Given the PV diagram of a fixed quanity of CO2 assumed to be an ideal gas. The temperature at point a A is 35 degrees celsius. What is the total mass of CO2? What ist he temperature at point C in degrees celsius? What is the heat added to the gas over the ABCA cycle? If 3200 J of heat flows out of the system when the system is taken in a straight path from C to A, what is the change in enteral energy along that path?
Given the PV diagram of a fixed quanity of CO2 assumed to be an ideal gas. The temperature at point a A is 35 degrees celsius. What is the total mass of CO2? What ist he temperature at point C in degrees celsius? What is the heat added to the gas over the ABCA cycle? If 3200 J of heat flows out of the system when the system is taken in a straight path from C to A, what is the change in enteral energy along that path?
University Physics Volume 1
18th Edition
ISBN:9781938168277
Author:William Moebs, Samuel J. Ling, Jeff Sanny
Publisher:William Moebs, Samuel J. Ling, Jeff Sanny
Chapter14: Fluid Mechanics
Section: Chapter Questions
Problem 53P: Verigy that the SI of hpg is N/m2.
Related questions
Question
Given the PV diagram of a fixed quanity of CO2 assumed to be an ideal gas. The temperature at point a A is 35 degrees celsius. What is the total mass of CO2? What ist he temperature at point C in degrees celsius? What is the heat added to the gas over the ABCA cycle? If 3200 J of heat flows out of the system when the system is taken in a straight path from C to A, what is the change in enteral energy along that path?
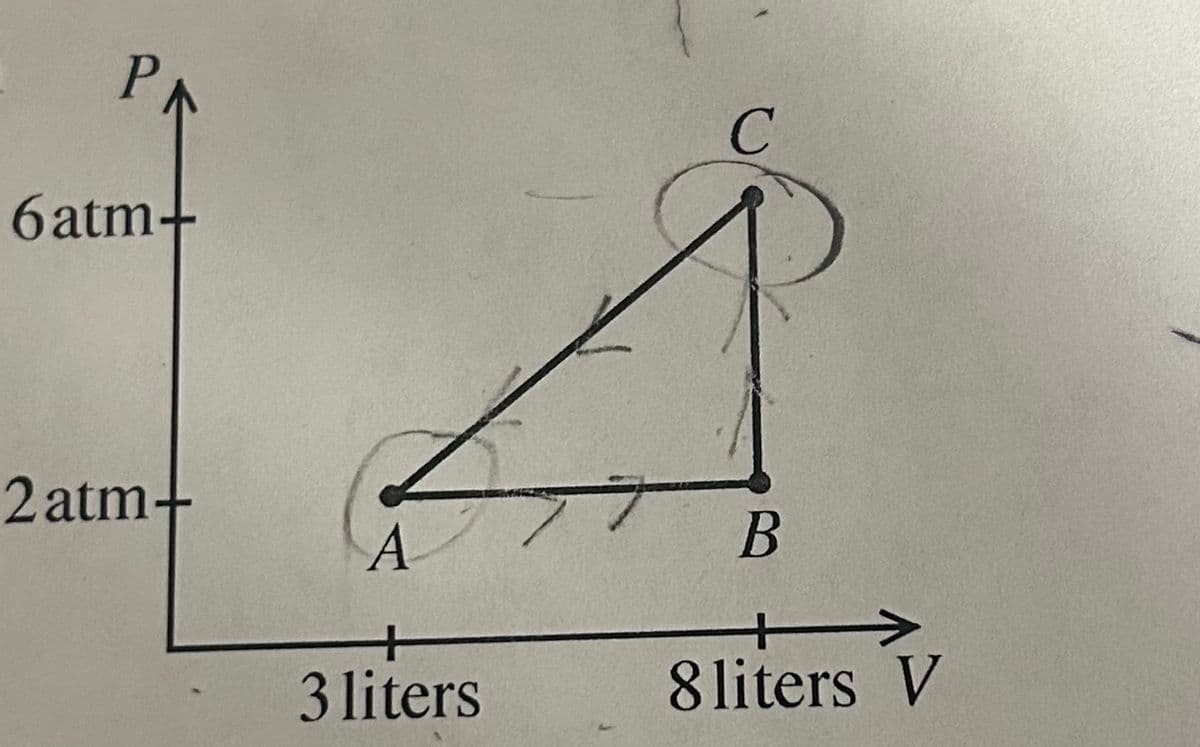
Transcribed Image Text:PA
6atm+
2 atm+
2
A
3 liters
C
B
8 liters V
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Step 1: Determine the given variables.
VIEWStep 2: Part (i) - Determine the total mass of the gas
VIEWStep 3: Part (ii) - Determine the temperature at C in degree celsius
VIEWStep 4: Part (iii) - Determine the heat added to the gas in a complete cycle.
VIEWStep 5: Part (iv) - Determine the change in internal energy for C to A
VIEWSolution
VIEWTrending now
This is a popular solution!
Step by step
Solved in 6 steps with 6 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you

University Physics Volume 1
Physics
ISBN:
9781938168277
Author:
William Moebs, Samuel J. Ling, Jeff Sanny
Publisher:
OpenStax - Rice University

College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College

Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning

University Physics Volume 1
Physics
ISBN:
9781938168277
Author:
William Moebs, Samuel J. Ling, Jeff Sanny
Publisher:
OpenStax - Rice University

College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College

Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning

Glencoe Physics: Principles and Problems, Student…
Physics
ISBN:
9780078807213
Author:
Paul W. Zitzewitz
Publisher:
Glencoe/McGraw-Hill

College Physics
Physics
ISBN:
9781285737027
Author:
Raymond A. Serway, Chris Vuille
Publisher:
Cengage Learning

College Physics
Physics
ISBN:
9781305952300
Author:
Raymond A. Serway, Chris Vuille
Publisher:
Cengage Learning