Q: What relationship do these two alkenes have with respect to each other? Conjugated Cumulated None of…
A: To find out the relation between the highlighted alkene
Q: When is conductance at its lowest possible value for a solution that can be analyzed via the…
A: Conductance is a measure of the ability of a solution to conduct an electric current.. The…
Q: When baking soda (solid sodium bicarbonate or sodium hydrogen carbonate, NaHCO₂) is heated, it…
A:
Q: In molecules, there are also atoms or groups of atoms that do not absorb radiation in the near UV…
A: Auxochromes are certain groups of atoms which have lone pairs that can be donated to the…
Q: 10 Organic Chemistry Please help
A: Acetylide ions are strong bases as well as strong nucleophiles. It is mainly used for new C-C bond…
Q: Combustion of hydrocarbons such as hexane (C6H₁4) produces carbon dioxide, a "greenhouse gas."…
A: 1.) Combustion reaction takes place in presence of oxygen , so we would write the reaction using…
Q: You are provided with 100 ml of 1 M HCl. You add a 1 ml of solution of 0.1M sodium acetate in water…
A: Let's analyze each option:The pH of the final solution does not change significantly:When we add 1…
Q: Determine the molecular formula and calculate the molar mass (in g/mol) of histidine, the essential…
A:
Q: Calculate the solubility of O₂ in water at a partial pressure of O₂ of 150. torr at 25°C. The…
A: Given information :Partial pressure of O2 = 150 torr at 250CHenry law constant (k) for O2 = 1.3 X…
Q: Which of the following molecules can exist as aromatic upon the loss of a proton (H+)?
A: Highly stable and highly conjugated cyclic compounds that do not give addition reactions and follow…
Q: Classify the following redox reactions: (a) P4+ 10 Cl₂ → 4 PC15 (b) Cl₂ + 2 KI →2 KCI + 12 (c) 2 NO…
A:
Q: H₂SeO₂ (aq) + 61- (aq) + 4H* (aq) Expt. # [H₂SeO₂] (M) 1 0.644 2 0.100 3 0.644 4 0.500 [-] (M) 0.820…
A: Answer:Power of concentration term of a reactant in the rate law is the order of reaction with…
Q: The airbags that protect people in car crashes are inflated by the extremely rapid decomposition of…
A: Word equation is Solid sodium azide ---> solid sodium + gaseous dinitrogen
Q: How many tertiary carbons are present in the compound shown below? CI 5 4 2 Br H
A: When a sp3 carbon atom forms compounds, it will get bonded to four other atoms. If that sp3 carbon…
Q: Part B.1: Measuring the volume of a sample of water. Measuring Device Measured Value Beaker…
A: The key aspect that should be dealt with while experimenting is precisely recording the readings.…
Q: You finish doing an experiemnt with Benzoin. These are the results you get: Boiling start point: 137…
A: The objective is to calculate benzoin's initial mass, mass of recrystallized, percent recovery,…
Q: Give the name of the complex substituent shown below. P (bold/red) represents the parent chain of…
A: For the branch chain of the any parent chain the numbering will start from the next carbon of the…
Q: How many grams of glucose must be added to 0.250 L of water to create a 0.150 m gluci se solution?…
A:
Q: Select the correct IUPAC name for the following organic substrate, including the R or S designation…
A: SN1 reaction goes through planar carbocation intermediate. Nucleophile (H2O) attack to positive…
Q: A chemist carefully measures the amount of heat needed to raise the temperature of a 1.97 kg sample…
A: Amount of heat,Q required to change the temperature of a substance of mass ,m and specific heat…
Q: 6 8 7 9 S 3 0 PPM 8.036 8.004 7.926 7.876 7.508 7.480 7.422 7.393 7.339 7.271 3.161 3.039 2.920…
A: To identify the structure based on the given 1H NMR
Q: What is the product of the following reaction? Br a. b. C. d. Br. Select one: a. b. C. d. ZI Br…
A: A nucleophilic substitution reaction is a type of organic reaction where one nucleophile (an…
Q: Explaining Vapor Pressure in Terms of IMFs Given the shown structural formulas for these four…
A: Answer:Vapor pressure is inversely proportional to strength of intermolecular forces present in…
Q: 19.6 Proposing a Synthesis for a Polysubstituted Benzene Ring COMPLETE THE FOLLOWING RETROSYNTHETIC…
A: To complete the retrosynthetic analysis by putting the missing intermediates
Q: Br Br CH₂CH₂CCH Br Br B CH₂CH₂CH₂CH B Br Br Br Br CH₂CHCHCH₂ Br Br CH3CHCHCH no reaction
A: Alkeene reaction with excess Br2 produced tetrabromoalkane.
