Four ice cubes at exactly 0°C with a total mass of 51.0 g are combined with 120 g of water at 85 °C in an insulated container (AH-6.02 kJ/mol Cwater-4.18J/g. C) If no heat is lost to the surroundings, what is the final temperature of the mixture? Express your answer using two significant figures. 197| ΑΣΦ T= Submit Previous Answers Request Answer * Incorrect; Try Again ? ROVERW "C
Four ice cubes at exactly 0°C with a total mass of 51.0 g are combined with 120 g of water at 85 °C in an insulated container (AH-6.02 kJ/mol Cwater-4.18J/g. C) If no heat is lost to the surroundings, what is the final temperature of the mixture? Express your answer using two significant figures. 197| ΑΣΦ T= Submit Previous Answers Request Answer * Incorrect; Try Again ? ROVERW "C
Chapter10: Liquids And Solids
Section: Chapter Questions
Problem 102E: A 0.250-g chunk of sodium metal is cautiously dropped into a mixture of 50.0 g water and 50.0 g ice,...
Related questions
Question
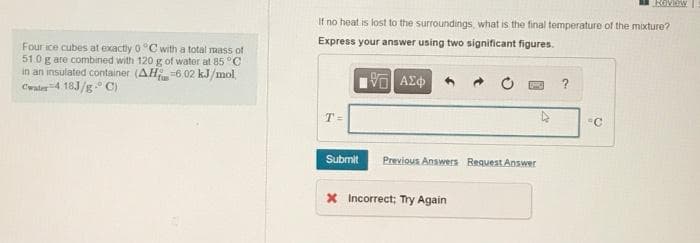
Transcribed Image Text:Four ice cubes at exactly 0°C with a total mass of
51.0 g are combined with 120 g of water at 85 °C
in an insulated container (AH-6.02 kJ/mol.
Cwater-4.18J/g. C)
If no heat is lost to the surroundings, what is the final temperature of the mixture?
Express your answer using two significant figures.
197| ΑΣΦ
T=
Submit
Previous Answers Request Answer
* Incorrect; Try Again
?
ROVERW
"C
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 6 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

World of Chemistry
Chemistry
ISBN:
9780618562763
Author:
Steven S. Zumdahl
Publisher:
Houghton Mifflin College Div

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning