Q: Design a synthesis for the following transformation. Be sure to include all the reagents, solvents,…
A: Given that, a synthesis scheme is shown below We have to carry out the above synthesis.…
Q: What is pH of 0.05 M HCL
A:
Q: a. For the following balanced chemical equation, calculate how many grams of the products would be…
A:
Q: Draw the structure of the major and minor organic product(s); provide the reactants, or provide the…
A: Since you have asked a question with multiple subparts, we will answer the first three subparts for…
Q: In the reaction HSO4 (aq) + OH(aq) SO42 (aq) + H₂O(), the conjugate acid-base pairs are A. B. C. D.…
A: According to the Bronsted - Lowery concept of acid - base, " the Bronsted acids are those species…
Q: How long will it take for the concentration of A to decrease from 0.500 M to 0.155 M in the…
A: Given -> Initial concentration (a°) = 0.500 M Final concentration (at) = 0.155 M Rate constant…
Q: A sample of rock was found to contain 2.88 x 10-5 mol of 40K and 2.32 x 10-5 mol of 40 Ar. If we…
A: Given,Number of moles of K40 = 2.88 × 10-5Number of moles of Ar40 = 2.32 × 10-5All the Ar40 comes…
Q: 3. Complete the following zig-zag structure so that it represents (2R,3R)-3-chlorobutan-2-ol. H3C.…
A: R/S nomenclature are used to assign the configuration at chiral centers. According to R/S…
Q: Provide the chemical equilibrium that describes the carbonic acid buffer system that exists at pH…
A: Answer: Buffer solution is a type of solution that resists the change in its pH on adding small…
Q: Would more, or less, heat be released if the oxidant were Oz instead of N₂04? (not a thorough…
A: Oxidation is a chemical process in which a substance loses electrons, resulting in an increase in…
Q: Observe and collect the following data for following a sandwich recipe and making sandwiches! # of…
A: For scenario B Form scenario A it is clear that we need Bread and Cheese in the ratio of 2:1 ( 2…
Q: (CH₂)₂CHCH₂OC-OCH₂CH₂ H₂SO4 H₂O
A: In the given reaction (c) and (d) an ester reacts with H2SO4 followed by H2O i.e. acid hydrolysis of…
Q: 3. Provide a complete arrow pushing mechanism for the following transformation. H. HO 황 NaOH heat
A: Given that, a reaction scheme is shown below We have to provide a complete mechanism of 5he above…
Q: What is the pH of 0.0233 M solution of HNO₂? K = 5.35 x10 3. (Hint: 'x' is not small) O2.06 0.728 O…
A: Given that, Ka of HNO2= 5.35×10-3 HNO2=0.0233 M then, pH =?
Q: Given these reactions, X(s) + -- 0₂(g) — XO(s) XO(s) + CO₂(g) XCO3(s)- what is AH for this reaction?…
A: (2) Enthalpy Change: The enthalpy change for a chemical reaction can be equal to the sum or…
Q: 14.40 The equilibrium-constant expression for a reac- tion is K [NH₂][0₂] [NO]4[H₂O]6 What is the…
A: The equilibrium constant (Kc) is a numerical value that represents the ratio of the concentrations…
Q: HCIO is a weak acid (K,= 4.0 x 108) and so the salt NaCIO acts as a weak base. What is the pl in…
A: Given, Molarity of NaClO = C = 0.029 M Acid dissociation constant of HClO = Ka = 4.0 x 10-8 pH…
Q: Draw the product of this reaction. Ignore any counter ions or inorganic byproducts. CH3COCI NH₂ Q
A:
Q: 1) The Name of the following Compound is NH₂ exc "Br
A: Nomenclature of substituted benzene: 1) naming is done by placing the substituent as a prefix to the…
Q: Question # 5: 25.00mL of a sulfuric acid solution was standardized by titrating it with 0.2500 M…
A:
Q: ملة 3. C—CH+H−O−CH,CH, H' T
A: The given reaction is an example of the acid catalysed reaction of carboxylic acid and alcohol to…
