Heat is removed from a molten metal of mass 2 kg at a constant rate of 10 kW till it is completely solidified. The cooling curve is shown in the fig. 1100 (0 s, 1023 K) 1000- 900 (20s, 873 K) 800- (10s, 873 K) 700 600- (30 s, 600 K) 500 20 30 Times (s) 10 40 Assuming uniform temperature throughout the volume of the metal during solidifications, the latent heat of fusion of the metal (in kJ/kg) is Temperature (K)
Heat is removed from a molten metal of mass 2 kg at a constant rate of 10 kW till it is completely solidified. The cooling curve is shown in the fig. 1100 (0 s, 1023 K) 1000- 900 (20s, 873 K) 800- (10s, 873 K) 700 600- (30 s, 600 K) 500 20 30 Times (s) 10 40 Assuming uniform temperature throughout the volume of the metal during solidifications, the latent heat of fusion of the metal (in kJ/kg) is Temperature (K)
Principles of Heat Transfer (Activate Learning with these NEW titles from Engineering!)
8th Edition
ISBN:9781305387102
Author:Kreith, Frank; Manglik, Raj M.
Publisher:Kreith, Frank; Manglik, Raj M.
Chapter6: Forced Convection Over Exterior Surfaces
Section: Chapter Questions
Problem 6.26P
Related questions
Question
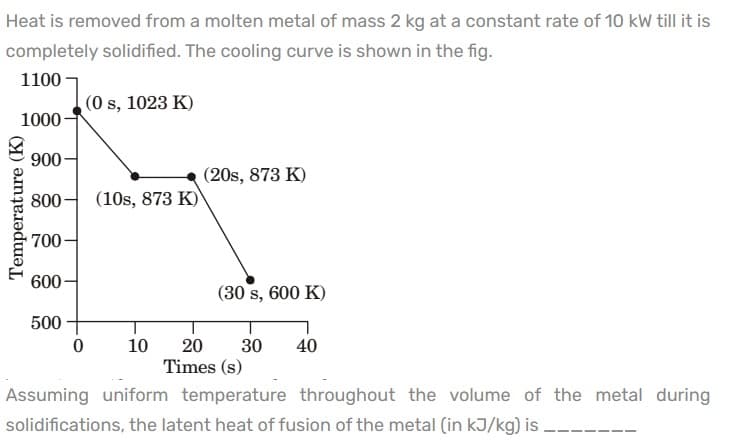
Transcribed Image Text:Heat is removed from a molten metal of mass 2 kg at a constant rate of 10 kW till it is
completely solidified. The cooling curve is shown in the fig.
1100
(0 s, 1023 K)
1000-
900-
(20s, 873 K)
800-
(10s, 873 K)
700
600-
(30 s, 600 K)
500
10
20
30
40
Times (s)
Assuming uniform temperature throughout the volume of the metal during
solidifications, the latent heat of fusion of the metal (in kJ/kg) is –-
Temperature (K)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Recommended textbooks for you

Principles of Heat Transfer (Activate Learning wi…
Mechanical Engineering
ISBN:
9781305387102
Author:
Kreith, Frank; Manglik, Raj M.
Publisher:
Cengage Learning

Principles of Heat Transfer (Activate Learning wi…
Mechanical Engineering
ISBN:
9781305387102
Author:
Kreith, Frank; Manglik, Raj M.
Publisher:
Cengage Learning