Q: Doctor’s order: Sulfasalazine Oral susp 500 mg q 6 hr; Directions for mixing: Add 125mL of water and…
A:
Q: Draw the full mechanism please in BOND LINE FORMULA. Give explanation please OH S C H₂O+
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: Add 2-methyl-2-propanol (Alcohol Halogenation folder) and ether (Et₂0) to the reaction flask and…
A: Based on the information provided, here are the statements that apply to the reaction:True…
Q: (b) Using the Crystallographer's formula to determine the density (in g/cm³) of Po: ρ = Σ Ζ * Μ…
A: Answer:Here:
Q: 22. The traditional reasoning for why phenol is more acidic than cyclohexanol resides in a resonance…
A: Phenol is more acidic than cyclohexanol due to differences in their respective structures and the…
Q: Predict the major organic products and propose mechanisms of the following reactions. d. e. C(CH3)3…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: Identify each of the following compounds as ionic or covalent. NH4Cl CBr4 K2CO3
A: The objective of the question is to identify whether the given compounds are ionic or covalent. This…
Q: What is the relationship between the following? structural isomers geometric isomers same molecule…
A: Structural isomerism :-Compounds having the same molecular formula but different connectivity of…
Q: Design a synthesis of 4-phenyl-1-butene from 4-phenyl-2-butanone and methyl iodide. Û and CH 31
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: What ions make up the ionic compound Mn(CN)2 ? Cation anion
A: The objective of the question is to identify the cation and anion in the ionic compound Mn(CN)2.
Q: How many atoms are in 135g of calcium?
A: 1 mole of a substance contains an Avagadro number () of particles.
Q: 42.9 g of Aluminum nitrite and 97.2 g of ammonium chloride react to form aluminum chloride,…
A:
Q: Draw the major product of the following Diels-Alder reaction. Use the Bicyclic ring tool and guide…
A: Diels-Alder reaction is the cycloaddition reaction between diene and dienophile. The Diels-Alder…
Q: mall amount of acetonitrile (CH3CN) is dissolved in a large amount of water. Imagine separating this…
A: The enthalpy of a solution is defined as the quantity of heat absorbed or liberated when a solute is…
Q: A solution containing 8.60 g of non-electrolyte (molar mass 119 g/mol) dissolved in 93.2 g of…
A: Given,Mass of non-electrolyte = 8.60 gMass of chloroform = 93.2 g = 0.0932 kg [ 1 kg = 1000 g ]…
Q: What is the formula of the compound formed between the ions PO43- and NH4+?
A: The objective of this question is to determine the formula of the compound formed between the ions…
Q: Suppose the gas mixture produced by the decomposition reaction is trapped in a container at 17.0°C;…
A:
Q: The chemical structure of cysteine (C₂H,NO₂S) is shown below. Highlight each atom that is in an…
A: As we know that,the general formula of primary amine group is --NH2, thia means that,When a group…
Q: Provide the correct systematic name for the compound shown here. H3C ОН ОН CH3
A: The given compound is an alcohol because of the presence of the OH functional group.The parent chain…
Q: How many moles of sodium are in 1.73 x 10^24 Na atoms?
A: We have to find how many moles of sodium are in 1.73 x 10^24 Na atoms.
Q: Are the following pairs of compounds as the same molecul constitutional isomers, geometric isomers,…
A: Isomers are compounds that have the same molecular formula but different arrangements of atoms or…
Q: What is the general formula for a compound if anions occupy the hcp lattice points and cations…
A:
Q: Which of the following is a CORRECT description about the reaction coordinate diagram? I. The…
A: A reaction is exothermic if the change in enthalpy of the reaction() is negative.A reaction is…
Q: Propose a structural formula for a compound of molecular formula C11H1 402 by the given spectra, and…
A: Double Bond Equivalence (DBE) for the molecular formula C11H14O2 can be calculated by using the…
Q: Try this Problem: Draw the lowest energy conformation and the highest energy conformation along the…
A: Conformations are molecules that can be interconverted to each other by rotation across…
3
Please help
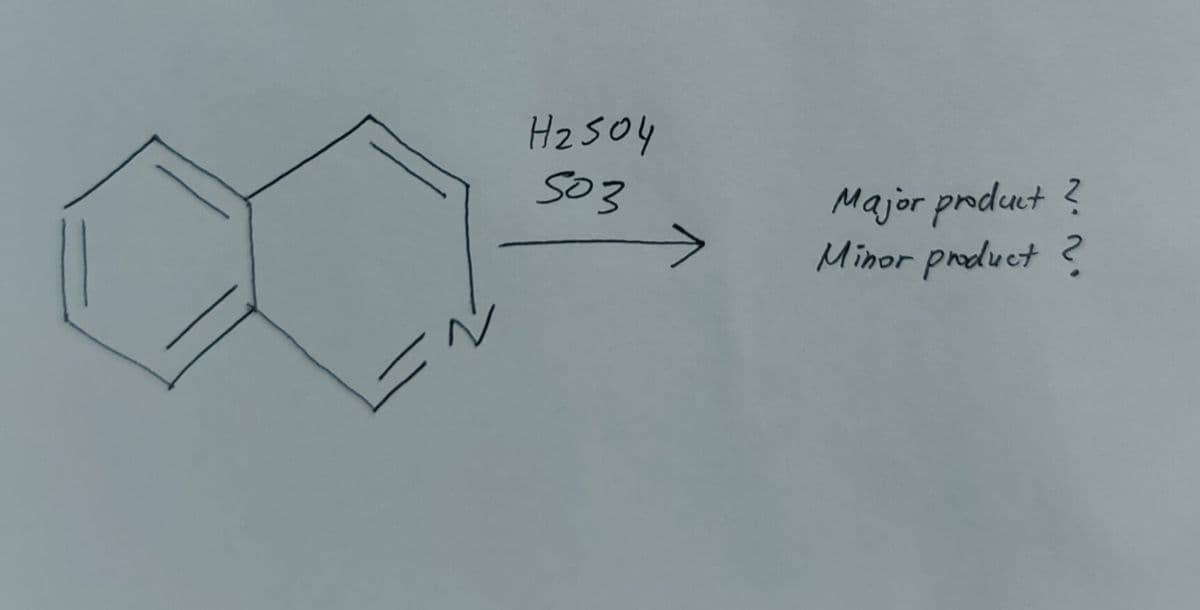
Step by step
Solved in 3 steps with 2 images

- Each H↔H eclipsing interaction in ethane costs about 4.0 kJ/mol. How many such interactions are percent in cyclopropane? What fraction of the overall 115 kJ/mol (27.5 kcal/mol) strain energy of cyclopropane is due to torsional strain.Give IUPAC names for the following substances (red = O, blue = N):CH3-CH2-OH reacts with H+/140C = ? + H2O
- CHEMISTRY AND ENVIRONMENTAL POLLUTION Explain how Hg2+ ion is transformed into methylmercury anddimethylmercury in the aquatic environment.Explain addition of H2O to an alkyne ?the action of halogenated derivatives of alkanes with KOH: CnH2n + 1 Cl + KOH = CnH2n + 2O + KCl is: a) radical substitution b) nucleophilic substitution c) electrophilic substitution d) addition reaction

