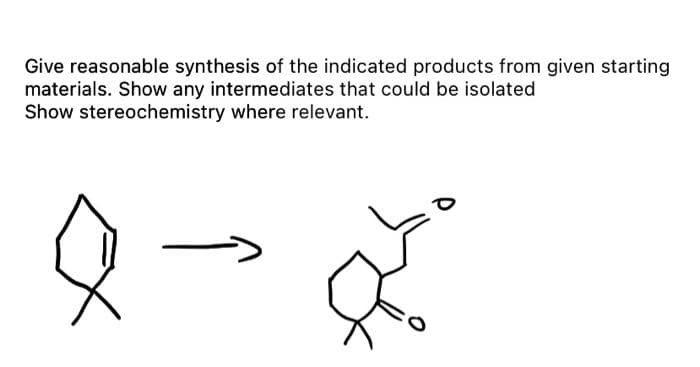
Give reasonable synthesis of the indicated products from given starting materials. Show any intermediates that could be isolated Show stereochemistry where relevant. Ž
Q: Calculate the entropy of each of the following states. (Figure 1) Figure (a) (b) (c) 1 of 1 ▼ Part A…
A:
Q: Solvent is purged with N₂ to remove 0₂. What electrochemical process are we trying to eliminate by…
A: The question asks about the electrochemical process that we are trying to eliminate by purging a…
Q: Pressure and temperature affect the amount of space between gas molecules, which affects the volume…
A: Density is a physical property that relates to the mass of an object to its volume. It is defined as…
Q: 9:23 PM Tue Mar 28 ← :0: Y Select to Add Arrows CH3CH2ONa, CH3CH2OH :0: Problem 95 of 20 Please…
A: The mechanism of a chemical reaction shows the detailed steps involved in it. The abstraction of…
Q: An aqueous solution contains 0.243 M hydrosulfuric acid and 0.134 M hydroiodic acid. Calculate the…
A: Hydrosulfuric acid (H2S) is a weak, diprotic acid and it dissociates partially in the solution. Its…
Q: Select the mechanism that has all curved arrows drawn correctly for the transformation shown below.…
A: In chemistry A bond is formed by donating a electron pair by a electron rich species called…
Q: HH ✡ :0: Select to Add Arrows H H Select to Add Arrows CH3CH₂ONA, CH3CH₂OH :0: CH3CH₂ONA, CH3CH2OH…
A: A reaction scheme is given. We have to draw the arrow pushing mechanism.
Q: Aldol condensation Indicate the products: CH3-CO-CH3 + CH3-CH2-CHO + NaOH Homo-aldol and…
A: Aldol condensation is a type of organic reaction that involves the condensation of two carbonyl…
Q: This dienol readily reacts under acidic condiuchs. Draw the product of this reaction. Include all…
A: Alcohol becomes dehydrated when it combines with protic acid to lose water molecules while being…
Q: 2C1, (g) + Sn(s) SnC1, (s) C(s) + O₂(g) → CO₂ (s) redox reaction? yes reducing agent: oxidizing…
A:
Q: Ⓒ Macmillan Learning Draw the organic product of the reaction. Include all hydrogen atoms in the…
A:
Q: What is the IUPAC name of each of the the following? CH3CH₂CH₂CH₂CH₂CNH₂ CH3CH₂CH₂CH₂CH₂CN NOHSCHI…
A:
Q: Cl 애 bryp C Pettog I ос o beioloo og с СсHздз Co н 3 23
A: The naming of organic compounds under IUPAC nomenclature is based on the structure of the molecule,…
Q: For the reaction 4HCl(g) + O₂(g) → 2H₂O(g) + 2Cl₂ (g) AH° = −114.4 kJ and AS° = 128.9 J/K The…
A: Answer: For any reaction sign of the value of Gibbs free energy change decides whether reaction is…
Q: Determine the product(s) formed when cyclohexene is treated with the following reagents. Br Br Br OH…
A: Cyclohexene + HBr ----> bromocyclohexane cyclohexane + Br2/H2O -----> halohydrin derivative of…
Q: 2N₂O(g) 2N₂(g) + O₂(g) k = 1 x 10³5 a. Write the equilibrium constant expression. b. State which…
A: To solve this problem we have to write equilibrium constant expression.
Q: A student determines the value of the equilibrium constant to be 8.55×10-31 for the following…
A:
Q: 3. Complete the following zig-zag structure so that it represents (2R,3R)-3-chlorobutan-2-ol. H3C.…
A: R/S nomenclature are used to assign the configuration at chiral centers. According to R/S…
Q: • In an analysis of inter halogen reactivity, 0.500 mol ICI (iodine chloride) was placed in a 5.00 L…
A:
Q: Draw the following organic compounds. You may choose to draw it as a full, condensed or line…
A: The IUPAC names contain Prefix + parent carbon + suffix Here, parent carbon chain is the longest…
Q: 3. Match the term on the left with the most appropriate description on the right. galvanic cell…
A: 1) Galvanic cell - An electrochemical cell that produces electricity. 2) Electrolytic cell - A…
Q: Which statements about primary batteries are correct? Select all that apply: "C" and "D" batteries…
A: Inroduction :- In Galvanic cell chemical energy is converted into electrical energy. i.e.…
Q: a) Stearic acid and oleic acid both have 18 carbons, but stearic acid melts at 71 °C, while oleic…
A:
Q: What is the equilibrium constant for the solubility of Ag₂CO₃ (Ksp = 8.1 × 10⁻¹²) in NH₃? (Kf of…
A: To solve this problem we have to calculate the equilibrium constant.
Q: a. For the following unbalanced equation, indicate how many moles of the first product are produced…
A:
Q: (CH,),CHCH,COOH + CH,CH(CH,)NH, T Reaction B Select Draw Templates More / ||||||| C H N O Q2Q Erase
A: When amine react with carboxylic acid then Amide and water is formed as product.
Q: Consider the reaction: 2H₂O2(1)→ 2H₂O(1) + O2(g) Using standard thermodynamic data at 298 K,…
A: Answer: Standard Gibbs free energy change for the reaction is equal to the difference between…
Q: 12.94 *How many grams of NaOH can react with 6.22 g of succinic acid? The structure of succinic acid…
A:
Q: Br. Br Answer H-C=C-H H-CC-H wwwww HCC H www. i) Excess NaNH. 2) EICI 3) H₂, Lindlar's catalyst 17…
A: Given reactants and reagents of different reactions
Q: The cell reaction for a dry cell battery is Zn(s) + 2 MnO2(s) + 2 NH4+(aq) → 2 NH3(aq) + Mn2O3(s) +…
A:
Q: 9:13 PM Tue Mar 28 Draw the major product of this Dieckmann condensation reaction. Ignore inorganic…
A: We know sodium ethoxide acts as a base and abstract the acidic proton of ketone leading to…
Q: CH3- -CH- CH3 -H Ag₂0 NH,OH, H2O
A: We know addition of oxygen is known as oxidation . Here ammoniacal solution of silver oxide acts as…
Q: Calculate the mass percent (m/m) of a solution prepared by dissolving 56.89 g of NaCl in 158.6 g of…
A:
Q: 2. Use the following chemical equation to answer the following question. UO₂+ (aq) + CH4 UO2 (s) +…
A:
Q: An aqueous solution contains 0.367 M hypochlorous acid. How many mL of 0.224 M potassium hydroxide…
A: It is based on the concept of buffer. Here we are required to find the volume of 0.224 KOH that need…
Q: Using the heat of fusion of acetic acid, 192 J/g, calculate the grams of acetic acid that will…
A: Given that The heat of fusion of acetic acid, ∆Hfusion = 192 J/g, The amount of acetic acid in…
Q: List the following types of electromagnetic radiation in decreasing wavelength. So, place a 1 by the…
A: The question is based on the concept of electromagnetic radiation. we need to arrange given rays…
Q: An aqueous solution contains 0.243 M hydrosulfuric acid and 0.134 M hydroiodic acid. Calculate the…
A: Hydrosulfuric acid H2S is a diprotic acid and it dissociates partially in the solution. Its acid…
Q: Macmillan Learning Write the systematic (IUPAC) names for the molecules. CH3 H3C IUPAC name: H₂C…
A:
Q: ISSED THIS? Read Section 1 7.5 (Pages 278-280) Watch (WE 7.5 When 0.445 of biphenyl (C₁3H10)…
A: Given, Mass of biphenyl (C12H10) = 0.445 g Initial temperature = Ti = 25.3 ∘C Final temperature =…
Q: Which correctly labels the Brønsted-Lowry acid, its conjugate base, the Brønsted-Lowry base, and its…
A:
Q: 3-ethyl-2-methylheptane Draw the molecule on the canvas by choosing buttons from the Tools (for…
A: Chemical structure refers to the arrangement of atoms, bonds, and functional groups in a molecule.…
Q: 5. Substitution vs. Elimination? Predict which mechanism will these reactions go through (SN2, SN1,…
A: To decide the substitution or elimination mechanism through which a reaction proceeds. The reaction…
Q: 2. b. Balance the following redox reactions using the half-reaction method. H₂O2 + Cr₂O7 → O₂+ Cr+³…
A: A redox reaction is balanced through the half-reaction method through following steps: The…
Q: 2. Uranium-238 undergoes radioactive decay to form thorium-234 and an alpha particle (He). The…
A:
Q: The element calcium is often used in fireworks to create an orange color. The particular color of…
A:
Q: Draw the major product of this reaction. Ignore inorganic byproducts. CI O excess CH3NH2 o
A: The given reaction is the example of nucleophilic attack of methyl amine to the carbonyl carbon of…
Q: An aqueous solution contains 0.444 M hypochlorous acid. Calculate the pH of the solution after the…
A: we have to calculate the pH of the solution
Q: A buffer solution contains 0.213 M NH3 and 0.265 M NH4Cl. What is the pH of the buffer solution…
A: 26.9 mL of 0.125 M NaOH was added to a buffer solution containing 0.213 M NH3 and 0.265 M NH4Cl.…
Q: CaC2 (s) + 2 H₂0 (1) → Ca(OH)2(aq) 2 Ca(OH)2(aq) + C₂H₂(g) AH = ??? Ca(s) + 2 C(graphite) → CaC2(s)…
A:
Step by step
Solved in 2 steps with 3 images

- Show how the following starting materials are converted to the givenproduct by a series of two pericyclic reactions. Account for the observedstereochemistry.Give the major product of the following reactions with the appropriate regiochemistry and steriochemistry.give the major product of the following reactions with the appropriate stereochemistry and regiochemistry.
- give the major products and include stereochemistry when appropriateProvide reagents/conditions to accomplish the following syntheses.Show how you would synthesize the following compounds, starting with benzene or toluene and any necessary acyclicreagents. Assume para is the major product (and separable from ortho) in ortho, para mixtures. p-aminobenzoic acid
- Show how you would synthesize the following compounds, starting with benzene or toluene and any neccessary reagents. Assume para is the major product (and separable from ortho) in ortho, para mixtures. ( A and B)Provide reagents/conditions to accomplish the following syntheses. Several steps are required in some cases.The following compound undergoes Benzilic Acid Rearrangementto yield ahydroxyacid salt. Proposea mechanism for the reaction, writethe major product,and provide an explanation as tothe preference of migration of one R group over the other.

