Given that Ka’s for hydrofluoric acid (HF) and boric acid (H3BO3) are 6.3 × 10^–4 and 5.4 × 10^–10, respectively, calculate the pH of the following solutions: (a) The mixture from adding 50 mL 0.2 M HF to 50 mL 0.5 M sodium borate (NaH2BO3). (b) The mixture from adding an additional 150 mL 0.2 M HF to the solution in (a), i.e., a total of 200 mL 0.2 M HF was added to 50 mL 0.5 M NaH2BO3.
Given that Ka’s for hydrofluoric acid (HF) and boric acid (H3BO3) are 6.3 × 10^–4 and 5.4 × 10^–10, respectively, calculate the pH of the following solutions: (a) The mixture from adding 50 mL 0.2 M HF to 50 mL 0.5 M sodium borate (NaH2BO3). (b) The mixture from adding an additional 150 mL 0.2 M HF to the solution in (a), i.e., a total of 200 mL 0.2 M HF was added to 50 mL 0.5 M NaH2BO3.
Chemistry: Principles and Practice
3rd Edition
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Chapter16: Reactions Between Acids And Bases
Section: Chapter Questions
Problem 16.48QE
Related questions
Question
Given that Ka’s for hydrofluoric acid (HF) and boric acid (H3BO3) are 6.3 × 10^–4 and 5.4 × 10^–10, respectively, calculate the pH of the following solutions:
(a) The mixture from adding 50 mL 0.2 M HF to 50 mL 0.5 M sodium borate (NaH2BO3).
(b) The mixture from adding an additional 150 mL 0.2 M HF to the solution in (a), i.e., a total of 200 mL 0.2 M HF was added to 50 mL 0.5 M NaH2BO3.
Expert Solution
Step 1

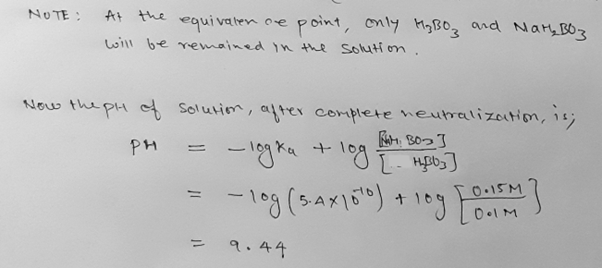
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning