Q: Which of the following is the weakest intermolecular force? ionic bonding dipole-dipole…
A:
Q: 12. Determine the normal boiling point for the liquid with the strongest intermolecular forces. 20…
A:
Q: 1 hentary reaction N₂0(g) → N₂(g) + O(g) - k₁ N₂(g) + O₂(g) k₂. Suppose also k₁k₂. That is, the…
A: We have to find overall, rate and rate constant values.
Q: Calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen,…
A: The formation of nitric acid occurs in two steps. From the chemical equation and enthalpies of these…
Q: 8.82*10^-2 of potassium bromide has an enthalpy change of 1620J when it dissolves in water. What is…
A:
Q: A chemist carefully measures the amount of heat needed to raise the temperature of a 0.85 kg sample…
A: Molar heat capacity = q/n∆T q = heat absorbedn = number of moles of substance∆T = T2 - T1 T1 =…
Q: Draw the skeletal (line-bond) structure for (S)-2- isopropylheptanoic acid. Use a dash or wedge bond…
A: Given,The name of the molecule is: (S) - isopropylheptanoic acid.
Q: 1.0804 2. NMO
A: Dihydroxylation involves using catalytic OsO4 and stoichiometric oxidant that will convert Os(VI)…
Q: Suppose 10.4 g of copper(II) acetate is dissolved in 50. mL of a 0.80 M aqueous solution of sodium…
A: Mass of copper (II) Acetate = 10.4 gVolume of sodium chromate solution = 50 mLConcentration of…
Q: = OTHERMOCHEMISTRY Calculating the heat of reaction from molar reaction enthalpy a... A chemist…
A: Endothermic reaction : It is a chemical reaction in which energy is absorbed by the system from its…
Q: Sometimes in lab we collect the gas formed by a chemical reaction over water (see sketch at right).…
A: Temperature = 45.0 oCVVolume of gas = 116 mL
Q: 5. For the reaction below, initially 3.5 mol of NH¸ are placed in a 4.0 L reaction chamber. After…
A:
Q: Examine the curved arrow mechanism below, and answer the question in the box. If the mechanism is…
A: During mechanism flow of electrons occur from higher electron density to lower electron densityA…
Q: For an electrochemical cell described as follows, what would be the balanced cell reaction indicated…
A: In an electrochemical cell, electrical energy is produced from the spontaneous cell reaction at the…
Q: When 605. mg of a certain molecular compound X are dissolved in 100 g of cyclohexane (CH₂), the…
A: GivenWeight of unknown compound X = 605. mg = 0.605 gWeight of solvent = 100. gFreezing point of…
Q: Draw the product of this reaction. Ignore inorganic byproducts. H 1. NaNHa
A: Sodium Amide (Sodamide, NaNH2), A Strong Base For The Deprotonation Of Terminal AlkyneA molecule…
Q: Draw the skeletal (line-bond) structure for 2-isopropylheptanoic acid. Drawing
A: A representation of molecular structure in which covalent bonds are represented with one line for…
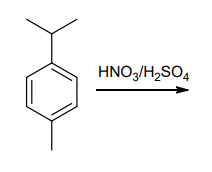
Q: NH₂ CH3 HNO3 H₂SO4
A:
Q: The pH at one half the equivalence point in an acid-base titration was found to be 6.57. What is the…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: In the laboratory, a general chemistry student measured the pH of a 0.350 M aqueous solution of…
A: Given:The concentration of the aqueous solution of pyridine (C5H5N) is 0.350 MThe value of pH is…
Q: Name all the intermolecular forces acting within the particles of the following substances.…
A: The intermolecular forces present in each substances are :---1. Pentane, (C5H12 ) :---Pentane is a…
Q: Which of the following is a characteristic of typical alkaline dry batteries? Question 1 options:…
A: An alkaline dry cell battery is a specific type of dry cell battery that utilizes an alkaline…
Q: When the Hg2+ concentration is 3.34×10-4 M, the observed cell potential at 298K for an…
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: You are studying the combustion of methane by the following unbalanced chemical reaction: a CH4(g) +…
A: In the combustion reaction, a fuel is burnt in oxygen and releases energy in the form of heat. This…
Q: he solubility of compound A in ether is 0.6 g/100 mL and its solubility in water is 0.12 g/100 ml.…
A: given the data,A dissolves in ether 0.6 g/100 mL.Water solubility of A is 0.12 g/100 mL.60 mL is the…
Q: What is X as Product?
