H₂C CH3 H3C Br Br: Br₂ CH₂Cl₂ H3C Br Electrophilic addition of bromine, Br₂, to alkenes yields a 1,2-dibromoalkane. The reaction proceeds through a cyclic intermediate known as a bromonium ion. The reaction occurs in an anhydrous solvent such as CH₂Cl₂. CH3 In the second step of the reaction, bromide is the nucleophile and attacks at one of the carbons of the bromonium ion to yield the product. Due to sta clashes, the bromide ion always attacks the carbon from the opposite face of the bromonium ion so that a product with anti stereochemistry is forme Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions 20 Br CH3 H3C Br: CH3 Br:
H₂C CH3 H3C Br Br: Br₂ CH₂Cl₂ H3C Br Electrophilic addition of bromine, Br₂, to alkenes yields a 1,2-dibromoalkane. The reaction proceeds through a cyclic intermediate known as a bromonium ion. The reaction occurs in an anhydrous solvent such as CH₂Cl₂. CH3 In the second step of the reaction, bromide is the nucleophile and attacks at one of the carbons of the bromonium ion to yield the product. Due to sta clashes, the bromide ion always attacks the carbon from the opposite face of the bromonium ion so that a product with anti stereochemistry is forme Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions 20 Br CH3 H3C Br: CH3 Br:
Chapter19: Aldehydes And Ketones: Nucleophilic Addition Reactions
Section19.SE: Something Extra
Problem 28VC
Related questions
Question
197.
Subject : - Chemistry
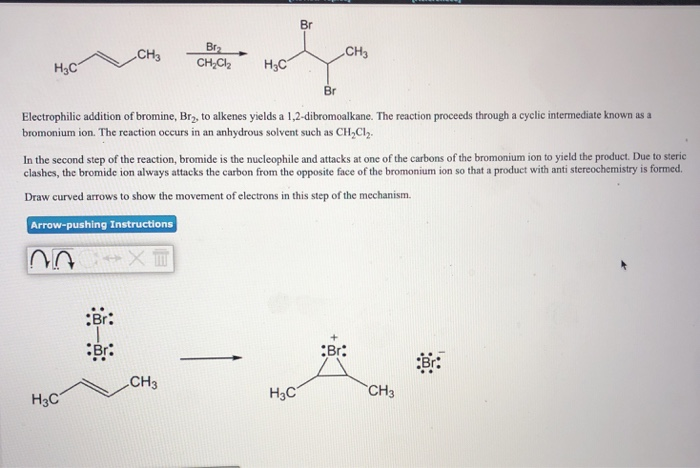
Transcribed Image Text:H₂C
CH3
H3C
Br
Br:
Br₂
CH₂Cl₂
H3C
Br
Electrophilic addition of bromine, Br₂, to alkenes yields a 1,2-dibromoalkane. The reaction proceeds through a cyclic intermediate known as a
bromonium ion. The reaction occurs in an anhydrous solvent such as CH₂Cl₂.
CH3
In the second step of the reaction, bromide is the nucleophile and attacks at one of the carbons of the bromonium ion to yield the product. Due to steric
clashes, the bromide ion always attacks the carbon from the opposite face of the bromonium ion so that a product with anti stereochemistry is formed.
Draw curved arrows to show the movement of electrons in this step of the mechanism.
Arrow-pushing
Instructions
20
Br
CH3
H3C
Br:
CH3
Br:
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

