Chapter16: Chemistry Of Benzene: Electrophilic Aromatic Substitution
Section16.SE: Something Extra
Problem 25VC: The following molecular model of a dimethyl-substituted biphenyl represents the lowest-energy...
Related questions
Question

Transcribed Image Text:2.
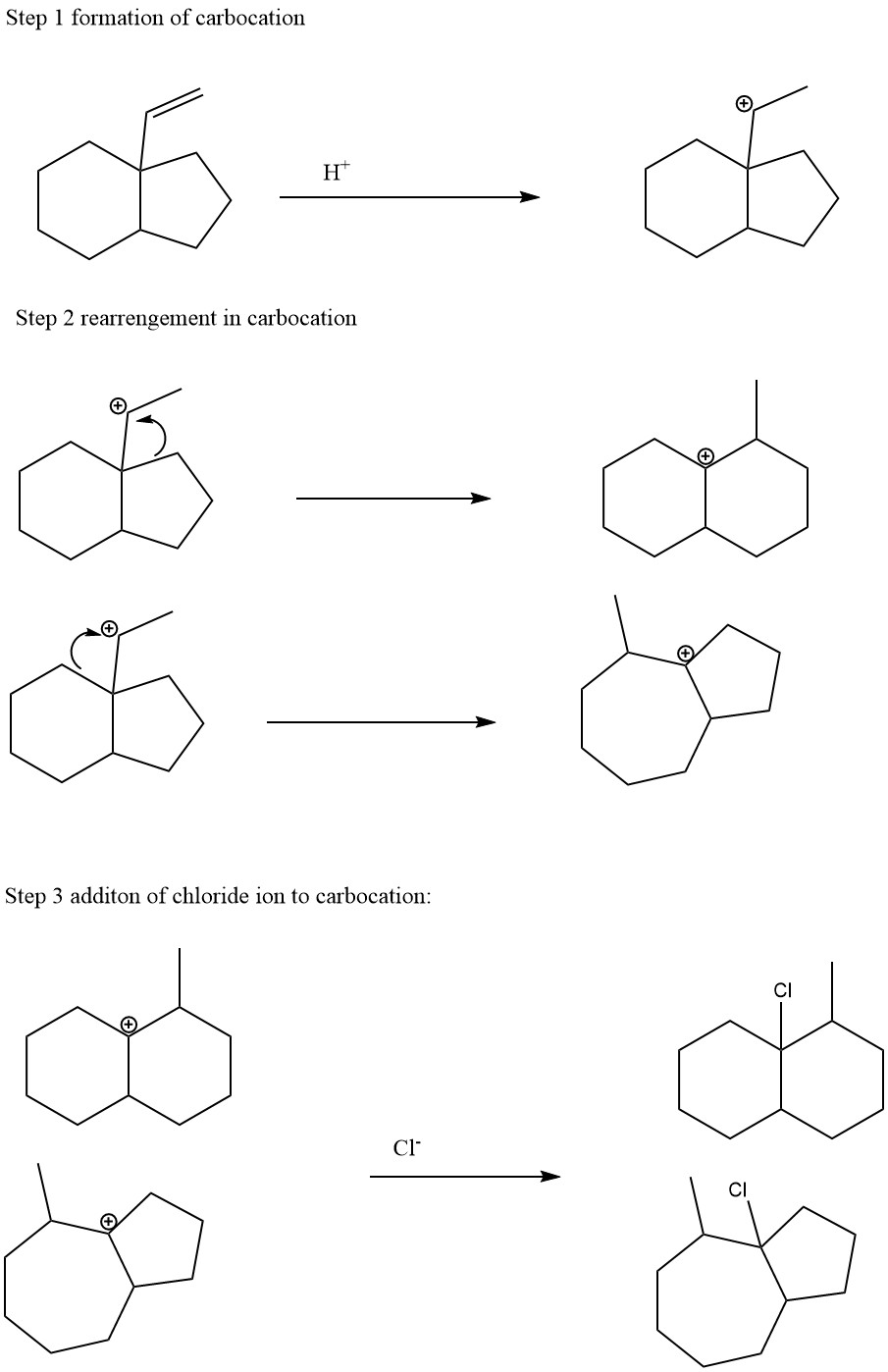
The following reaction results in two possible carbocation rearrangement products, each of which can be
characterized by a change in the ring size. Give structures for both products. There is no need to show the
mechanism of the reaction.
HCI (conc)
Expert Solution
Step 1
The electrophilic addition reaction of alkene proceeds in two steps. In the first step the proton attached to the double bond to form the more stable carbonation intermediate. This carbonation intermediate can be rearranged to even more stable carbonation. In the last step the chloride ion attached to the carbonation to form the final product.
Step 2
The reaction is given below:
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

