Given the balanced equation with an unknown compound represented by X, which compound is represented by X? * enzyme C6 H12O6 (aq) 2X + 2CO2 (g)
Given the balanced equation with an unknown compound represented by X, which compound is represented by X? * enzyme C6 H12O6 (aq) 2X + 2CO2 (g)
Human Physiology: From Cells to Systems (MindTap Course List)
9th Edition
ISBN:9781285866932
Author:Lauralee Sherwood
Publisher:Lauralee Sherwood
Chapter3: The Plasma Membrane And Membrane Potential
Section: Chapter Questions
Problem 1SQE: Using the Nernst equation, calculate the equilibrium potential for Ca2 and for C1 from the following...
Related questions
Question
100%
Given the balanced equation with an unknown compound represented by X, which compound is represented by X?
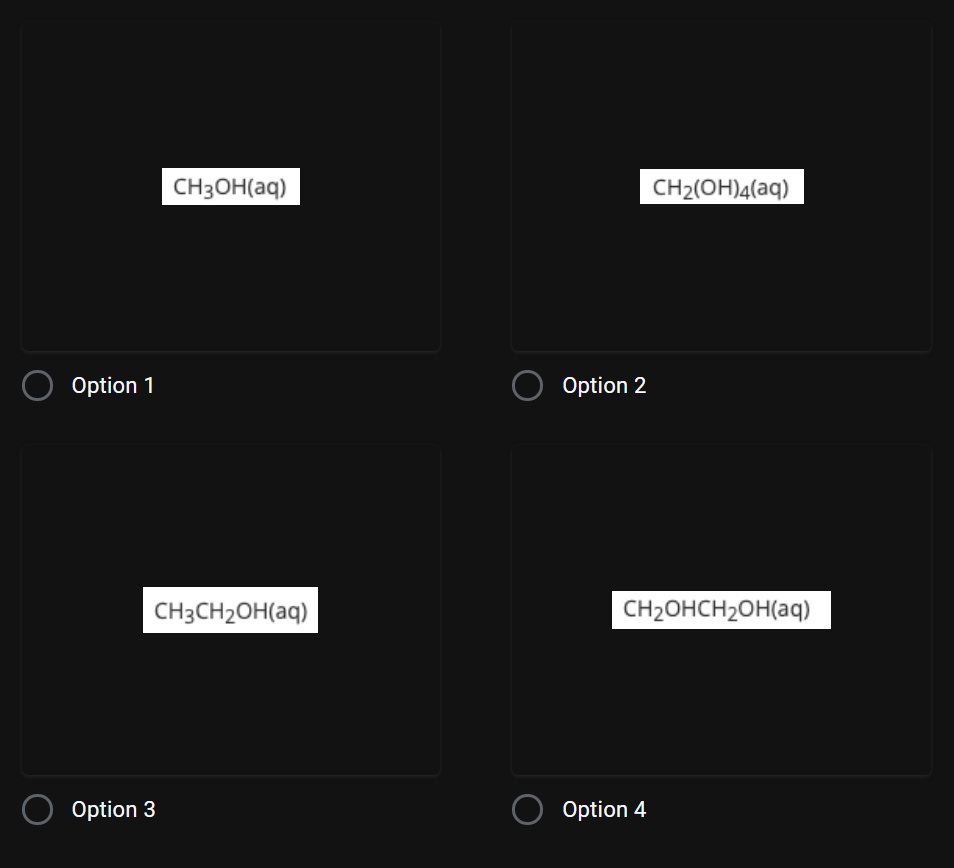
Transcribed Image Text:CH3OH(aq)
CH2(OH)4(aq)
Option 1
Option 2
CH3CH2OH(aq)
CH2OHCH2OH(aq)
Option 3
Option 4
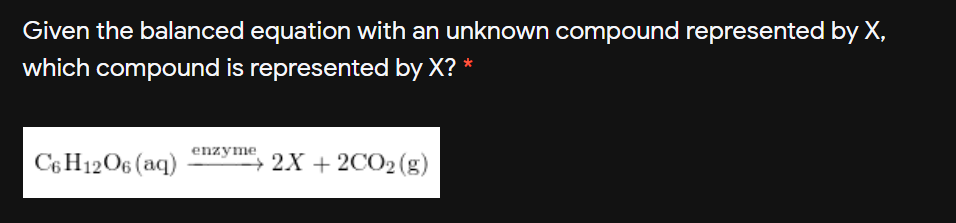
Transcribed Image Text:Given the balanced equation with an unknown compound represented by X,
which compound is represented by X? *
C6 H12O6 (aq)
enzyme
+ 2X + 2CO2 (g)
Expert Solution
Step 1
Sugars such as glucose, sucrose, fructose, etc., can be converted to alcohol (ethanol) by a biochemical process called ethanol/alcohol fermentation. Alcohol fermentation occurs in yeast in the absence of oxygen. Hence, this process is called an anaerobic process.
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Human Physiology: From Cells to Systems (MindTap …
Biology
ISBN:
9781285866932
Author:
Lauralee Sherwood
Publisher:
Cengage Learning

Human Physiology: From Cells to Systems (MindTap …
Biology
ISBN:
9781285866932
Author:
Lauralee Sherwood
Publisher:
Cengage Learning