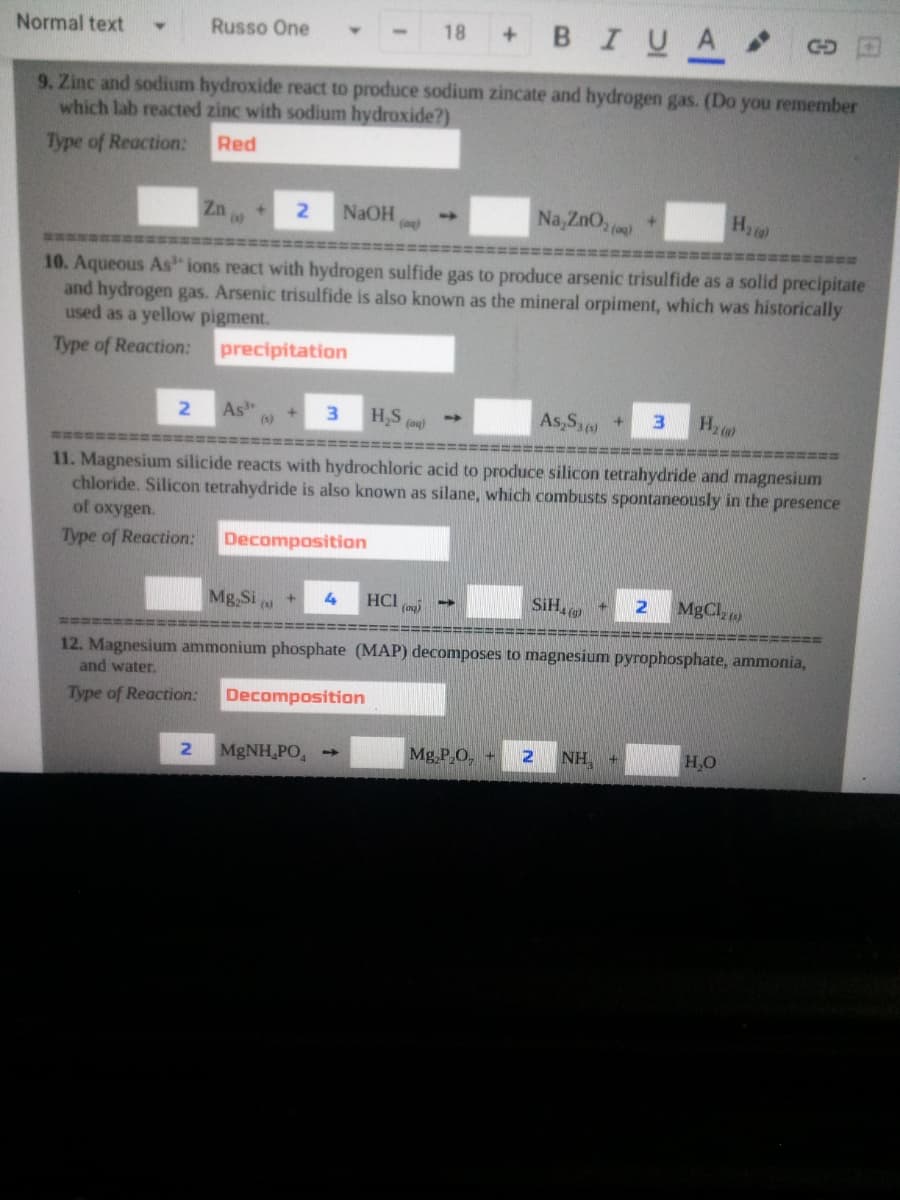
gas. (Do you remembe which lab reacted zinc with sodium hydroxide?) ype of Reaction: Red Z0 Na,ZnO, () NaOH +. 0. Aqueous As" ions react with hydrogen sulfide gas to produce arsenic trisulfide as a solid precipita and hydrogen gas. Arsenic trisulfide is also known as the mineral orpiment, which was historically used as a yellow pigment. Type of Reaction: precipitation As +] (1) 3. H,S As,S, As,S+ 3. (og) 11. Magnesium silicide reacts with hydrochloric acid to produce silicon tetrahydride and magnesium chloride. Silicon tetrahydride is also known as silane, which combusts spontaneously in the presence of oxygen. Type of Reaction: Decomposition Mg,Si HCI (o) SIH MgCl 12. Magnesium ammonium phosphate (MAP) decomposes to magnesium pyrophosphate, ammonia, and water. Type of Reaction: Decomposition 2. MgNH,PO, Mg,P,O, NH, H,O
gas. (Do you remembe which lab reacted zinc with sodium hydroxide?) ype of Reaction: Red Z0 Na,ZnO, () NaOH +. 0. Aqueous As" ions react with hydrogen sulfide gas to produce arsenic trisulfide as a solid precipita and hydrogen gas. Arsenic trisulfide is also known as the mineral orpiment, which was historically used as a yellow pigment. Type of Reaction: precipitation As +] (1) 3. H,S As,S, As,S+ 3. (og) 11. Magnesium silicide reacts with hydrochloric acid to produce silicon tetrahydride and magnesium chloride. Silicon tetrahydride is also known as silane, which combusts spontaneously in the presence of oxygen. Type of Reaction: Decomposition Mg,Si HCI (o) SIH MgCl 12. Magnesium ammonium phosphate (MAP) decomposes to magnesium pyrophosphate, ammonia, and water. Type of Reaction: Decomposition 2. MgNH,PO, Mg,P,O, NH, H,O
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter9: Chemical Quantities
Section: Chapter Questions
Problem 36QAP
Related questions
Question
Answer in the same form as of the question please
Transcribed Image Text:+BIUA
Normal text
Russo One
18
GD
9. Zinc and sodium hydroxide react to produce sodium zincate and hydrogen gas. (Do you remember
which lab reacted zinc with sodium hydroxide?)
Type of Reaction:
Red
Zn
Na, ZnO,
H
2.
NAOH
林性
10. Aqueous As ions react with hydrogen sulfide gas to produce arsenic trisulfide as a solid precipitate
and hydrogen gas. Arsenic trisulfide is also known as the mineral orpiment, which was historically
used as a yellow pigment.
Type of Reaction:
precipitation
As
(1)
H,S (ag)
As,Sa
Hz
3
11. Magnesium silicide reacts with hydrochloric acid to produce silicon tetrahydride and magnesium
chloride. Silicon tetrahydride is also known as silane, which combusts spontaneously in the presence
of oxygen.
Type of Reaction:
Decomposition
Mg,Si
HCI
(ag)
SiH.
MgCl,)
===
12. Magnesium ammonium phosphate (MAP) decomposes to magnesium pyrophosphate, ammonia,
and water.
Type of Reaction:
Decomposition
MgNH,PO,
Mg,P,O, +
NH,
H,O
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning