Q: Draw a skeletal (line - bond) structure of (S) - 2, 2, 3trimethylhexane, Use a dash or wedge bond to…
A: 11. (S)-2,2,3-trimethylhexane12. (S)-4-ethyloctane
Q: Tw_wmm IR Spectrum (liquid film) 4000 00 80 60 -40 20 % of base peak 3376 40 10 3000 59 13C NMR…
A: The question is based on the concept of organic spectroscopy. We need to analyse the spectral data…
Q: Draw the enolate anion and the carbonyl that would be needed to make this product through an aldol…
A: An enolate ion is an anion formed by the removal of an alpha-hydrogen atom from a carbonyl compound,…
Q: Draw a tetramer of this alternating copolymer. OH OH + OH H3O+, heat *OH 쌓 (0)
A: Tetramer and copolymer
Q: Draw the major organic product for the reaction: Br NaCl H₂O Select Draw с Rings H Cl More Erase
A: Given that, the reaction is:
Q: What is the name of the compound shown below? (A) (B) n-octane n-pentane n-propane n-hexane H, H…
A: The Newman projection represents the dihedral angles of different conformations of a molecule. In…
Q: 4) Determine cell potential, at 298 K, for the following cell if the copper ion concentration is…
A: The electrochemical cell described involves a redox reaction between copper (Cu) and iron (Fe)…
Q: Draw and name all of the monochorination products that you might obtain from the radical…
A: The monochorination of the given company are as follows:
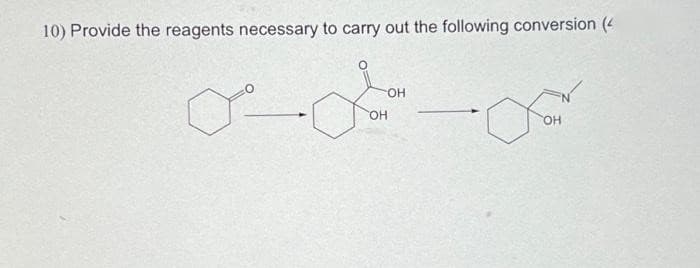
Q: Synthesize the (target) molecules on the right from the starting molecules the left. Give reagents…
A: The question is based on the concept of organic synthesis. We need to synthesize the product using…
Q: 69.0 g of AgNO₃ (FW 169.87 g/mol) is added to a solution containing 45.0 g of CaBr₂ (FW 199.89…
A: The question is based on the concept of reaction stoichiometry.We need to calculate mass of…
Q: Consider the reaction H₂(g) + F₂(g) 2HF(g) Using standard thermodynamic data at 298K, calculate the…
A:
Q: Which of the following syntheses are suitable to prepare the given target molecule (as the major…
A: Given is organic compound. Given compound is alkene.
Q: Calculate the equilibrium constant at 25.0°C for each of the following reactions from the value of…
A: The equilibrium constant (K)=?
Q: What values of J may occur in the terms 3D, 4D, and 2G? How many states (distinguished by quantum…
A: The four types of quantum numbers principal, azimuthal, magnetic and spin. The principal quantum…
Q: Aqueous sulfuric acid (H₂SO4) will react with solid sodium hydroxide (NaOH) to produce aqueous…
A: First calculate the number of moles for H2SO4 and NaOH. Use that moles to calculate the mass of…
Q: Propose structures for compounds that fit the following ¹H NMR data: (b) C³H5Br (a) C5H10O 0.95 8 (6…
A: Givena): Molecular formula C5H10Ob): Molecular formula C3H5Br
Q: [A], (M) 0.150 0.450 0.450 Using the information in the table, the value of the rate constant for…
A:
Q: 9.30 Draw out the products of the following reactions and then use curved arrow notation to show the…
A: Given are organic reactions.Let's see products in next step.
Q: How many grams of dipotassium phthalate (242.3 g/mol) must be added to 700 mL of 0.175 M potassium…
A:
Q: What is the osmotic pressure of a solution made from dissolving 65.0 g of glucose, C6H12O6, in 475…
A: Given: Mass of glucose = 65.0 g Volume of solution = 475.0 ml = 0.475 L (Because 1ml =…
Q: Specify the reagent you would use in each step of the following synthesis: a. LiAIH4 b. H₂SO4 c. HCI…
A: Given,The two step reaction is:
Q: According to the ideal gas law, a 1.039 mol sample of nitrogen gas in a 1.269 L container at 266.5 K…
A: Answer:Given data:
Q: Classify the following reaction as: addition, elimination, substitution or rearrangement. Nal A)…
A: The question is based on organic reactions. We need to identify the product and explain its…
Q: A miniscule pinhole develops in the wall of a tank, and every molecule of the gas inside that…
A: Given,Diameter of round hole = 0.01 mmTemperature of the gas = = (273 + 25) K = 298 KPressure of…
Q: Which is an AB3 molecule that has μ ‡0? O PC13F2 O SeCl₂ O NBr3 O SeO₂ O Bl3
A:
Q: Calculate the total volume of gas (at 29 °C and 749 mm Hg) produced by the complete decomposition of…
A: The Balanced Equation:Temperature = 29 oC and Pressure = 749 mm Hg Complete decompostion of 1.36 Kg…
Q: Problem 1.8 What is the formal charge on the O atom in each of the following species that contains a…
A: Formal charge: The formal charge of an atom in a polyatomic molecule or ion may be defined as the…
Q: Isotonic saline solution, which has the same osmotic pressure as blood, can be prepared by…
A: mass of NaCl = 0.923 gVolume of the solution (V) = 100. mL = 0.1 LTemperature (T) = 25°C = (25 +…
Q: (c) 4-Methyl-4-hexene The name is incorrect because the numbering of the chain is not correct V .…
A: The given compounds contain an alkene functional group. For the IUPAC name of an alkene, first of…
Q: Select the right statement about the following reaction. th 1 equ. HBr Br O The given product is…
A: Given,The reaction:
Q: 5. The following reaction forms three possible products. Please indicate which are stereoisomeric…
A: Methanol is a weak nucleophile and weak base so it favours SN1 and E1 mechanism . SN1 favours…
Q: Consider the following system at equilibrium where ΔΗ° - 111 kJ, and == Kc = 0.159, at 7.23 × 10² K:…
A: The question is based on the concept of Le-chatlier principle. It States that when a system at…
Q: The equilibrium constant, Kc, for the following reaction is 55.6 at 698 K. H₂ (9) + I₂ (g) → 2HI(g)…
A: The initial moles of H2 = 0.316 molThe initial moles of I2 = 0.316 molThe equilibrium constant of…
Q: What do the masses of 2-naphthyl-butylether say about what functional groups are present?
A: A question based on mass spectrum. An organic molecule and its mass spectrum are given that are to…
Q: Provide the hybridization and molecular geometry for the indicated atoms in the following molecules.
A:
Q: Choose the reagent(s) that would be most likely to complete this reaction. Q
A: The alkene can undergo an addition reaction to given substituted alkane. When bromine (Br2) reacts…
Q: P Please don't provide the handwriting solution
A: Whether equilibrium lies on the right side or left side depends upon the acidity of the reactant and…
Q: Identify the MAJOR product that would be obtained from the following reaction. Br Br نے Br Br₂ CH3OH…
A:
Q: Osmosis is the process responsible for carrying nutrients and water from groundwater supplies to the…
A: osmotic pressure is the minimum pressure required to stop osmosis i.e. flow to solute molecule from…
Q: whivh of the following is not a bronsted lowry base? al ch3oh bl ch3och3 c) ch3nh2 d) ch3ch3…
A: Substance that can accept proton, H+ are called Bronsted-Lowry base.
Q: How much energy is contained in 1.00 mole of 505 nm photons? (answer is assumed to be in Joules)
A: The energy of photon can be calculated by the formula shown below.
Q: Devise an efficient synthesis of the diol shown below starting with…
A: To draw the structure of 1,1,3,3-tetramethyl-2-ethylcyclohexane, take cyclohexane and add four…
Q: Predict the product for the following reaction. To preview the image click here :0: HO OA B :ÖH A H…
A: NaBH4 is a weak reducing agent and it reduces aldehyde and ketone to alcohol but it cannot reduce…
Q: The equilibrium constant, Kc, for the following reaction is 7.00 × 10-5 at 673 K. NH₂I(s) ⇒ NH3 (g)…
A: Answer:For any given reaction, value of equilibrium constant KC is equal to the ratio of molar…
Q: Yesterday in class, we talked about Zeop (Z*) increasing in the following systems: = 738 KJ/mol =…
A: According to Slater's rule,Where Zeff is the effective nuclear charge, Z is the atomic number, and S…
Q: You mix 55.0 mL of 0.10 M HCl and 110.0 mL of 0.050 M NH3. What is the pH of the resulting solution?…
A:
Q: 9 it Ho H(43)2 -Br + a 10
A: Given is organic reaction ..We have to synthesize following compound,
Q: 6. Calculate the number of moles of NaOH that must be added to 1.0 L of 0.50 M HC₂H₂O₂ to produce a…
A:
Q: a. 94Be + 4₂He → ¹26C + ? b. Be +¹1H → ₂He + ? C. 3015P →3014Si + ?
A: In nuclear reactions, the sum of the atomic number and mass number of all the species will be same…
Q: The equilibrium constant, Ke, for the following reaction is 7.00 x 105 at 673 K. NH,I()NH₂(g) +…
A: Then given equilibrium reaction is The equilibrium constant of the reaction,
Please don't provide hand written solution....
Step by step
Solved in 3 steps with 2 images


