Q: Sulfur tetrafluoride gas is collected at 23.0 °C in an evacuated flask with a measured volume of…
A: Given that :The temperature of sulfur tetrafluoride gas = 23The volume of the gas = 15.0 LPressure =…
Q: Indicate which of the following is NOT a valid resonance contributor of the boxed compound. t A In B…
A: There are several electronic parameters to explain the stability of the organic compound. Resonance…
Q: An analytical chemist weighs out 0.032 g of an unknown monoprotic acid into a 250 mL volumetric…
A:
Q: -5 The equilibrium constant, Kc, for the following reaction is 7.00 x 10 at 673 K. NH₂I(8) NH3 (g) +…
A: moles of NH4I = 0.599 molvolume of vessel = 1.00 LT = 673 KKc = 7.0010-5
Q: draw heterolytic cleavage of manganese and imidazole mechanism
A: In the given question we have to draw the heterolytic cleavage of manganese and imidazole…
Q: Use the standard reaction enthalpies given below to determine AH° rxn for the following reaction:…
A: Answer:Enthalpy change is an extensive property and it is additive in nature, therefore, enthalpy…
Q: How would you use ¹H NMR spectroscopy to distinguish between the two compounds shown? O Golf H H…
A: NMR spectroscopy is a very important tool for the determination of the structure of the organic…
Q: TOM CHS TO Use a dash and/or wedge bond to indicate the stereochemistr of substituents on asymmetric…
A: Allkene reacts with bromine (Br2) and water (H2O) to form a bromohydrin which react with strong base…
Q: Given that K, for CH₂CH₂NH₂ is 6.3 x 10-4 at 25 °C, what is the value of Ka for CH₂CH₂NH3 at 25 °C?
A: The Kb value for it's conjugate acid =?
Q: Consider the reaction when aqueus solutions of sodium hydroxide and chromium(III) nitrate are…
A: Steps to write a net ionic equation and complete ionic equation for a molecular reaction : Write…
Q: Draw thw product, and give the reaction mechanism for the acid - catalysed esterification of…
A:
Q: C 2.1 mol He (g) 2.8 atm F 1.9 mol Kr (g) 1.6 atm elect the set of graphs below that show the…
A: The objective of the question is to select the graphs that show the distributions of the speed of…
Q: 17.24 • Provide a systematic name for each of the following compounds: a. b. C. d. e. > Answer OH…
A: Benzene is considered as parent chainNumbering is done to the ring so that substituents gets…
Q: Highlight in red each acidic location on the organic molecule at left. Highlight in blue each basic…
A: In accordance with the Bronsted-Lowry concept, an acid is characterized as a provider of protons,…
Q: 1. Propose the mechanisms and predict the products of the following reactions. a) b) OH OH H₂SO4…
A: The given reactions are dehydration reactions of alcohol, which lead to the formation of an…
Q: An ideal solution is repaired by mixing 7.00 g of toluene with 25.0 g of benzene. Use the following…
A: The objective of this question is to calculate the vapor pressure of an ideal solution prepared by…
Q: A 1/20 inch thick lead sheet is used for protection from medical X-rays. If a single lead atom has a…
A: To find out how many lead atoms thick the sheet is, we need to convert the thickness of the lead…
Q: Cholesterol (386.7 g/mole) is a non-electrolyte and a critical component of the cell membranes of…
A: The objective of the question is to calculate the change in boiling point of a solution containing…
Q: 3. While not carbonyl compounds themselves, nitriles can be hydrolyzed in a similar. fashion.…
A: The mechanism of the reaction has various steps which are shown in step 2. In this reaction, nitrile…
Q: Which major IR absorption(s) is/are present above 1500 cm for the following compound? Check all that…
A: Given ,Molecule : Objective : Which major IR absorptions are present above 1500 cm-1.
Q: A beaker with 1.40x10² mL of an acetic acid buffer with a pH of 5.000 is sitting on a benchtop. The…
A: The objective of this question is to determine the change in pH when a strong acid (HCl) is added to…
Q: A 23. A 04100 M solution was made of each of the following solutes: NaCIO, KNO2, MgI2, and CsHsNHBr…
A: The question is based on the concept of the pH of the solution.It is defined as a negative logarithm…
Q: A cup containing 95.0 mL of H2O is initially at room temperature 22.9°C. A chilled steel rod at…
A: The objective of this question is to find the mass of the steel rod. We can use the principle of…
Q: What charges would the ions need to make the hypothetical ionic compound XY₂? The X ion has a charge…
A:
Q: - CH3 why does this decompose to methanol and carbon dioxide? | CH₂CH₂ OCH3 + H₂O → Draw the…
A:
Q: The salt calcium chloride is soluble in water. When 1.55 g CaCl₂ is dissolved in 116.00 g water, the…
A: Given,The amount of water = 116.00 gTemperature (T1) = 250CTemperature (T2) = 27.470C. = (27.47-…
Q: The cyclobutenyl dichloride deriviative below reacts with the powerful Lewis acid antimony…
A: Given that, the reaction is:
Q: Over-the-counter (OTC) antacids are chemicals that neutralize acidity to relieve a painful reflux.…
A: Antacids :Our stomach produces hydrochloric acid(HCl). It helps in the digestion of food without…
Q: 2. Rank the compounds from lowest to highest C=O stretching frequency. Defend your answer. a. 3.…
A: The question has three parts. The first part is asking to rank compounds based on the C=O stretching…
Q: Classify each reaction as addition, elimination, substitution, rearrangement, oxidation, or…
A: Looking at the reactant and product, it is clearly seen that there occurs rearragement.
Q: draw mechanism for the reaction B. 1) NBS 2) KNO3 Conc. H₂SO4
A: To find out the structure of product and mechanism associated with the formation of it from the…
Q: A student finds that a piece of metal has a mass of 12.54g. When the metal is placed inside a flask…
A:
Q: Draw the product for the following aldol reaction: LDA ?
A: The given reaction is:Determine the product of the reaction.Organic reactions are the reactions in…
Q: 2. b) Suggest a sequence of reagents by which you expect you would best effect the following…
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: The rate constant for this first-order reaction is 0.370 s¹ at 400 °C. A products How long, in…
A: Given information:The rate constant for first-order reactions = 0.370 s-1 at 400 °C.Initial…
Q: Balance: Li(s) + Co2 + (aq)-> Li+(aq) + Co(s) and CrO42-(aq) + H+(aq) + Co(s) - > Cr3+ (aq)+ H2O(1)…
A: To balance an equation one has to balance number of atoms as well as charge.For balancing ionic…
Q: 01 O. III What is the product of the following reaction? MeO OMe MeOH acid, heat COMe OMe III OMe IV
A:
Q: Draw the product of the reaction shown below. Use a dash or wedge bond to indicate stereochemistry…
A: The objective is to determine the product formed by the given reaction. Trienes present in…
Q: The equilbrium constant for the reaction 2NO(g) N₂(g) + O2(g) is 2.4 x 10³ at 1000°C. The initial…
A:
Q: Predict the major products of this organic reaction. NBS hv ? Draw only the major product or…
A:
Q: A reaction at 16.0 °C evolves 703. mmol of chlorine pentafluoride gas. Calculate the volume of…
A: The ideal gas equation gives the relation between pressure, volume, temperature, and the number of…
Q: How much heat must be removed from a 90.5 g sample of benzene (M = 78.11 g mol-1) to cool the sample…
A: The objective of the question is to determine the heat removed from the given sample of benzene.It…
Q: Automotive air bags inflate when sodium azide decomposes explosively to its constituent elements:…
A: Given reaction,Mass of nitrogen gas produced = 2.80 gThe molar mass of Mass of sodium azide required…
Q: Calculate ΔrH for the following reaction: P4(g) + 10Cl2(g) → 4PCl5(s) Express your answer to 4…
A: The thermochemical equations can be treated as algebraic equation which can be added, subtracted,…
Q: Which one is H NMR of Ethyl acetate ?
A: The objective of this question is to identify the correct H NMR of ethyl acetate.
Q: The interactions between the tert-butyl and ethyl groups in the chair below is called type your…
A: The objective of the question is to fill in the blank with the suitable type of interaction seen in…
Q: Evaluate the equation and enter the final answer using the correct significant figures in scientific…
A: In a number, all non-zero digits are considered significant. Zeros are significant if they are…
Q: Balance the half-reaction in basic solution. Use e as the symbol for an electron. half-reaction: SO…
A:
Q: Which of the following acids would produce the highest pH at the equivalence point in a weak…
A: The objective of the question is to predict which of the given acids would produce the highest pH.
Q: This energy diagram shows the allowed energy levels of an electron in a certain atom. (Note: the SI…
A:
help me please
Please fill in the reagents,intermediates, and/or products of the following reactions.
all the reactions shall be covered in Chapter 12-15 from Organic Chemistry, 6th Edition by Marc Loudon and Jim Parise.
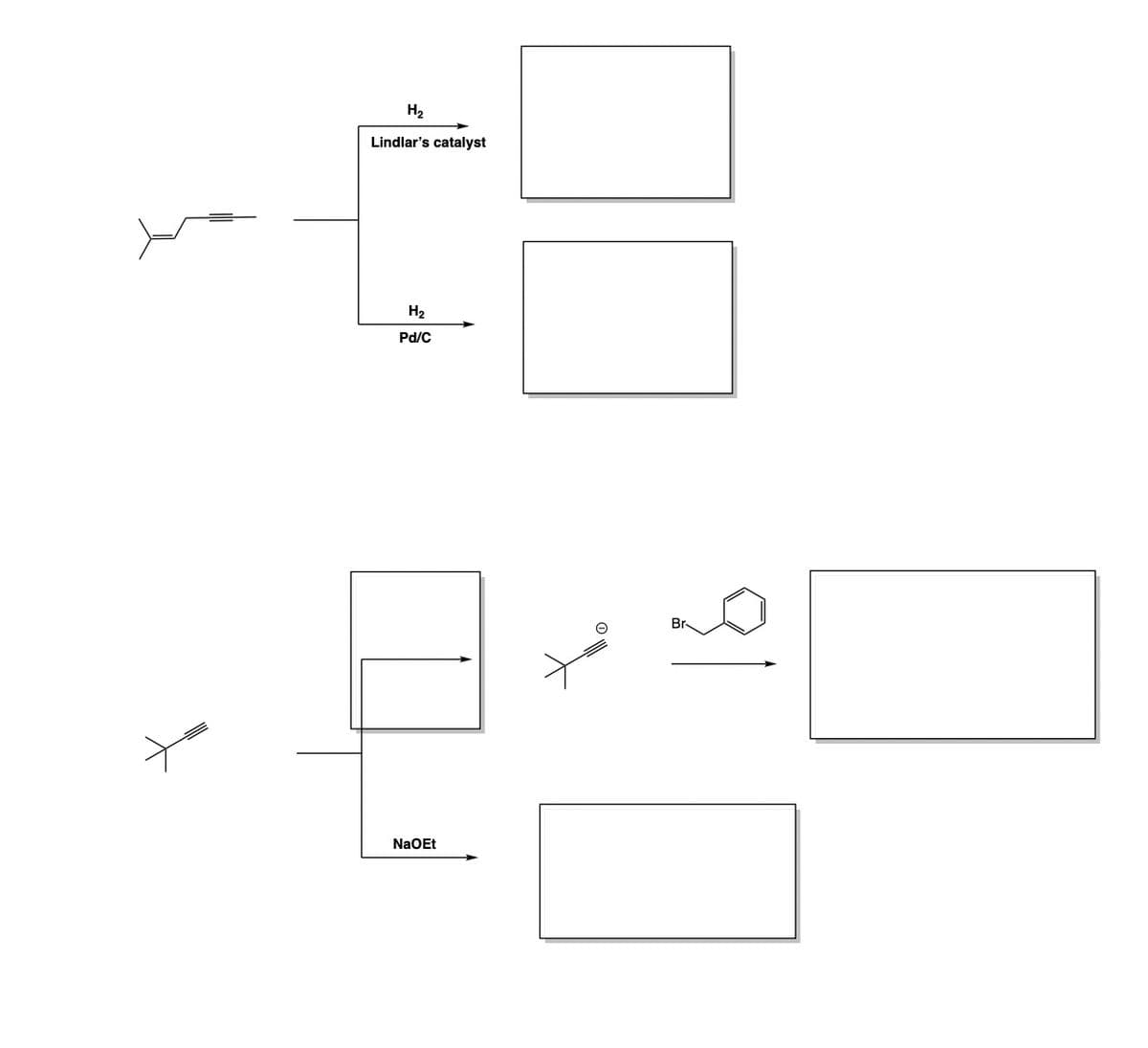
Step by step
Solved in 3 steps with 2 images

- An enzyme-catalyzed reaction has a Km of 1.4 mM and a Vmax of 7 nM/s. What is the initial velocity when the substrate concentration is 0.6 mM?Qmax is maximum adsorption of acetic acid on activated carbon.If there is another organic compound that has high affinity to activated carbon in the same acetic acid solutions. How Qmax of acetic acid adsorption will change?Lower, Higher or same? EXPLAINWhat is an adsorption isotherm? What is the texture of a catalyst?
- What is a ‘shape-selective catalyst’?An enzyme-catalyzed reaction has a Km of 1 mM and a Vmax of 4 nmole/L sec-1. The reaction velocity (nmole/L sec-1) when the substrate concentration is 0.25 mM is: A. 1.25 B. 10.0 C. 5.0 D. 0.50 E. 1.0 F. 100 G. 150 H. 55 I. 751. What is the difference between the Freundlich and Langmuir isotherm models in adsorption? 2. What is the difference between Pseudo-First Order and Pseudo-Second Order of kinetics?
- If the concentration of Sn2+ ions in a solution of Sn(OH)2 was measured to be (1.41x10^-3), what is the Ksp value for Sn(OH)2?Suppose it is known that ozone adsorbs on a certain surface in accord with a Langmuir isotherm. How could you use the pressure dependence of the fractional coverage to distinguish between adsorption (i) without dissociation, (ii) with dissociation into O + O2, (c) with dissociation into O + O + O?A typical diffusion constant for small molecules in aqueous solution at 25 °C is 6 × 10−9 m2 s−1. If the critical reaction distance is 0.5 nm, what value is expected for the second-order rate constant for a diffusion-controlled reaction?
- what is solution homogenous catalysis?2 what is the purpose of BSC Level 1 & 2, proper layout, performance, and its application in biosafety assessmentUsing data in Appendix E, calculate the standard emf foreach of the following reactions:(a) H21g2 + F21g2 ¡ 2 H+1aq2 + 2 F-1aq2(b) Cu2+1aq2 + Ca1s2 ¡ Cu1s2 + Ca2+1aq2(c) 3 Fe2+1aq2 ¡ Fe1s2 + 2 Fe3+1aq2(d) 2 ClO3-1aq2 + 10 Br-1aq2 + 12 H+1aq2 ¡ Cl21g2 +5 Br21l2 + 6 H2O1l2

