Given the compound: CI CI a. Provide the chemical formula of the molecule. Note that there are hydrogen atoms as part of the formula but these are not shown in the drawing. b. The picture does not show lone pairs of electrons. Redraw the structure and indicate where all lone pairs are located. Include all hydrogen atoms in your drawing. Note that there are 42 valence electrons.
Given the compound: CI CI a. Provide the chemical formula of the molecule. Note that there are hydrogen atoms as part of the formula but these are not shown in the drawing. b. The picture does not show lone pairs of electrons. Redraw the structure and indicate where all lone pairs are located. Include all hydrogen atoms in your drawing. Note that there are 42 valence electrons.
Living By Chemistry: First Edition Textbook
1st Edition
ISBN:9781559539418
Author:Angelica Stacy
Publisher:Angelica Stacy
ChapterU2: Smells: Molecular Structure And Properties
Section: Chapter Questions
Problem 6RE
Related questions
Question
100%
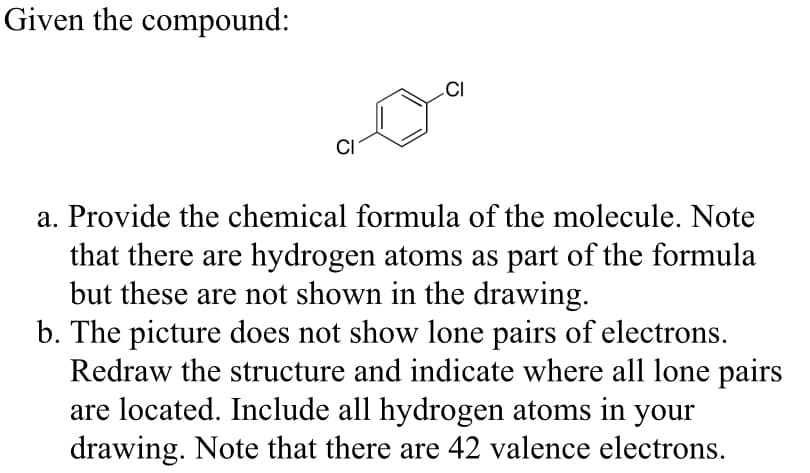
Transcribed Image Text:Given the compound:
CI
CI
a. Provide the chemical formula of the molecule. Note
that there are hydrogen atoms as part of the formula
but these are not shown in the drawing.
b. The picture does not show lone pairs of electrons.
Redraw the structure and indicate where all lone pairs
are located. Include all hydrogen atoms in your
drawing. Note that there are 42 valence electrons.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:
9781559539418
Author:
Angelica Stacy
Publisher:
MAC HIGHER


Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:
9781559539418
Author:
Angelica Stacy
Publisher:
MAC HIGHER


Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning