Q: How many grams of oxygen are formed when 4.063 grams of KO are reacted? 4 KO(s) + 2H2O() 4 KOH(s) +…
A: Molarity= moles of solute/ volume It expresses in mol/L. Limiting reagent is the reagent which…
Q: Iodine and chlorine react to form Iodine pentachloride as shown below Unbalanced equation: I2 +…
A: Answer:- This question is answered by using the simple concept of stoichiometry which involves the…
Q: How many moles of NH3 are produced when 0.45 moles of H2 gas react with N2 gas? N2 + 3H2 -->2 NH3…
A: The balanced reaction taking place is given as, => N2 + 3 H2 -----> 2 NH3
Q: For the reaction 2 C4H10 + 13 O2 → 8 CO2 + 10 H2O, if 51.60 g of CO2 is produced, how much C4H10 was…
A: 2C4H10 + 13O2 → 8CO2 + 10H2O From this Balanced chemical equation we get that ; 2 mol C4H10 produce…
Q: Hydrogen gas and nitrogen gas react to give ammonia gas. How many moles of hydrogen are needed to…
A: The mole (symbol: mol) is the unit of measurement for amount of substance in the International…
Q: copper reacts with silver nitrate. if 5.5 moles of copper are used to react, how many moles of…
A: The reaction of copper with silver nitrate is given as , 2AgNO3 + Cu ------> Cu(NO3)2 + 2 Ag .
Q: Iron (III) sulphate reaction with sodium hydroxide according to the balanced equation below. What…
A: The chemical reaction taking place is: Fe2(SO4)3 + 6NaOH ⟶ 2Fe(OH)3 + 3Na2SO4 The given information…
Q: 2 N2H4(1) + N204(1) → 3 N2(g) + 4 H2O(g) Suppose we mix together 50 molecules of N2H4 and 45…
A: N2H4 reacts with N2O4 to form N2 and water. The equation for the balanced chemical reaction is as…
Q: O2 + 2 Mg → 2 MgO If 4 moles of magnesium totally reacted with more than enough O2, how many moles…
A:
Q: What mass of carbon dioxide could be produced from the combustion of 28.15 g heptane (C7H16)…
A: The balanced reaction given is, Given: Mass of C7H16 reacting = 28.15 g.
Q: A container holds 100. g of P₄ and 150g of O₂.The following reaction occurs: P₄ + 3O₂ → P₄O₆. Which…
A: When a chemical reaction between the reactants occurs, they combine in a specific stoichiometric…
Q: How many moles of calcium chloride are produced if 0.112 mol of calcium reacts? Ca + Cl2 = CaCl2
A: 1 Ca + Cl2 = 1 CaCl2 From the balance reaction, we can see that 1 mol of Ca produced 1 mol of…
Q: How many moles of potassium nitrate are produced when 2.08 moles of potassium phosphate react ?
A: given: K3PO4+Al(NO3)3-->3KNO3 + AIPO4
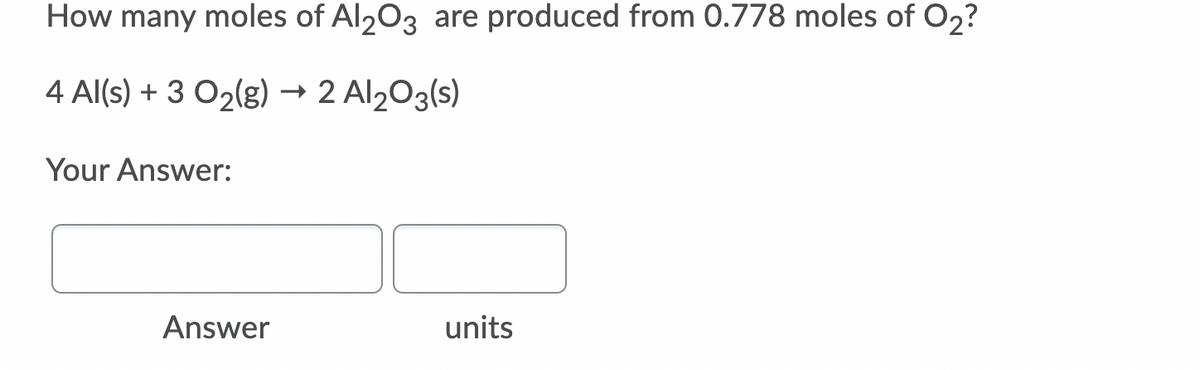
Q: How many moles of Al203 are produced from 0.497 moles of O2? 4 Al(s) + 3 O2(g) → 2 Al203(s) Your…
A:
Q: You react 100.0 g of O2 and 50.0 g of H2 in a container and produce water. 2H2 + O2 → 2H2O How…
A: Limiting reactant :- reactant which is present in less amount than required as per balanced chemical…
Q: How many grams of SO3 are produced when 20.0 g FeS2 react with 16.0 g O2 according to this balanced…
A: The answer to the following question is- Reaction : 4 FeS2 + 15 O2 -----> 2 Fe2O3 + 8 SO3
Q: In the acetylene torch, acetylene gas (C2H2) burns in oxygen to produce carbon dioxide and water. 2…
A: The reaction is given below.
Q: How many moles of water are produced if 8.0 grams of oxygen react completely with excess hydrogen?…
A: Solution stoichiometry is mainly based on the calculation of moles and volumes. These two values are…
Q: How many moles of C6H12O6 are formed when 7.46 moles of O2 are produced in the following reaction ?…
A: The balance chemical equation : 6CO2 + 6H2O ----> C6H12O6 + 6O2 Number of moles of O2 = 7.46…
Q: 2KClO3 →2KCl + 3O2 How many grams of potassium chloride are produced 25 g of potassium chlorate…
A: Balanced Reaction 2KClO3 ---> 2KCl + 3O2 Mass of KClO3 = 25g Molar mass of KClO3 = 122.55g/mol…
Q: For the reaction 2 H2O → 2 H2 + O2, how many moles of O2 are produced with 1.45 moles of H2? Your…
A: Given Reaction 2H2O → 2H2 + O2 Number of mole of H2 = 1.45 mole Number of mole of O2 = ?…
Q: 2H2 + O2 → 2H20 Given 3.0 moles of H2, how many grams of O2 are need for the reaction to take place…
A: Number of moles of a chemical compound is used to measure the amount of the compound in a given…
Q: In a particular reaction 5.00 moles of NH3 were produced. How many moles of H2 and how many moles of…
A: Nitrogen reacts with hydrogen to form ammonia. The equation for the balanced chemical reaction is…
Q: Using the equation below, find how many grams of Ag2S can be produced from 48.63 g of AgI. Round to…
A:
Q: how many moles of O2 is in excess when the reaction is over? Do not include
A: Solution: The given reaction: 4NH3 + 5O2 → 4NO + 6H2O From the stoichiometry we can find theoretical…
Q: How many grams of water are produced if 32.0 g of CH4 are completely consumed, according to the…
A: Answer :- B) 72.0 gm 1 mole of CH4 produces 2 moles of H2O
Q: How many moles of H3PO4 are produced when 73.6 g P4010 reacts completely to form H3PC P4010(s) +…
A: The given data : The mass of P4O10 = 73.6 g To determine : The moles of H3PO4
Q: Solve this
A:
Q: 2Mg + O2 --> 2MgO How many grams of MgO are formed when 10 g of Mg react with an excess of O2?
A: The number of moles of the substance is equal to the ratio of mass to the molar mass of the…
Q: How many liters of carbon dioxide gas are produced if 0.250 moles of oxygen react completely in the…
A: Here ethane reacts completely with the oxygen to form carbon dioxide and water molecules .A…
Q: How many moles of Al203 are produced from 0.836 moles of Al? 4 Al(s) + 3 O2(g) → 2 Al203(s) Your…
A:
Q: Aluminum carbide reacts with water to produce methane and aluminum hydroxide in the reaction shown…
A: Mass of Al4C3=112g Mass of H2O=174g Mass of CH4=? Molar mass of Al4C3 = 143.96gm/mole Molar mass of…
Q: Consider the following reaction: CH4 + 2O2 --> 2H2O + CO2 How many moles of water can be formed…
A: Given Reaction CH4 + 2O2 → 2H2O + CO2 Number of mole of CH4 = 3 mole Number of mole of…
Q: 1HgO(s) + 1 Na2SO3(aq) 1Hg(l) + 1Na2SO4(aq) Use the balanced reaction above to calculate the answer…
A:
Q: Determine the number of molecules of Cr2o3 that form when 1.34x10^3g of oxygen completely reacts…
A: Moles of oxygen=1.34×10316=83.75 mol4Cr(s)+3O2(g) →2CrO3(s)From the chemical reaction,3 moles of…
Q: How many moles of Al203 are produced from 0.349 moles of Al? 4 Al(s) + 3 O2(g) → 2 Al203(s) Your…
A:
Q: For the reaction shown, how many grams of oxygen will be required to reach with 15.5 grams of…
A:
Q: Using the equation below, find how many grams of tin (Sn) can be produced from 13.32 g of H2. Round…
A:
Q: For the reaction 2Na + 2H20 --> 2NAOH + H2 How many moles of H2 are produced from 3.36 moles of H2O…
A: The amount of substance can be expressed in moles. 1 Mole = 6.023 ×1023 particles. These particles…
Q: A sample of 4.00g of methane (CH4) is mixed with 15.0g of chlorine (Cl2). Determine which is the…
A: CH4(g) + 4Cl2(g) --------> CCl4(g) + 4HCl(g) Formula to be used; Moles = Given weight ÷ Molecular…
Q: Potassium superoxide, KO2, reacts with carbon dioxide to form potassium carbonate and oxygen:…
A:
Q: Aqueous sulfuric acid (H2SO4) reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium…
A: Given, H2SO4(aq) + NaOH(s) → Na2SO4(aq) + H2O(l) Balancing the above equation, we get H2SO4(aq) +…
Q: How many molecules of H2O will be produced if 22.6 moles of NH4NO3 are decomposed in the reaction…
A:
Q: sulfur reacts with oxygen to form sulfur trioxide. if 8.4g of sulfur react, how many moles of sulfur…
A: Mass of Sulfur = 8.4 g Molar mass of Sulfur = 32.065 g mol-1
Q: Use this equation for problems c. and d.: 2HCI + Mg(OH)2 - MgCl2 + 2H20. c. How many grams of H20…
A:
Q: How many moles of O2 will be formed from 5.60 moles of KCIO3 for the following reaction? Answer to 2…
A:
Q: Using the following balanced equation - 3Cl2 + 6NaOH --------> 5NaCl + NaClO3 + 3H20 How…
A: An important part of chemistry that includes mass of substance involved in any particular reaction…
Q: Glucose oxidation is a chemical process that provided energy for an organism to carry out all of its…
A:
Q: How many grams of oxygen are formed when 1.522 grams of KO are reacted? 4 KO() 2H20() 4 KOH() O28)…
A: Here we have to calculate the grams of O2 formed, during this reaction: 4 KOs+2H2Ol→4KOHs+O2g Where…

Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- In the acetylene torch, acetylene gas (C2H2) burns in oxygen to produce carbon dioxide and water. 2C2H2 (g) + 5O2(g) à 4CO2(g) + 2H2O (g) Find the moles of CO2produced when 2.5 moles C2H2are burnt? Your Answer: Question 16 options: Answer unitsA container holds 15.0 g of P₄O₆ and 6.00 g of O₂. The following reaction occurs: P₄O₆ + 2O₂ → P₄O₁₀.if 17.5 g of P₄O₁₀ is actually produced, calculate the % yield.6 CO2 + 6 H2O→ 6 O2 + C6H12O6; A) Determine the moles of CO2 used to make 100 moles of C6H12O6. B) Determine the moles of O2 produced when 100 moles of C6H12O6 is produced.
- 2H2 + O2 ⟶⟶ 2H2O How many moles of O2 are needed to produce 10 moles of H2O?Given the combustion of gasoline: 2C8H18 + 25 O2 →16CO2 +18H2O How many moles of O2 is required to react with 4.00 moles of C8H18? Select one: a. 2.00 b. 50.0 c. 25.0 d. 12.5A container holds 15.0 g of P4O6 and 6.00g of O2 The following reaction occurs: P4O6 +2O2=P4O10 if 17.5 g of P4O10 actually produced, calculate the % yield
- Balance the reaction shown below: C2H6+O2→CO2+H2OC2H6+O2→CO2+H2O Once balanced, given 0.826 moles C2H6C2H6, calculate the moles of H2OH2O produced. A. 2.48 B. 1.65 C. 0.275 D. 0.413 E. 0.826Given a balance chemical equation , 2 C2H6 (g)+7 O2 (g) —> 4 CO 2 (g)+6 H2O(g) If reaction mixture contains 5 moles of O2 (g) with 6 moles of C2H6 (g) how many moles of CO2 will be produced? Hint: Identify the limiting reactant first Round and report your answer with a number having one decimal point. Enter that number into the answer box Don't include unit.e) If only 0.105 grams of hydrogen gas actually form, what is the percent yield for this reaction? [ Select ] ["1.9 %", "3.55 %", "28.1 %", "114 %"]
- A chemistry student performed the reaction: SnCl4(l) + 2H2Se(g) -> SnSe2(s) + 4HCl(g) in lab. The student started with 0.34 moles of SnCl4 and 0.46 moles of H2Se. The amount of HCl(g) actually produced by the student was 0.26 moles of HCl. a. what is the limiting reagent? b. what is the % yield for this reaction?Determine the balanced chemical equation for this reaction. C8H18(g)+O2(g)→CO2(g)+H2O(g) Enter the coefficients for each compound in order, separated by commas. For example, 1,2,3,4 would indicate one mole of C8H18, two moles of O2, three moles of CO2, and four moles of H2O.observe the following balanced chemical equation: 6 NH3(g) + 8 O3(g) --> 3 N2O5(g) + 9 H2O(g) if the reaction of 2.0 mol NH3(g) and 2.5 mol of O3(g) is run with 100% efficiency which of the followng is correct? all NH3(g) will be consumed 60.0 g of water will be produced all of the O3(g) will be consumed 1.0 mole of N2O5(g) will be produced



