Q: to the image below 3. Which of the following best describe the solution to your final answer in PRO...
A:
Q: HBr, (t-BuO)2
A: Major product of following reaction
Q: tu 1or tIs question. A student ran the following reaction in the laboratory at 1096 K: 2S03(g)2s02(g...
A: Given equilibrium is 2SO3(g) <----> 2SO2(g) + O2(g) Initial concentration of SO3 = 9.37×10-2...
Q: What mass of copper is contained in 111. g of copper(II) fluoride? Enter your response rounded to th...
A: Copper (II) fluoride contains 37.42% F by mass.
Q: This series demonstrates the addition of 0.25 M solution of lead (II) nitrate (??(?? ) ) to 0.5 M so...
A: 1. When aqueous solutions of lead(II) ion are treated with potassium chromate solution, we notice th...
Q: (a) C7H10B12 (c) C3H6O3 (b) C3H4O2 (d) CSH12N2
A: Given that : We have to calculate the degree of unsaturation for each of the following molecules : (...
Q: A concentrated salt solution has a mass of 5.29 g for a 5.00 mL sample. . What is the specific gravi...
A:
Q: What is the concentration of OH- remaining in solution after 120.0 mL of 0.100 M HCl is mixed with 8...
A: Molarity = number of moles of solute/volume of solution (in L) So, number of moles = molarity × vol...
Q: In what absolute temperature will a given sample of ammonia at 300K and a critical temperature of 13...
A: The two gases will be in a corresponding state when they have the same reduced temperatures.
Q: a) Equimolar amounts of H, and CO are mixed. Using data from Table A-V calculate the equilibrium mol...
A:
Q: Fill in the blanks to produce a balanced chemical reaction for solid P4 and H2 gas is: _____P4 + __...
A:
Q: What is the main product of the following reaction? NaSCH3 acetone SCH; 1SCH5 1SCHy- I. I. II. IV.
A:
Q: The following initial rate data are for the reaction of ammonium ion with nitrite ion in aqueous sol...
A:
Q: 25.00 mL portion of a H2SO4 solution was titrated with a 0.256 M KOH solution. To reach the equivale...
A: A) chemical equation is shown below B) The Molarity of sulfuric acid 0.160 M.
Q: 5) A 5.00 mL aliquot of an acetic acid solution requires 42.3 mL of 0.24 M NaOH for neutralization, ...
A:
Q: explain what advantage the technique of reflux has over conventional boiling
A: Refluxing is a technique that is used to boil a mixture over a long period of time. In this techniqu...
Q: A.This series shows a progression as 0.5 M copper sulfate (???? ) added to a test tube containing 0....
A:
Q: Write the formula for the conjugate acid of each of the following bases.
A: Given :- Base i.e. H2PO3- To determine :- Chemical formula of conjugate acid
Q: A sealed container holding 0.0255 L of an ideal gas at 0.997 atm and 65 °C is placed into a refriger...
A:
Q: Choices: Solute and Solvent Caffeine in a cup of coffee ...........Solute Nitrogen in the a...
A: When one substance dissolves into another, a solution is formed. The solute is the substance that is...
Q: A student needs to determine density of a liquid. The student determines mass of an empty flask to b...
A:
Q: number of moles of (c) acetate ions, CH3COO-, in 0.43 mol of Ca(CH3COO)2.
A: 2) In 1 mol of Ca(CH3COO)2 has 2 moles of CH3COO- ion.
Q: 3. Write and balance the chemical equation for the oxidation of I ion in aqueous solution by chlorin...
A: Here balance the equation by oxidation and reduction reaction.
Q: [OH-] = 7.0 × 10-10 Express the concentration to two significant figures and include the appropriate...
A: Given is, [OH-] = 7.0×10-10
Q: The equilibrium constant, K., for the following reaction is 9.52×10-2 at 350 K. CH4 (g) + CC14 (g)=2...
A: We are given equilibrium condition of two reaction along with their equilibrium constant, we have to...
Q: Can you help me how to balance this equation CS2 + Cl2 ---> CCl4 + SCl
A: The given equation is: CS2+Cl2→CCl4+SCl
Q: The compound is CH4N2O. If the molecular weight of the compound is 120.12 g/mol, what is the molecul...
A:
Q: Which of the following hypothetical isotopes have the same mass number? Note: the number before the ...
A:
Q: What is the conjugate base of HSO4 ? Express your answer as a chemical formula. View Available Hint(...
A:
Q: A. The Internal System of Units Fill in the space provided with the correct SI units and symbol for ...
A: As per our guideline we can answer only one question Kindly post other questions seperately We ha...
Q: Balance each of the following redox reactions in acidic solution. For each balanced reaction, identi...
A:
Q: Arrange the following aqueous solutions, all at 25 °C, in order of decreasing acidity. Rank from mos...
A: Rank as acid and bases
Q: 1.What is the lactic acid concentration (in M) of the titrated sample? 2. Determine the lactic acid ...
A:
Q: for a real gas at 25 degrees celsius whose critical temperature and critical pressure are 30 K and 0...
A:
Q: What is the half-life (in seconds) of a zero-order reaction which has an initial reactant concentrat...
A: Given that: Initial reactant concentration [A]∘ = 0.824 M k = 5.43×10-2M/s Half-life of zero order r...
Q: Some formulas could be either molecular or empirical formulas; however, some can only be molecular f...
A: Empirical formula is the formula of a compound having the simplest whole number ratio of elements. M...
Q: How can the following compounds be distinguished using their mass spectra? CH3 H3C ČH3 H3C. CH3 H3C ...
A: Mass spectrometry is used for differentiate compound based on their mass to charge ratio.
Q: Circle the followig molecules that can be used to forma Grianard reagent. Then choose one you circle...
A: Preparation of grignard reagent Alkyl halides react with mg in the present of dry ether to form RMg...
Q: If the reaction between zinc chloride and sodium phosphat produces 25.7 g of product, then what is t...
A:
Q: Provide the mechanism for the following transformation. D = 2H
A: At first D+ present in D-Br adds to the alkene to give tertiary carbocation. Then 1,2-Me- shift occu...
Q: The equilibrium constant, K., for the following reaction is 2.44×10-2 at 1140 K. 2SO3(g) 2S02(g) + O...
A: We have to calculate the equilibrium constant for given reaction.
Q: 2. Predict the shapes of the following compounds: a) H,S b) AsH; c) AIBR,
A:
Q: Discuss about Carbohydrate Metabolism and Diabetes Mellitus: prevention during the pandemic
A: Discuss of carbohydrate mechanism and diabetes mellitus -prevention during the pandemic is given bel...
Q: A certain chemical reaction releases 25.4 kj/g of heat for each gram of reactant consumed. How can y...
A: Given, chemical reaction releases 25.4 kj/g of heat for each gram of reactant comsumed. We have t...
Q: 05. Aluminum has an atomic number of 13 and a mass number of 27 a. It has 14 protons b. It has 27 ne...
A:
Q: Provide 2 additional resonance structures for the following molecule and draw the electron pushing a...
A: Given that : We have to provide 2 additional resonance structures for the following molecule includi...
Q: СООН HO (а) or Cl `Cl I II HO HO (b) or `CH3 `CI I II
A:
Q: Phenol, C6H5OH, methanol, and CH3OH both have O-H groups, but phenol is a stronger acid. Draw the st...
A:
Q: Please answer this NEATLY, COMPLETELY, and CORRECTLY for an UPVOTE. Topic: Acid-Base Equilibria and...
A: Solving first three as per the guidelines. The mass balance equations are as follows: 0.1 F H2A ...
Q: At what temperature does sulfur hexafluoride have a density of 0.3350 g/L at 0.8210 atm?
A: Given Density ( d ) = 0.3350 gram/Liter Pressure ( P ) = 0.8210 atm Temperature ( T ) = ...
Predict the product of the reaction.
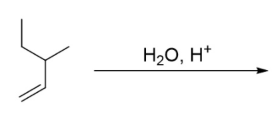
Step by step
Solved in 2 steps with 1 images