Q: Identify the locants for the methyl groups in compound A. Explain why the locants are not 2, 4, and…
A:
Q: Consider a titration of 25 mL of 0.3 M NH3 using 0.1 M HCI. What is the pH-relevant component at the…
A:
Q: 2. Uranium-238 undergoes radioactive decay to form thorium-234 and an alpha particle (He). The…
A:
Q: Solvent is purged with N₂ to remove 0₂. What electrochemical process are we trying to eliminate by…
A: The question asks about the electrochemical process that we are trying to eliminate by purging a…
Q: Ⓒ Macmillan Learning Modify the structure to represent the organic product, if any, expected from…
A: An oxidizing agent is a chemical species that causes another chemical species to undergo oxidation,…
Q: CH3OH solvent HCl (catalyst)
A:
Q: Pressure and temperature affect the amount of space between gas molecules, which affects the volume…
A: Density is a physical property that relates to the mass of an object to its volume. It is defined as…
Q: following is a possible set of quantum numbers for a 3d electron? (n, l, ml, ms ) A) 3, 3, 2, +
A: The four quantum numbers are as follows: Principal quantum number (n): It defines the number of…
Q: Draw the structure(s) of the major organic product(s) of the following reaction. ui BrMg 1. Dry THF…
A:
Q: An aqueous solution contains 0.402 M hypochlorous acid. Calculate the pH of the solution after the…
A:
Q: Ⓒ Macmillan Learning Draw the structures of the carboxylic acid and the alcohol that produce the…
A: Esterification is the process of combining an organic acid (RCOOH) with an alcohol (ROH) to form an…
Q: What is Sº for B in the reaction 3A 2B if AS (rxn) =-221.8 J/mol K? [S° (A)= (205.0 J/mol .K)]
A: Given, 2 A ----> 2 B ∆S°rxn = -221.8 J/mol.K S°(A) = 205.0 J/mol.K S°(B) = ?
Q: 25) of what is the major product the following reaction он CH₂ CH3 HNO3 H2504 point t jass
A:
Q: Use the following information to answer the next question List of Organic Compounds 1. Ethane 2.…
A: Stronger the bond higher is the thermal stability.
Q: 18) which of the following s groups are ortho/ Para directers J.-NO₂. 1)--OCH 3 ID-CO₂ CH 3 IN - CH…
A: we have to select the groups that are ortho para directors
Q: a) Identify H and predict the products. ON CHO 4대ㅇ HCN Y li Al H3 #30+ ↑ b) Cive the mechanism of…
A:
Q: Cl 애 bryp C Pettog I ос o beioloo og с СсHздз Co н 3 23
A: The naming of organic compounds under IUPAC nomenclature is based on the structure of the molecule,…
Q: (a) Modify the given copies of the starting material to draw the aldol condensation product. Note:…
A:
Q: No explanation or anything needed just name the compounds (organic Chem)
A: Naming of organic compounds based on IUPAC nomenclature system. There are some important rules which…
Q: 2. Complete the following statements: agreb a. As hydronium ion concentration increases, pH…
A: Note: As per the guidelines solution of question-2 has been made. For the expert solution of 2nd…
Q: 2-bromopropane Draw the molecule on the canvas by choosing buttons from the Tools (for bonds),…
A: To draw any organic compound from its IUPAC name, we need to identify the functional group first,…
Q: The reaction was studied at 25 °C and the following results were obtained: a) determine the rate…
A: we have to determine the rate law of the reaction
Q: Provide a synthesis for the given target molecule, starting with an alkyl halide of your choice.…
A: Primary or secondary halide generally undergoes substitution with strong nucleophile by SN2…
Q: Give the starting material and mechanism arrows needed to produce the given product by coordination.…
A: organic reaction mechanism.
Q: Consider a solution of 2.0 M HCN and 1.0 M NaCN (Ka for HCN=6.2x10^-10). Which of the following…
A: Answer: Buffer solution is a type of solution that resists the change in its pH on adding small…
Q: Choose the best description for the selectivity/specificity of the transformation shown below: Br…
A:
Q: [Review Topics] [References] Use the References to access important values if needed for this…
A: Given Concentration of acetic acid = 0.370 M Volume = 150 ml pH = 4.4 Concentration of NaOH = 0.361M…
Q: All of the following contain a sp-hybridized functional group except a carboxylic acid. a nitrile.…
A: Hybridization is a process of combining two atomic orbitals for the generation of new degenerate…
Q: Part A ΔΗ 131 kJ , ΔSx = – 250 J/K , T = 295 K . Express your answer in joules per kelvin as an…
A: The formula to find the entropy of surroundings ∆Ssurr is: ∆Ssurr=-∆HrxnoT The formula to find the…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

- Define what an organometallic compound is. What types of chemical connections do organometallic compounds have? Exemplify each type.Draw and name all of the structural isomers possiblefor an alkyl halide with no branches and the molecularformula C 5 H 10 B r 2Write the structures of the products when Butan-2-ol reacts with the following:(a) CrO3(b) SOCl2
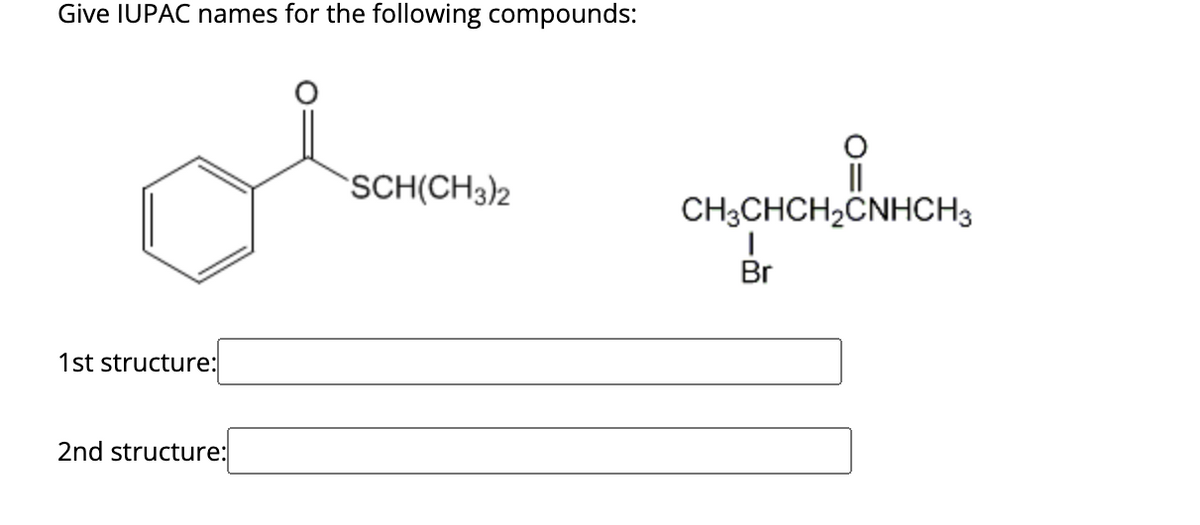
- the number of elements of unsaturation in the molecular formula C4H6. Giveall nine possible structures having this formula. Remember thata double bond = one element of unsaturationa ring = one element of unsaturationa triple bond = two elements of unsaturationidentify the following organic compound according to its class. 1.) CH3COOH 2.) CH3CH2CH2CH3 3.) CH3CHO 4.) CH3CH2CL 5.) CH3OCH3Give IUPAC names for the following compounds:
- A 2-bromobutane react with methanol and form a enantiomeric pair of 2-methoxybutane. Draw the structures of the enntiomeric pairs of ethers.Under certain reaction conditions, 2,3-dibromobutane reacts with two equivalents of base to give three products, each of which contains two new p bonds. Product A has two sp hybridized carbon atoms, product B has one sp hybridized carbon atom, and product C has none. What are the structures of A, B, and C?Draw the structure of the major product of benzene + CH3CH2CH2Cl in AlCl3
- Write equations for the reaction of cyclohexane with:a. Br2/H2O indoorsb. Br2/H2O in the presence of sunlight.Give a common name (when possible) and a systematic name for each compound. ) (h) CH3C‚CCH2OCH3Draw the structure of a molecule that fi ts each description: a. a 2 ° alcohol of molecular formula C 6H 12O b. a cyclic ether with molecular formula C 5H 10O c. a 1 ° alkyl halide with molecular formula C 5H 11Cl

