Q: Calculate the osmotic pressure of a 0.0112 M solution of NaCl in water at 0 °C. Assume the van't…
A:
Q: 3. Which one of the following particles can drift and why. Briefly describe and write down the drift…
A: In a conductor , free electrons are present which have random velocities and move in random…
Q: H₂O(g) + Cl₂O(g) = 2 HOCK(g) Ke=0.0900 at 25°C Calculate the equilibrium concentrations of each gas…
A:
Q: is organic chemistry important? what is covalent bond, explain with diagram
A: Organic compounds are those in which carbon and hydrogen bond is involved. These organic compounds…
Q: Please answer the question as fast as you can. Thank you very much. Which of the following best…
A: The electronic configuration of Flourine is 9F : 1s22s22p5
Q: Without any specific reaction conditions, potentially what functional groups could be formed between…
A: There is no reaction between two acids always acid reacts with base . -> Carboxylic acid reacts…
Q: What are the properties of a buffer solution? Explain.
A:
Q: Would you expect nitroethylene to be more or less reactive than ethylene in a Diels-Alder reaction?…
A:
Q: "Reduction of Cr(VI) to Cr(III) is environmentally favorable as the latter species is not toxic to…
A:
Q: F sp² hybridized trigonal planar Problem 2.30 Which species are Lewis bases? a. NH3 b. CH3CH₂CH3 c.…
A: According to the Lewis definition, the species which donates lone pair of electrons acts as a Lewis…
Q: in a Precipitation Titration of Cobalt Chloride lab Suppose you did not have any indicator…
A: The end point of the titration can easily observed by using indicators, which shows a sharp colour…
Q: The specific rotation of quinine, an anti-malarial drug, is -165°. How much quinine (in %) is…
A: we have to calculate the amount of quinine in % in the sample
Q: Part 3: For each of the following representations of butane, determine what strain energy is…
A:
Q: (9) Given a Covation is c(x₁+)-Co 1-erf ( x 2 Jot CS-Co (9) A Solution of Fick's first lov (b) A…
A:
Q: What is the percent yield of a reaction in which 200. g of phosphorus trichloride reacts with excess…
A:
Q: Modeling lonic and Covalent Bonds 1 In each box, enter the appropriate number of valence electrons…
A:
Q: Problem 4.7 More Practice: Problem 4.8 Give the IUPAC name for each compound. inyt b. C. x d. •Y Try…
A:
Q: Rutile, TiO2, crystallizes in a structure characteristic of many other ionic compounds. How many…
A: First look at the structure, and calculate no. of effective Ti4+ and O2- ions in the unit cell of…
Q: What is the water pressure at a depth of 30 m under sea water (similar to pure water), at 25°C? We…
A:
Q: A 0.100 M solution of an enantiomerically pure chiral compound D has an observed rotation of +0.18°…
A: Given, Molarity : 0.100M observed rotation : +0.18o path lenght= 1 dm Molar mass of the compound :…
Q: A 50.0 mL solution of Ca(OH)₂ with an unknown concentration was titrated with 0.340 M HNO₃. To reach…
A: Calcium hydroxide reacts with nitric acid to form calcium nitrate. This reaction is an example of a…
Q: "Reduction of Cr(VI) to Cr(III) is environmentally favorable as the latter species is not toxic to…
A: We will find order one by one and then rate constant
Q: Calculate the molecular and molar mass of hydrogen fluoride with scientific and decimal notation…
A: Molar mass of compound can be formed by adding mass of each element with its stochiometric…
Q: what's the optimum stucture for NH3s
A: NH3 has a pyramidal structure (on the basis of VSPER theory) where, N has sp3 hybridisation. Three…
Q: Determine the major product of the following reaction Br OH SOCI₂, CH₂Cl₂ reflux ?
A: reagents are is thionyl chloride (SO2Cl2) and dichloromethane act as solvent. In this reaction,…
Q: Na₂CO3 (s) + 2 HCI (aq), Sona CO₂ (g) + H₂O (1) + 2 NaCl (aq)
A: A balanced chemical equation represents number of atoms for each element in the reaction, the total…
Q: One way in which the useful metal copper is produced is by dissolving the mineral azurite,…
A: Given, metal copper is produced is by dissolving the mineral azurite, which contains copper(II)…
Q: Answer the questions in the table below about the shape of the fluoroform (CHF3) molecule. How many…
A: VSEPR theory: The structure of a molecule can be determined by using the VSEPR theory. According to…
Q: H3PO4 + 3NaOH >>> Na3PO4 + 3H2O If you start the reaction with 100. grams of each reactant, which…
A:
Q: With an isothermal process, we expand Nitrogen at 25°C and 1 atm from 0.5 L to 1 L. Then at a…
A: Using corresponding formulas for change in entropy at constant temperature, and at constant…
Q: In this structure, which hydrogen has the higher chemical shift and why?
A: NMR spectroscopy is branch of chemistry in which we deal with shielding and deshielding effect of…
Q: Draw the organic product of the Bronsted acid-base reaction. Include all lone pairs and charges as…
A:
Q: 1. Given the following solvents and their dielectric constants: Hexane 1.88 Benzene 4.6 Ethanol 25…
A: In hexane carbon is Sp3 hybridized Hybrid orbitals are involved in bond formation. The C-C bond is…
Q: 3. Sodium carbonate decahydrate Na CO₂ · 10H₂0 .
A:
Q: A 2.00 liter vessel is filled with NH3(g) to a pressure of 4.00 atm and the reaction N2(g) + 3 H2(g)…
A: Answer: Partial pressure of a gas in reaction mixture is directly proportional to its number of…
Q: the [M+ 2] + Peak be the most useful in the MS analysis of which compound? •) bromo aminoindane 1)…
A: The relative intensity of elements is as O18 = 0.2 % Cl37 = 32 % Br81 = 97 %
Q: 4. Given the half-reactions 2Hg²+ (aq) + 2e2 Cr³+ (aq) + 3e2 below, provide the answers to the…
A: 4. Recall the half-reactions, 2 Hg2+ aq + 2 e- ↔ Hg22+ aq E°…
Q: 4. Calculate the standard potential energy for this cell: 2Na(s) + Fe²(aq) ---> 2Na²+ (aq) + Fe(s)
A:
Q: Complete and balance the following redox equation. What is the coefficient of H₂S when the equation…
A:
Q: Example Q. Salicylic acid (pKa of 3.0). 1. What is the percentage of acid at a pH of 4.5? 2. What is…
A:
Q: Starch is the indicator in 3 component system Select one: True False
A: Starch is a viable indicator in the titration process because it turns deep dark blue when iodine is…
Q: For the reaction, MoS2(g) + 2H2(g) → Mo(s) + 2H₂S(g) AH25°c = 46,670 cal, AG25°C -38,460 cal, and Cp…
A: Given: Cp=-12.95+3.75×10-3 T-0.503×105T2
Q: What is the water pressure at a depth of 30 m under sea water (similar to pure water), at 25°C? We…
A:
Q: Draw the structure of an alkane or cycloalkane that has more than three but fewer than ten carbon…
A: Organic compounds are made up of carbon and hydrogen. These organic compounds are called…
