Q: The reaction A (g) = B(g) has an equilibrium constant of 5.8 and under certain conditions has Q =…
A: The equation for the reaction is: A(g) --->B(g) has an equilibrium constant of 5.8…
Q: 0.2306m to in show the sol
A:
Q: Cation Na+ K+ Al³+ Which one of the following ionic compounds has the largest lattice formation…
A:
Q: Find the molecular weight Find the percent composition of the of each element in the compound.…
A: we need to calculate the molecular weight and find the percentage composition for the given…
Q: Do we always have to convert Celsius into kelvin when using Gay-Lussacs law?
A: Answer:- This question is answered by using simple concept of Gay- Lussac law of gases which states…
Q: explain the combustion of methane gas COMBUSTION OF METHANE GAS: CH4 + 2O2 → CO2 + 2H2O + Energy
A:
Q: Gaseous methane (CH4) will react with gaseous oxygen (O₂) to produce gaseous carbon dioxide (CO₂)…
A:
Q: At a certain temperature, Kc = 0.500 for the reaction SO3(g) + NO(g) NO₂(g) + SO₂(g) If 0.100 mol…
A:
Q: 7. Which compound is more soluble in water-CH₂COOH or CH CH CH CH? Explain. 8. Which compound is…
A:
Q: How much of natural uranium is U-235? Group of answer choices 0.7% 13 % 33 % 99.3 % 100%
A:
Q: he pH of a 0.82M solution of hypobromous acid (HBrO) is measured to be 4.36. alculate the acid…
A:
Q: Let's use calorimetry to measure heats of reaction but this time let's have a limiting reactant.…
A: a.) Exothermic reaction is that in which heat is released and endothermic is that in which heat is…
Q: Complete the following table for aqueous solutions at 25 °C: hydronium ion concentration hydroxide…
A:
Q: "Synthesis gas" is a mixture of carbon monoxide and water vapor. At high temperature synthesis ges…
A: At high temperatures, carbon monoxide (CO) and water vapor react with each other to form carbon…
Q: Complete the table below, using the diagram of an atom shown at right. name symbol 0 0 proton e 0…
A: Electrons are type of subatomic particles with a negative charge . Proton is a type of subatomic…
Q: An unknown amount of acetylene gas occupies 12.40 mL. When the amount of this gas is changed to 0.57…
A:
Q: A photon has a frequency of 8.97 x 108 Hz. What is the energy of this photon in Joules? (h = 6.626 x…
A: Given, Frequency of a photon (v) = 8.97 × 108 Hz = 8.97 × 108 s-1 Energy of a photon (E) = ? Note:…
Q: SrSO4 SO³- SO4 Sr² + Sr³+ so²- Sr+
A:
Q: Use the data found in the Thermodynamic Values link under Reference Materials to calculate AH, AS,…
A: Given data :
Q: Diesel fuel for automobiles and trucks is a mixture of hydrocarbons that can be modeled by C16H34.…
A:
Q: Gaseous methane CH4 reacts with gaseous oxygen gas O2 to produce gaseous carbon dioxide CO2 and…
A:
Q: A certain first-order reaction is 60.% complete in 55 s. What are the values of the rate constant…
A: Initial concentration ( XA= 100%) Concentration after time 55 s ( XB =100 — 60% = 40%) Time = 55 s…
Q: What is the rate reaction of D? The rate expression for the reaction is: rate = k [NO][0₂]. 2NO(g) +…
A: Rate expression is given by the slowest step. Rate expression should not contain any intermediate…
Q: What is the frequency of a photon if the energy is 7.21 x 10-¹⁹ J? (h= 6.626 x 10-34 J·s)
A: Frequency is directly proportional to energy
Q: what is the conjugate acid of H2PO4
A: Conjugate acid - when base take proton its conjugate acid form
Q: 1. A gas system has an initial volume of 2400mL with the pressure unknown. When the volume changes…
A:
Q: The best definition of a chiral molecule is a molecule that has NO plane of symmetry. Sometimes,…
A:
Q: Question 20 of 26 Submit What is the energy (in J) of a mole of photons that have a wavelength of…
A: Given -> Wavelength= 701nm h = 6.626 × 10-34 J.s c = 3.00 × 108 m/s
Q: For a liquid vaporizing to a gas at a temperature higher than the liquid's boiling point, what are…
A:
Q: Liquid octane CH3CH26CH3 reacts with gaseous oxygen gas O2 to produce gaseous carbon dioxide CO2…
A:
Q: How many significant figures are in the following? 20.300 cm 0.00201 m 250 cm 1 in = 2.5 cm 25…
A:
Q: What set of reagents can best accomplish the following transformation? oor A) PCC in CH₂Cl₂ D)…
A:
Q: A 0.394 g sample of potassium hydrogen phthalate, KHCH404 (MM=204.44 g/mol), is dissolved in 100 mL…
A: Given: The mass of potassium hydrogen phthalate, KHCH4O4 = 0.394 g The volume of NaOH solution is…
Q: Multiply or divide the following measurements. Be sure each answer you enter contains the correct…
A: Rules for determining the correct no. Of significant figures in answers: Rule 1. The result of an…
Q: Maleic acid (H2C4H2O4, represented by H2M) is the precursor of malic acid widely used in industry.…
A: #(a): Maleic acid, H2M is a diprotic weak acid. NaOH is a monoacidic strong base. The balanced…
Q: What is the wavelength (in nm) of a photon if the energy is 7.17 x 10-¹9 J? (h= 6.626 X 10-34 J. s)
A:
Q: For the reaction Ca(OH)2 (aq) + 2HCl(aq) → CaCl₂ (s) + 2H₂O(1) AH° = 30.2 kJ and AS° = 205.9 J/K The…
A: The reaction is ; Ca(OH)2(aq) + 2HCl (aq)---> CaCl2(s) + 2H2O(l) ∆H০ = -30.2 kJ ∆So= 205.9 J/K…
Q: what is the change in entropy when150 ml of cold (278 k) water is added to 150 ml of nearly…
A:
Q: 5 Work Problem Hydrogen peroxide decomposes according to the following reaction: H₂O2 (1) = H₂O)…
A:
Q: What is the electron configuration for phosphorus? 1s²2s²2p 3s 3p Copy and paste the valence into…
A: Exactly half filled and fully filled configuration are more stable due to symmetrical filling and…
Q: According to Boyle's Law, as pressure decreases what do you expect will happen to the volume? A.…
A:
Q: The beautiful Taj Mahal in Agra, India, is made of marble, which consists primarily of calcium…
A: nitric acid, HNO3(aq) is a strong monoprotic acid. marble, CaCO3(aq) is a weak diprotic base. So…
Q: 165, How many protons and neutrons are in an atom of Number of protons=0 Number of neutrons = [ Ho ?…
A:
Q: Add or subtract the following measurements. Be sure each answer you enter contains the correct…
A: The significant figure refers to particular digits in a number. According to the measurement rule,…
Q: species H₂PO4 HSO4 2- SO4 H₂O OH H₂PO4 HIO 3 103 relative pH of 0.1 M aqueous solution (Choose one)…
A: pH can be defined as -log [H+]. Also stronger a acid is, more H+ ion will produce and lower will be…
Q: How many grams of CO2 are produced from 7.15 g of ethanol?
A:
Q: How was x calculated, was quadratic or the 5% assumption used?
A: Here the value of "x" is calculated by using the quadratic equation which gives x = 0.0125 M
Q: Write the correct balanced net ionic equation for the following reaction; Pb(ClO4)2 (aq) with…
A: We have to write the correct balanced net ionic equation for the following reaction; Pb(ClO4)2 (aq)…
Q: Write a balanced reaction equation including the states for the reaction between Al and (i) H+ (ii)…
A: Al is more reactive than hydrogen. Hence Al(s) reacts with acids to form H2(g) and itself gets…
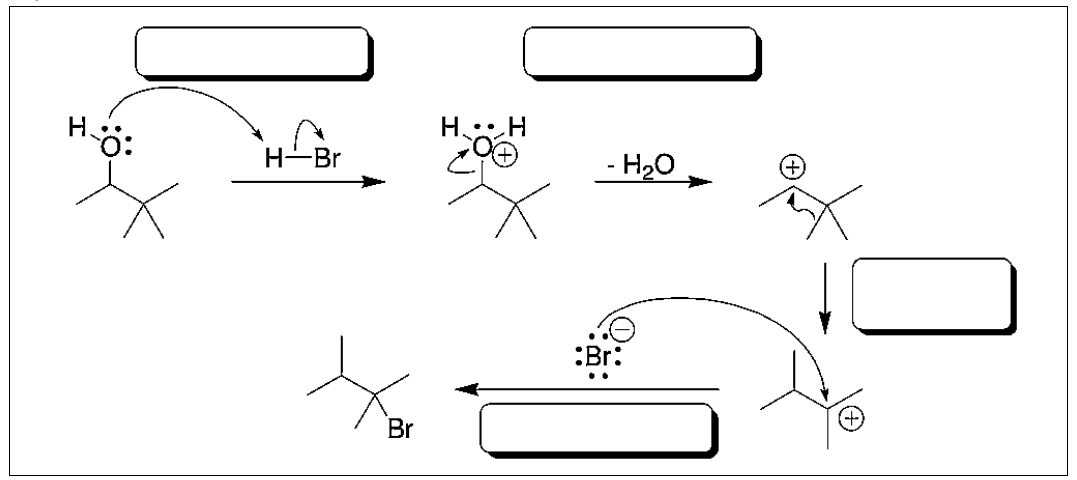
Identify each of the following arrow pushing patterns.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Rank the following groups in order of decreasing priority. a. – COOH, – H, – NH2, – OH b. – H, – CH3, – Cl, – CH2CI c. -CH2CH3, -CH3, -H, -CH(CH3)2 d. – CH = CH2, – CH3, – C ≡ CH, – HDraw 2,3 - dimethyl hexane and fill in all H’sPlace the following compunds in order from lowest to highest boling points. Explain why you have ranked each compound C6H14 CH3OH H2O C3H8

