Chapter1: Temperature And Heat
Section: Chapter Questions
Problem 100P: One easy way to reduce heating (and cooling) costs is to add extra insulation in the attic of a...
Related questions
Question
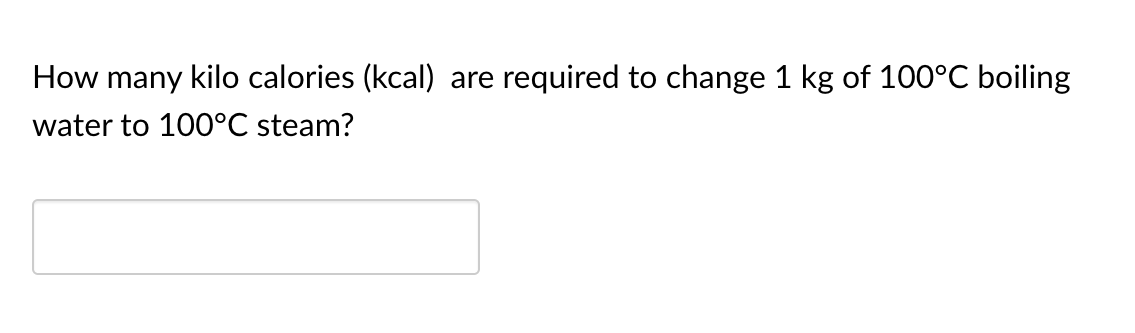
Transcribed Image Text:How many kilo calories (kcal) are
required to change 1 kg of 100°C boiling
water to 100°C steam?
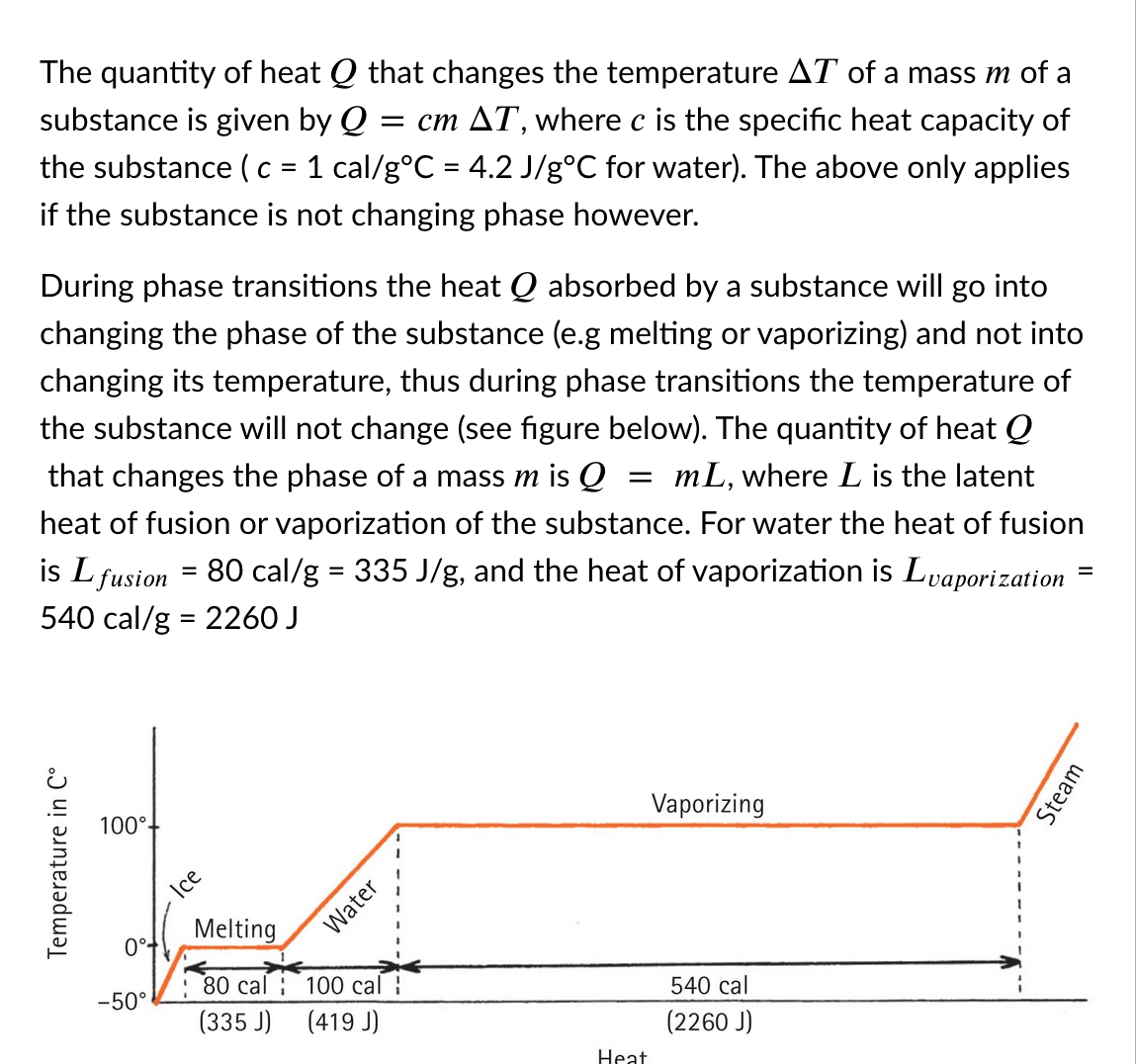
Transcribed Image Text:The quantity of heat Q that changes the temperature AT of a mass m of a
substance is given by Q
= cm AT, where c is the specific heat capacity of
the substance (c = 1 cal/g°C = 4.2 J/g°C for water). The above only applies
if the substance is not changing phase however.
During phase transitions the heat Q absorbed by a substance will go into
changing the phase of the substance (e.g melting or vaporizing) and not into
changing its temperature, thus during phase transitions the temperature of
the substance will not change (see figure below). The quantity of heat Q
that changes the phase of a mass m is Q
mL, where L is the latent
heat of fusion or vaporization of the substance. For water the heat of fusion
80 cal/g = 335 J/g, and the heat of vaporization is Lvaporization
is L fusion
540 cal/g = 2260 J
100°-
Vaporizing
Ice
Melting
0°-
80 cal ; 100 cal :
-50°
540 cal
(335 J) (419 J)
(2260 J)
Heat
Temperature in C°
Water
Steam
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you


An Introduction to Physical Science
Physics
ISBN:
9781305079137
Author:
James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar Torres
Publisher:
Cengage Learning


An Introduction to Physical Science
Physics
ISBN:
9781305079137
Author:
James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar Torres
Publisher:
Cengage Learning