A: Alkenes undergo oxidative cleavage by reaction with KMnO4 to form carboxylic acid or ketone.
Q: Which of the following does not have an electron configuration ending with 2S^2 2P^6? chlorine ion…
A: we have to select the ion that does not have an electron configuration that ends as
Q: Which is the least stable A. 'Br B. E. O Br ? Br O C. O Br D. O =0 O Br
A: The delocalization of electrons through pi bonds is simply called resonance. The resonance is driven…
Q: In the following net ionic equation, identify each reactant as either a Bronsted-Lowry acid or a…
A: To Solve this problem first we have to know about Brønsted–Lowry acid–base theory . According to…
Q: Problem 4: Identify the expected major product(s) for the following reaction. HBr 40 °C ?? Br + En…
A:
Q: Using the information below, calculate AHf for PbO (s) PbO (s) + CO (g) Pb (s) + CO2 (g) AHf for CO₂…
A:
Q: Draw the major product of this reaction. Ignore inorganic byproducts. DMP (Dess-Martin Periodinane)…
A: Dess–Martin periodinane (DMP) is used for the oxidation of primary alcohols to aldehydes and…
Q: Which of the following structures contribute(s) to the resonance hybrid of the intermediate formed…
A:
Q: What are the major species present when 10 mL of 0.1 M NaOH are added to 40 mL of 0.05 M acetic acid…
A: When a strong base like NaOH is added to a weak acid like CH3COOH, it undergoes a neutralization…
Q: A. Types of Unsaturated Hydrocarbons Models of Unsaturated Hydrocarbons Compound Condensed…
A: As per guidelines, we can answer first question only
Q: w the missing products and/or reagents in the following multistep synthesis. 8 OH Select to Draw NaH…
A:
Q: 4. A chalcone derivative shows the following UV spectrum. Solvent is ethanol, 1 cm cuvette and the…
A: According to Beer's Lambert law, A = €Cl Where A = Absorbance € = Molar extinction…
Q: A chemist measures the energy change AH during the following reaction: 2 Fe₂O3(s)→ 4 FeO(s) + O₂(g)…
A: If ∆H<O, reaction is exothermicIf ∆H>0, reaction is endothermic
Q: Suppose 0.452 g of sodium iodide is dissolved in 100. mL of a 14.0 m M aqueous solution of silver…
A: Mass of NaI = 0.452 gVolume of AgNO3 solution = 100 mLConcentration of AgNO3 = 14.0 M
Q: what is the stochiometry from triphenylmethanol to triphenylmethyl methyl ether? Please explain?
A: triphenylmethanol to triphenylmethyl methyl ether.
Q: Determine the number of grams of HCl that can react with 0.750 g of Al(OH), according to the…
A:
Q: During an experiment, vinegar was added to baking soda in a container. The container's temperature…
A: .......
Q: Unknown analysis Complete the table below to classify your unknown substance. Write "+" if the test…
A: 1. .....
Q: A compound has a molar mass of 28.06 g. What is its chemical formula if carbon and hydrogen are…
A: Molecular formula: In a molecular formula we describe the number of atoms of all the elements…
Q: Rank the compounds in each set from most to least reactive in an EAS reaction
A: Rate of electrophilic aromatic substitution depends on the electron density of the benzene ring.…
Q: In the following equation for a chemical reaction, the notation (s), (I), or (g) indicates whether…
A: A process in which reactants are converted into products is known as a chemical reaction.
Q: Draw the possible products of this epoxide ring- opening reaction. Use a dash or wedge bond to…
A: Epoxide ring opening is a chemical reaction in which an epoxide ring (a three-membered cyclic ether)…
Q: An aqueous solution has a hydroxide ion concentration of 1.0 × 10-¹1 M. What is the hydronium ion…
A:
Q: Consider the titration of 500 mL of 100 mM boric acid. The pka of boric acid is 9.23. What will the…
A: Boric acid formula is which react with sodium hydroxide to form .Reaction is
Q: Apply Hess' Law using the step-wise reactions below to determine the change in enthalpy associated…
A:
Provide the missing structures
Step by step
Solved in 3 steps with 2 images


