Chapter20: Applications Of Oxidation/reduction Titrations
Section: Chapter Questions
Problem 20.6QAP
Related questions
Question
Hello, I need help taking all the information and turning it into a balanced molecular equation. Thank you

Transcribed Image Text:1-A. Zinc with 6M Hydrochloric Acid
Solids do dissolve
h calar Change
Solids do hat preciptes
i.
Observations a.
b.
C.
-Jay
रुपनि
AJo Gas zvolves
No odor
d.
e.
f.
ii. Write a balanced molecular equation for the reaction.
7| EXPT 04
mbes OF protans in
electinn
Expert Solution
Step 1
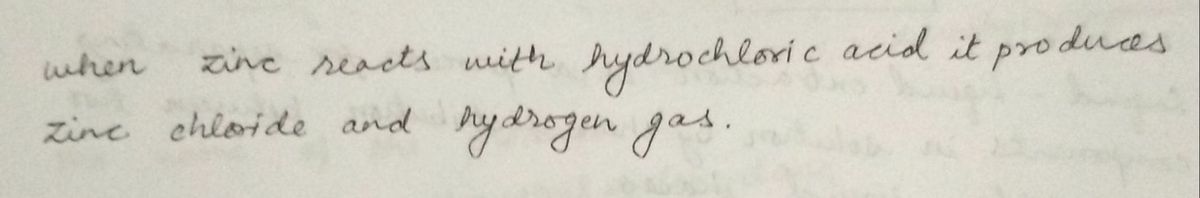
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