Q: Problem 2.32 Problem 2.33 Problem 2.34 For each reaction, label the Lewis acid and base. Use curved…
A:
Q: 1. How many grams of NH4OH must be dissolved in enough water to make 350 mL solution such that the…
A: 1. A certain amount of NH4OH is dissolved in enough water to make 350 mL of solution in which…
Q: The solubility of ammonium formate, NH4HCO2, in 100. g of water is 102 g at 0 °C and 546 g at 80 °C.…
A: Answer:- This question involves the solubility of a compound at different temperatures. Firstly mass…
Q: What would be the overall charge associated with the tripeptide shown below if it were in a solution…
A: we have to calculate the overall charge of the tripeptide N-lys-glu-phe-C
Q: Which household products are strong acids? Which are strong bases? What pH values are considered…
A:
Q: Propose an efficient synthesis the following transformation: The transformation above can be…
A:
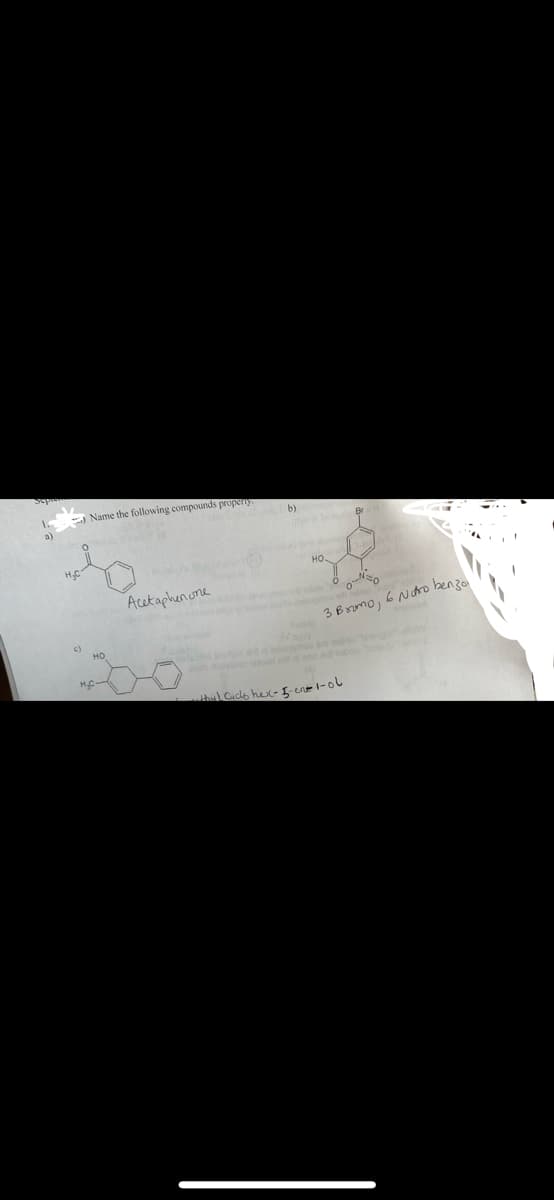
Step by step
Solved in 2 steps

- KMnO4, warm, conc'd reacts with hept-1-ene to yield __________. CO2, hex-1-ene CO2, hexanoic acid Formic acid, pentanoic acid Ethanoic acid, pentanal Formic acid, hexanoneWhich choice best describes the alkene below?A chemist allows some pure (2S,3R)-3-bromo-2,3-diphenylpentane to react with a solution of sodium ethoxide(NaOCH2CH3) in ethanol. The products are two alkenes: A (cis-trans mixture) and B, a single pure isomer. Under thesame conditions, the reaction of (2S,3S)-3-bromo-2,3-diphenylpentane gives two alkenes, A (cis-trans mixture) and C.Upon catalytic hydrogenation, all three of these alkenes (A, B, and C) give 2,3-diphenylpentane. Determine the structuresof A, B, and C; give equations for their formation; and explain the stereospecificity of these reactions
- M 6 write the principal product in a, c & e and the neccesary reactives for b, d & f in the following reactions :Explain the mechanism-Carboxylation—Reaction of RMgX with CO2Name (including E/Z stereochemistry) the five alkenes that can produce 3-bromo-3-methylhexane on reaction with HBr. Draw the skeletal structure of each molecule.
- Predict the major products formed when 2- methyl-1-butene reacts with: H2, Pt/25°C. Show the reaction mechanism the given alkene reactionsA chemist allows some pure (2S,3R)-3-bromo-2,3-diphenylpentane to react with a solution of sodium ethoxide(NaOCH2 CH3) in ethanol. The products are two alkenes: A (cis-trans mixture) and B, a single pure isomer. Under the same conditions, the reaction of (2S,3S)-3-bromo-2,3-diphenylpentane gives two alkenes, A (cis-trans mixture) and C. Upon catalytic hydrogenation, all three of these alkenes (A, B, and C) give 2,3-diphenylpentane. Determine the structures of A, B, and C; give equations for their formation; and explain the stereospecificity of these reactions.As a rule, axial alcohols oxidize somewhat faster than equatorial alcohols. Which would you expect to oxidize faster, cis-4-tert-butylcyclo-hexanol or trans-4-tert-butylcyclohexanol? Draw the more stable chair conformation of each molecule.

