Q: Question 37: Consider the HNO3 molecule. Which of the following would not be a major step when…
A:
Q: Draw arrows to indicate the movement of electrons in the following reaction. Which direction will…
A: A question based on reaction mechanisms. A reaction scheme is given whose mechanism is to be…
Q: 3. Indicate whether each of the following structures is (R)-2-chlorobutane or (S)-2-chlorobutane (if…
A: The R and S configuration is a nomenclature used for naming enantiomers of a chiral compound. you…
Q: Macmillan Learning Predict the major product for each of the following reactions: Cl₂ Product 1 Cl₂…
A: Alkynes on addition with halogen first form vicinal dihalides alkene, which on further reaction with…
Q: Convert 5.32 × 10⁻⁷ s to microseconds . Use only the metric system.
A: Time = Convert the given time into microseconds =?
Q: 0.120 moles of C is reacted with 0.117 moles of O₂ to form CO and CO₂. 3C(s) +20₂(g) → CO₂(g)…
A: The limiting reagent is the compound which is completely consumed during the reaction. It can be…
Q: A bottle of nitric acid contains 0.250 L of 0.500 mol/L HNO3. The bottle is diluted up to 1.00 L…
A: After dilution of any stock solution, the distance between solute particles in the solution…
Q: In the following reaction, what are the correct coefficients for S and Mn²*? H* + MnO4² +S→→Mn²+ +…
A:
Q: Questions Draw the structures and provide the IUPAC names of the product(s) of the reaction of…
A: Since, there is multiple questions are present. We solve only first question for you according to…
Q: One of the intermediates in the synthesis of glycine from ammonia, carbon dioxide, and methane is…
A: The reaction in which the reactant is totally consumed when the reaction is completed is known as…
Q: QUESTION 14 Does Substrate 3 undergo a carbocation rearrangement in an SN1 mechanism? SUBSTRATE 1…
A: stands for substitution nucleophilic unimolecular. Substitution is the reaction where one group is…
Q: Calculate the pH of a weak acid solution (quadratic equation). Calculate the pH of a 0.0167 M…
A:
Q: SiH4 (g) + 2 O2 (g) -> SiO2 (s) + 2 H20 (l) at 25ºC; where ΔН°rxn = -1 516 kJ and ΔSºrxn = -432.8 J…
A:
Q: It takes 42min for the concentration of a reactant in a first-order reaction to drop from 0.45 M to…
A:
Q: The K for benzoic acid is 6.5 x 105. Calculate the pH of a 0.24 M benzoic acid solution. Round your…
A: The Ka for benzoic acid is 6.5×10-5.We have to calculate the pH of 0.24 M benzoic acid solution.
Q: What set of reagents would be needed to perform the following reaction? ? OH Either H₂O/H3O+ or 1)…
A: 1. Hg(OAc)2, H2O 2. NaBH4: Converts alkene to alcohol. Product is Marovnikoff1. BH3 2. H2O2, OH-:…
Q: M HN J
A: This is an example of epoxide opening reaction. It involves SN2 mechanism that leads to inversion in…
Q: Make a 1.0 L of a 0.10 M sodium phosphate buffer at pH = 7.8. ck solutions are H2PO4- = 1.0 M and…
A: "Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Does Substrate 2 undergo a carbocation rearrangement in an SN1 mechanism? CI ya SUBSTRATE 1 Yes No…
A: Carbocation rearrangement refers to the intramolecular migration of a carbon atom with a positive…
Q: Consider a binary mixture for which the excess Gibbs energy is given by GEIRT = 2.6x1x2. For each of…
A: Binary liquid mixtures are liquids that are composed of two different molecular components, A and B.…
Q: A 650.-mL sample of nitrogen gas is collected at 27.0°C and a pressure of 638 torr. What volume will…
A: Provided, the initial conditions are asPressure (P1)=638 torrTemperature(T1)= (27+273.15) K or…
Q: Which of the structures shown below the IR spectrum matches the IR spectrum below. Relative…
A: Spectroscopy is an analytical technique that is aimed at elucidating the structure of a compound.…
Q: Calculate the number of moles of aluminum and silicon required to produce 373.0 g of chromium based…
A: Number of moles is given by:
Q: 4. Write a balanced equation for the combustion of propane, C3Hs. When balanced, the equation…
A: According to Q&A guidelines of Bartleby, we are supposed to answer only the first question out…
Q: Give only typing answer with explanation and conclusion
A: A question based on preparation of amines. An enamine is given whose product of reaction with…
Q: Consider the reaction carried out at a standard temperature of 25 °C. Use the thermodynamic data…
A:
Q: Which of the compounds match the IR spectrum shown below? Answer choices are shown below the…
A: Find the compound based on IR spectrum Based in Standard Ir frequencies of functional groups, we can…
Q: Combustion reactions involve reacting a substance with oxygen. when compounds containing carbon and…
A: The enthalpy of combustion of a substance is defined as the heat energy given out when one mole of a…
Q: D Question 2 Classify each property as physical or chemical. a) the explosiveness of hydrogen gas…
A:
Q: What would be the major product of the following elimination reaction? Br O O NaOH A There is no…
A:
Q: Assume that we have a vapour consisting of isolated atoms. Which of the following vapours would…
A: An atom will be a paramagnetic, if it contains unpaired electron. If it contains paired electrons,…
Q: Use the dropdowns to create a correct and complete isotope notation. An isotope has 108 neutrons…
A: Given,Number of neutrons in an isotope = 108Number of protons in an isotope = 72
Q: Write the equilibrium-constant expression for the reaction shown in terms of [SO2], [CO2], [CS2],…
A:
Q: The half-life for a first order reaction is 85min. What is the rate constant?
A:
Q: How many grams of Al(NO3)3 are present in 12.1 g of a(n) 18.4% by mass Al(NO3)3(aq) solution? Enter…
A:
Q: Which reaction below would be the fastest? A. CH3CH2 B. С. CH3CH2 OA OB CH₂CH₂ C OTOS C-CH2CH3 + он…
A: The leaving group will be a good leaving group if it is more stabilised. So more stable the anion,…
Q: A gas mixture contains the noble gases Kr, Ne, and Xe. The total pressure of the mixture is 4.76…
A: The relation between partial pressure, total pressure, and mole fraction is given as:Based, on this…
Q: Multiply or divide the following measurements. Be sure each answer you enter contains the correct…
A: There are some rules of multiplying and dividing with units . If we multiply the term with units ,…
Q: he values of ∆H◦, ∆S◦, and the equilibrium constant, K, at 50.0◦C. At what temperature will the…
A: we can use the Van't Hoff equation, which relates the equilibrium constant (K) to the standard…
Q: Click the "draw structure" button to launch the drawing utility. Draw the organic product formed in…
A: IntroductionThe study of substances containing carbon is the focus of the chemical subfield known as…
Q: Consider the reaction shown below. At a particular time, What is rate of appearance of Br2 at the…
A:
Q: How to prepare 250cm3 of a solution of carbonate with concentration 0.100 mol dm-3
A: Here , we have asked that how to prepare 250cm3 of a solution of carbonate with concentration…
Q: Inside an industrial unit, the hourly average concentration of NO2 is measured at 0.12 ppm (by…
A: The relation between and ppm can be given as, According to the ideal gas equation,PV = nRTR =…
Q: Question 11 Draw all the product(s) for each reaction below. If there are two identical molecules…
A: This reaction is the hydrolysis of acetal in acid condition.At first in this reaction presence of…
Q: Show ions that will not have ion -dipole intermolecular force by finding net ionic equation for…
A: Answer:-This question is answered by using the simple concept of writing the net ionic equation.
Q: Write a balanced chemical equation for each reaction.
A: A balanced chemical equation is an equation in which all the atoms of each species should be equal…
Q: Specify the reagent you would use in each step of the following synthesis: a. LIAIHA b. H₂SO4 c. HCI…
A: A question based on reactions of ketones. A reaction scheme is given in 2 steps, for which…
Q: A 15.2815.28 g sample of a compound contains 9.759.75 g of iron, Fe,Fe, 1.801.80 g of phosphorus,…
A: Total mass of compound = 15.28 gMass of iron = 9.75 gMass of phosphorus = 1.80 g
Q: Consider the hypothetical reaction A + 3B → 2C, and match the various rates with the correct…
A:
Q: A tank with 43.2 kg of water is leaking at a rate of 0.0135 kg/s. How many hours will it take until…
A:
name of the compound and its pharmaceutical applications?
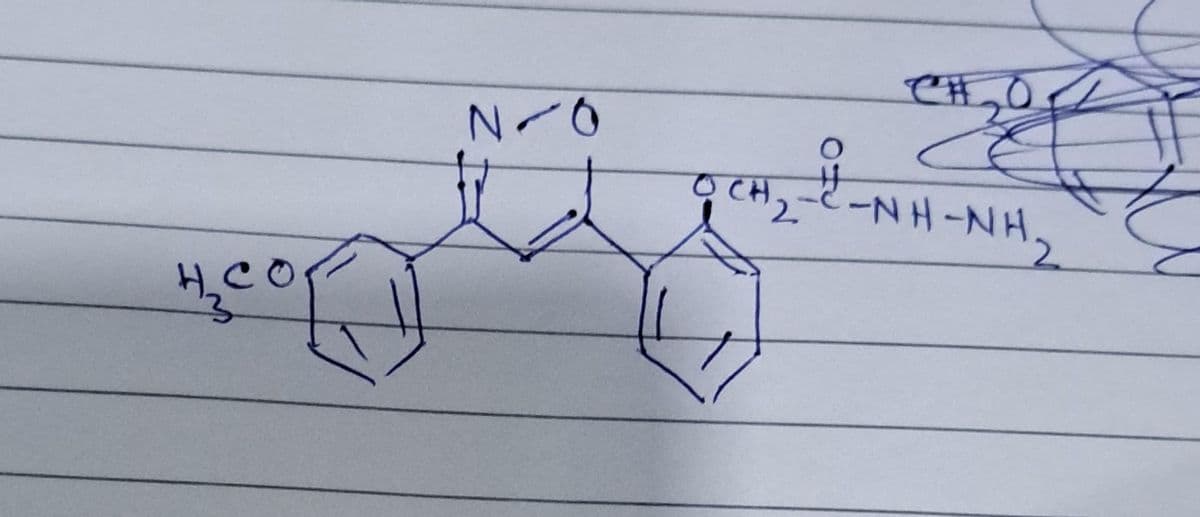
Step by step
Solved in 4 steps with 1 images


