Histamine is synthesized from one of the 20 protein-derived amino acids. Suggest which amino acid is its biochemical precursor and the type of organic reaction(s) involved in its biosynthesis (e.g., oxidation, reduction, decarboxylation, nucleophilic substitution). CH,CH,NH, H. Histamine
Histamine is synthesized from one of the 20 protein-derived amino acids. Suggest which amino acid is its biochemical precursor and the type of organic reaction(s) involved in its biosynthesis (e.g., oxidation, reduction, decarboxylation, nucleophilic substitution). CH,CH,NH, H. Histamine
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter27: Amino Acids And Proteins
Section: Chapter Questions
Problem 27.19P
Related questions
Question
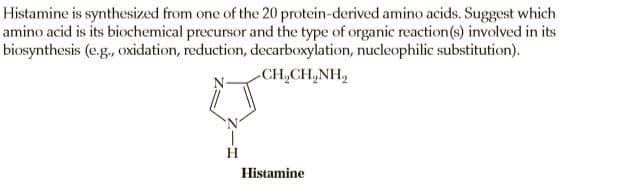
Transcribed Image Text:Histamine is synthesized from one of the 20 protein-derived amino acids. Suggest which
amino acid is its biochemical precursor and the type of organic reaction(s) involved in its
biosynthesis (e.g., oxidation, reduction, decarboxylation, nucleophilic substitution).
CH,CH,NH,
H.
Histamine
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 3 images

Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole