Q: Q/ The intensity of the incident light radiation is reduced by 20% when it passes through a 1 cm…
A: The Beer-Lambert law : It states that for a given sample the path length and concentration of the…
Q: The freezing point of pure benzene (CH) is 5.49 °C. The freezing point of a solution made using…
A:
Q: Nitrogen monoxide, NO, reacts with bromine, Br₂, to give nitrosyl bromide, NOBr. 2NO(g) + Br₂(g) →…
A:
Q: Please answer all of the following neatly clearly and thoroughly! Thanks Consider the given reaction…
A: Le Chatelier’s principles : In reversible reactions When factors like concentration, pressure,…
Q: Kp for the following reaction at a give temperature is 0.263 at 1000 K. C(s) + 2 H₂(g) CH4(g) What…
A:
Q: 02A: 2.1-2.5 and Accuracy Handout (70 min) culate Figures of Merit for Trueness ▾ Part A. Absolute…
A: Given data is The concentration of standard ethyl alcohol sample = 0.174 mg/L The average value of…
Q: Express the rate of the reaction 2NO(g) + O₂(g) → 2NO₂ (g) a. in terms of A[NO₂]. rate = b. in terms…
A: Given -> 2NO(g) + O2(g) ---> 2NO2(g)
Q: 1.48. Use the approximation (1-x)¹1 + x + x² + to determine an expression for the virial coefficient…
A:
Q: Use the data to determine the entropy change ASrxn for this process: NH3(aq) + HOCl(aq) = NH₂Cl(aq)…
A: Given -> NH3(aq) + HOCl(aq) ---> NH2Cl(aq) + H2O(l)
Q: What is the effective mass yield for the reaction shown? The density of ethyl L-lactate is 1.03…
A: We are given an organic synthesis reaction and we have to calculate the effective mass yield of it.
Q: 3. 2. нс. нс- н.с. Н.С- ostwork CH Complete the statement: "The reason organic acids and bases can…
A: Note: Since you have posted a question with multiple sub-parts, we will provide the solution only to…
Q: Draw the structures of the following Alkynes: 2,5-Dimethyl-3-hexyne Ethyne 1-Propyne…
A: Alkynes are a class of hydrocarbons that contain a carbon-carbon triple bond. The basic formula for…
Q: Assume the half-life of a substance is 29 days and the initial amount is 139.399999999998 grams. (a)…
A: Given data is The half life of the substance = 29 days Initial amount of the substance =…
Q: What is the pH of a solution containing 0.10 M cyanide ion, CN1-? CN1- + H2O HCN + OH1- Kb of…
A: According to the question, The concentration of the CN- is = 0.10 M The Kb of CN- ion = 1.6 x 10-5…
Q: A student performed three aqueous layer titrations in Part 1 using a standard sodium thiosulfate…
A: Given data is Molarity of sodium thiosulphate solution = 0.01155 M Final burette reading of…
Q: An ethylene glycol solution contains 30.4 g of ethylene glycol (C2H6O2) in 94.4 mL of water. (Assume…
A:
Q: Calculate the effusion rate of He atoms through a 1.0 mm circular aperture (e.g., a pinhole in a…
A: Given, Pressure=5.0 atm Temperature=298 K Diameter of circular aperture=1.0 mm Molar mass of…
Q: 4. COOH COOH
A: The given reaction is Diels-Alder reaction.this is also called (4+2) cycloaddition reaction.
Q: D. Give the quantum numbers for the last electron of each element given in Part II following the…
A: Since, For representation of electron in an orbital we use four type of quantum number-- 1)Principle…
Q: Q/A transparent medium has a refractive index of 1.5 and the wavelength of the magnetic ray passing…
A:
Q: Starting from benzene, how will you prepare the target? Show all the steps in the correct order. NH₂…
A:
Q: D. Give the quantum numbers for the last electron of each element given in Part II following the…
A:
Q: The elements nickel (Ni) and boron (B) form two binary compounds with the following compositions:…
A: Here two elements Ni and B combine with each other to form two compound. Grams of Ni per grams of B…
Q: 1.34. A derivative with respect to the same variable can be taken more than once: (a) and is called…
A: The given terms are We have to find the expression of the following terms for the ideal gas…
Q: True/False : ______ Zaitsev’s Rule says the most substituted double bond is the favored product in…
A:
Q: Please modify this drug (tamoxifen) to make it water soluble and easy to be absorbed in stomach? mal…
A: In the given molecule we have to tell that ,how this compound can be easily soluble in water and…
Q: H3C. ОН CH3 H3C. nigh you use on low CH₂ Grignard
A:
Q: ОН н ОН н H₂N "I 5 - 10 min 22 °C Acetic Acid Acetic Anhydride heat ОН Н NaBH4 Ethanol 22°C ОН н H2O
A: The given reaction is a condensation reaction followed by acetylation on the amine group. The first…
Q: The following information is given for mercury, Hg, at latm: AHvap(357 °C) = 59.3 kJ/moi boiling…
A: Answer: This question is based on the phase transition where gaseous mercury is being condensed into…
Q: 7. Using the bond dissociation energies provided below, draw an energy diagram for the two…
A: Suppose a reaction A2 + B2 –> C2 Then ΔHrxn given as ΔHrxn = Σ Bond energy of reactants -Σbond…
Q: Consider the heating curve provided for 1.00 mole of a substance that begins as a solid at -14.8 °C…
A: The specific heat capacity of the liquid is 97.25 J/mol.oC, which states that 97.25 J of heat…
Q: Name and draw a structural formula for the major product of each alkene addition reaction: CH 3 (a)…
A: In the given question, two addition reactions are given and it is asked to write the name and draw…
Q: Then, rank the solids in order of decreasing melting point. That is, select "1" next to the solid…
A: This question belongs to Solid State Chemistry.
Q: Which of the following types of stretches are shown in this spectrum ?
A: A question based on IR spectroscopy. An IR spectrum of an unknown organic compound is prescribed for…
Q: 1.72. Calculate the average grade using equation 1.32 if the scores on a 10-point quiz are 5, 5, 5,…
A: Since, Average of the given sample or data is calculated as sum of all the data divided by the…
Q: Iodine and bromine react to give iodine monobromide, IBr. I2(g) + Br2(g) 21Br(g) What is the…
A: The given equilibrium reaction is as follows: I2(g) + Br2(g) ⇌ 2IBr (g) Kc of reaction = 55.8…
Q: The boiling point of water is 100.00 °C at 1 atmosphere. A student dissolves 13.53 grams of…
A:
Q: Part A 0 H-C-O-CH₂-CH₂ Enter the name of the carboxylic acid first, then the name of the alcohol.…
A: As per the answer guidelines i have to answer only 1 subpart of the particular question until…
Q: Calculate the freezing point of a solution containing 11.4 g FeCl3 in 180 g water. Calculate the…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: What is the name of the molecule shown below? Do not add spaces or parentheses in your name. Include…
A: IUPAC nomenclature is used for naming the organic compound as recommended by international union of…
Q: Data for the following reaction are given in the table below. CO(g) + NO₂(g) → CO₂(g) + NO(g) Trial…
A: Suppose a reaction M +N –> O Then rate law equation of this reaction given as Rate = k[M]a[N]b…
Q: which one is correct: a) The entropy change of the system can be negative in a spontaneous process.…
A: Entropy is a thermodynamic property that quantifies the amount of thermal energy in a system that is…
Q: Match the reagents to the reaction (place letter on reaction arrow) Only letters C. & D. Are left to…
A: In the given reaction the hydroxy group is substituted by the chloride ion from the back side…
Q: For each of the following reactions, choose which expression represents Ke. a. TIC13 (8) TICI(s) +…
A: We have been given three reactions and we have been asked to choose expression that represent Kc…
Q: H₂ H₂ (a) H₂C=C-C-C-CH3 1-pentene (b) (c) (d) H3C C C CH3 ΤΗ CH3 2-butene cyclobutene…
A: The structures of the given molecules are
Q: Hydrocarbon Reactions mayd •*-||||||| HHHHHHH HH CH₂ C10H22 (1)→ C2H6 (9) + C7H16 (1) + C(s)
A: 1.Cracking in chemistry refers to the process of breaking down large hydrocarbon molecules into…
Q: The density of helium gas under certain conditions is found to be 0.164 kg/m³. What is the volume in…
A:
Q: A solution of CaCl₂ in water forms a mixture that is 35.0% calcium chloride by mass. If the total…
A: According to the question, The solution of CaCl2 = 35.0 % by mass The total mass of the…
Q: H₂CH₂C H H product 1: product 2: CH₂CH₂ HCI This equation shows the reaction of…
A:
Q: Based on the nature of reactants and products, determine which of the following potential energy…
A: To determine the nature of the reaction i.e Exothermic or endothermic based on the potential diagram…
Step by step
Solved in 3 steps with 2 images

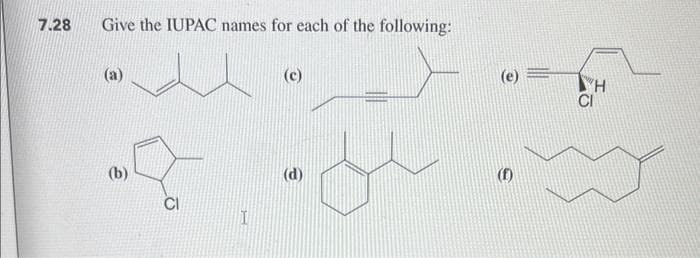
- Give the series of reactions below, identify and give the iupac name for the following compounds, in short identify and name A,B,CAnswer the following Compounds CORRECTLY by giving the IUPAC NAME OF IT. •Label with proper notations (like hyphen, commas, periods, and grouping symbols) thank u❤️1.- a) Write the reaction to obtain aromatic polyamides (Aramids), include the molecular structure of Kevlar (p-substituted) and Nomex (m-substituted). b) Which one of the isomers do you expect to have higher crystallinity and why (what intermolecular forces cause this)? c) Which one do you expect to have more chain flexibility and why (although is quite limited on both)? d) Describe the process to obtain Kevlar fibers industrially.
- Which of the following best estimates the percentages of the three isomeric methylanilines from the reaction shown in Image 10? a. B b. A c. D d. CGiven the solution to Example 8.5, predict the structure of the product(s) formed when 3-hexene is treated with NBS.Give the IUPAC name for the compound below, including the configurational labels E or Z as appropriate
- Using the information provided below, deduce the identity of the compound I, II, III, IV and V. What is the IUPAC name of compound I, II, III, IV and V?In the following three compounds(1,2,3) arrange their relative reactivity towards the reagent CH3Cl / AlCl3. and justify it.question 1 i) Explain the acidity of terminal alkanes, alkenes and alkynes. ii) Compound A has the formula C10H16. On catalytic hydrogenation over palladium, it reacts with only 1 molar equivalent of H2 to yield C. Compound A also undergoes reaction with ozone, followed by zinc treatment, to yield a symmetrical diketone, B (C10H16O2). By showing the reaction steps, predict the structures of A, B and C. iii) Show the reaction mechanism of cyclohexene with Br2.
- An optically active unknown compound B, whose molecular formula is C6H10, reacts with H2/Ni produce compound C, whose molecular formula is C6H14. Compound C is optically inactive. What are the structures of compound B and C ?What is the IUPAC for each compound. Specify E/Z if relevantOct-1-yne (HC≡CCH2CH2CH2CH2CH2CH3) reacts rapidty with NaH, forming a gas that bubbles out of the reaction mixture, as one product. Oct-1-yne also reacts rapidty with CH3MgBr, and a different gas is produced. Write balanced equations for both reactions and identify the gases formed.

