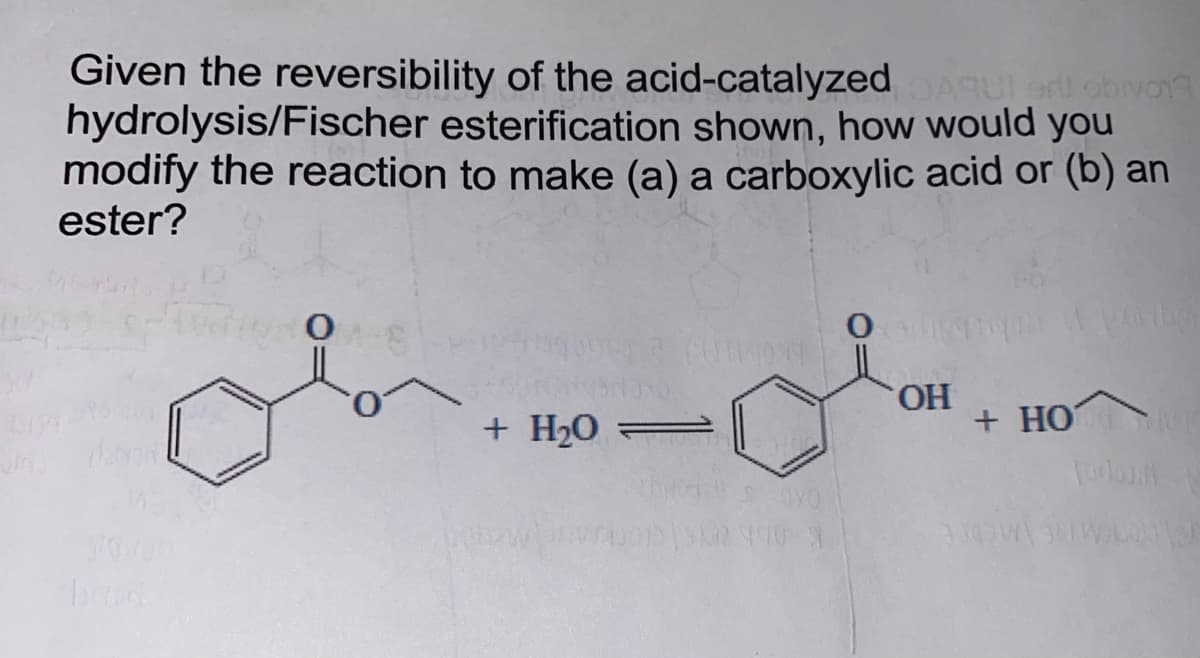
Given the reversibility of the acid-catalyzed DAGUI er! obivo17 hydrolysis/Fischer esterification shown, how would you modify the reaction to make (a) a carboxylic acid or (b) an ester? o 133 (014049 + H₂0 = -0X0 362790-9 O OH on Wag + HOVIC Forlo 3007032015/
Given the reversibility of the acid-catalyzed DAGUI er! obivo17 hydrolysis/Fischer esterification shown, how would you modify the reaction to make (a) a carboxylic acid or (b) an ester? o 133 (014049 + H₂0 = -0X0 362790-9 O OH on Wag + HOVIC Forlo 3007032015/
Chapter21: Carboxylic Acid Derivatives: Nucleophilic Acyl Substitution Reactions
Section21.6: Chemistry Of Esters
Problem 16P
Related questions
Question
Please answer all and show all steps.
Transcribed Image Text:Aqui er! obivor
Given the reversibility of the acid-catalyzed
hydrolysis/Fischer esterification shown, how would you
modify the reaction to make (a) a carboxylic acid or (b) an
ester?
họ
303 (0114034
Noxo
+ H₂0 =
0x0
952790-9
090
OH
(01 Vanhe
+ HOVIC
Todo
3000 30000 150
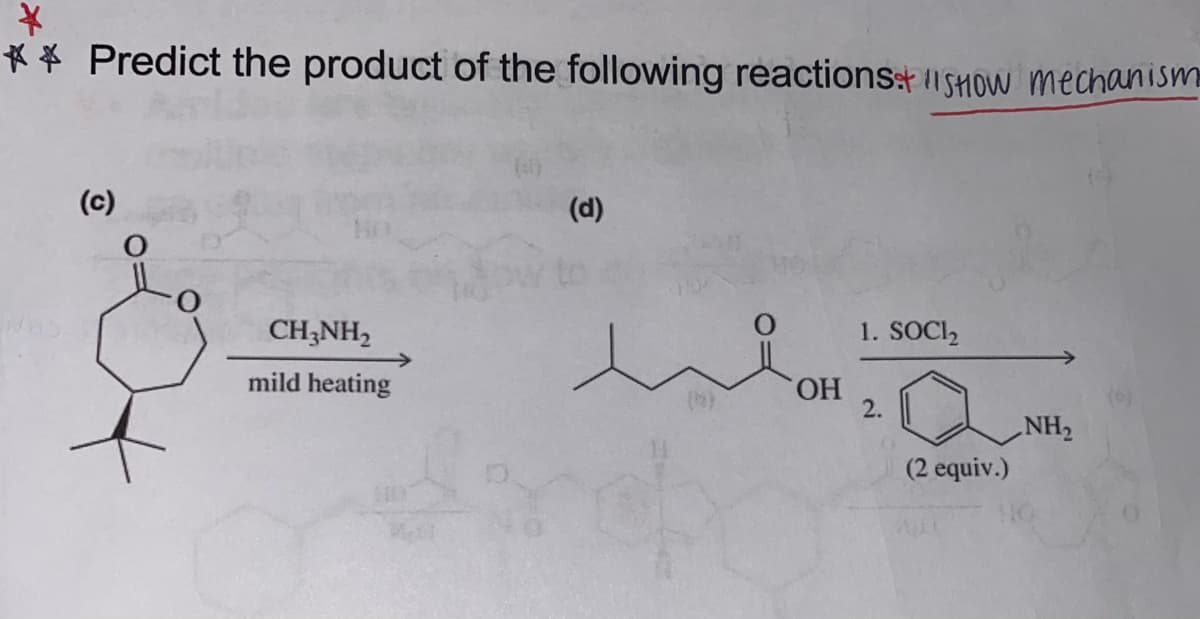
Transcribed Image Text:*
** Predict the product of the following reactions:+SHOW mechanism
(c)
&
CH3NH₂
mild heating
(d)
m
OH
1. SOCI₂
2.
(2 equiv.)
NH₂
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps with 4 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning