How could you convert butanoic acid into the following compounds? Write each step showing the reagents needed. (a) 1-Butanol (b) 1-Bromobutane (c) 1-Butene
How could you convert butanoic acid into the following compounds? Write each step showing the reagents needed. (a) 1-Butanol (b) 1-Bromobutane (c) 1-Butene
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter7: Alkynes
Section: Chapter Questions
Problem 7.27P: Following is the structural formula of the tranquilizer meparfynol (Oblivon). Propose a synthesis...
Related questions
Question

Transcribed Image Text:How could you convert butanoic acid into the following compounds?
Write each step showing the reagents needed.
(a) 1-Butanol (b) 1-Bromobutane (c) 1-Butene
Expert Solution
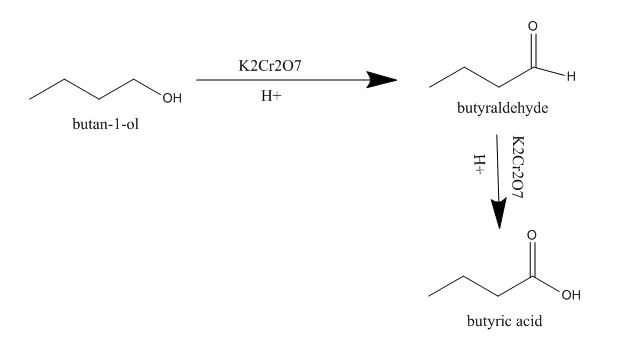
Step 1 Butanol to Butanoic Acid
Butanol or 1-Butanol is a primary alcohol. The aim is to convert it into Butanoic Acid. Basically, alcohol can be converted into carboxylic acids by oxidation process. Some strong oxidizing agents like KMnO4/H+ directly convert the alcohols into acids whereas some mild oxidizing agents like K2Cr2O7/H+ does the job in a step wise manner , i.e. by first converting it into aldehyde and again converting it into carboxylic acid.
Step 2
Butanol to Butanoic Acid-
In the first step, Butanol is converted to Butanal or Butyraldehyde and then it is converted to Butanoic acid or butyric acid
Step by step
Solved in 4 steps with 5 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning