Q: 1. How many moles of oxygen would be required to react with calcium to produce 8 moles of calcium…
A: Calcium reacts with oxygen to form calcium oxides. We will first write the balanced equation, then…
Q: Calculate how many milliliters of carbon dioxide would be produced theoretically from 2.0 g of…
A:
Q: Given the following balanced chemical equation, how many grams of Hg would be produced from the…
A: First of all, we will calculate the molar masses of each HgO and Hg in the reaction and then based…
Q: How many grams of sodium azide are required to produce 275g of N2?
A:
Q: What is the limiting factor for the preparation of methyl orange?
A: sodium sulphanilate dihydrate is limiting factor for the preparation of methyl orange.
Q: How many moles of N2 are produced by the decomposition of 2.88 mol of sodium azide?
A: The Number of Moles of N2 produced from the decomposition of 2.88 mol of Sodium Azide can be…
Q: How many moles of phosphine are prepared from 5.00 moles of Ca3P2
A: The balanced reaction of formation of phosphine from Ca3P2 is given as
Q: Write the balanced equation, then outline the steps necessary to determine the information requested…
A: Hello. Since your question has multiple sub-parts, we will solve first three sub-parts for you. If…
Q: What mass of NaHCO3 reacts in order to produce 450. g of Na3PO4?
A: Balanced chemical reaction is given by:3NaHCO3 + H3PO4 = Na3PO4 + 3H2O + 3CO2
Q: What mass of NaHCO3 must decompose to produce 448 grams of Na2CO3?
A: The balanced equation for the decomposition of NaHCO3 is: 2NaHCO3→Na2CO3+H2O+CO2
Q: What is the molar ratio between O2 and H2O? 4 NH3 + 5 O2 4 NO + 6 H20 ->
A:
Q: How many grams of NH3 can be obtained from 12.0 g N2 using thẻ TölloWIng synthesis of ammonia? N2 +…
A: According to the reaction, 3H2(g) + N2(g) ------------------:>2NH3(g) 12.0 gm…
Q: How many mole of AlCl3 can be produced from 1.4 moles of Al reacting with HCl?
A: The balanced reaction of aluminium with HCl is as :- 2Al + 6HCl ----> 2AlCl3 + 3H2
Q: What mass of methane is required to produce 1.130 kg of carbon dioxide during combustion? (Assume…
A:
Q: How many moles of oxygen would be produced by the decomposition of 75.0g of copper (II) nitrate?
A: Two moles of copper (II) nitrate undergoes decomposition to form one mole of oxygen gas according to…
Q: Calculate the number of grams of SO2 that must react to produce 350. g of SO3.
A: 2SO2 + O2 ---------> 2SO3 Given weight of SO3, W(SO3) = 350 g Molecular weight of SO3, Mwt(SO3) =…
Q: When 2.50g Iron (II) hydroxide is treated with 2.50g of phophoric acid. 1.99g of iron(II) phosphate…
A:
Q: How many moles of phosphine are prepared from 2.00 moles of H2O?
A: Phosphine can be prepared by the reaction of calcium phosphide and water. The balanced reaction of…
Q: Calculate the number of moles in 16.0g of Fe2O3.
A:
Q: write the balanced equation, then calculate the number of moles and the mass of Mg required to react…
A: Given : Mass of Mg = ? Mass of HCl = 5g
Q: Explain why Nitrogen is produced from the top of the cryogenic distillation column while Oxygen is…
A: Nitrogen is obtained on the top while oxygen is obtained on the bottom if the column.
Q: How many moles of hydrogen gas would be needed to react with excess carbon dioxide to produce 30.6…
A: The reactants given are hydrogen gas i.e H2 (g) and carbon dioxide gas i.e CO2 (g) and the product…
Q: How many moles of H2 gas can be formed from 0.39 grams of Mg? M g ( s ) + 2 H C l ( a q ) ⟶ H 2 ( g…
A:
Q: Calculate the number of I of hydrogen gas required to make 4. N2+ H2
A:
Q: How many moles of phosphine are prepared from 7.00 moles of Ca3P2?
A: Phosphine can be prepared by the reaction of calcium phosphide and water. The balanced reaction of…
Q: What mass of aluminium oxide formed when 10.0g of Al is burned in 20.0g of O2?
A: Given: The amount of Al = 10.00 g The amount of O2 = 20.00 g Al burns in O2 to given an oxide of Al…
Q: What is the concentration of HOCI required to oxidize 30 mg/L of dissolved nitrogen in NH3 to form…
A:
Q: Write the balanced equation, then calculate the number of moles and the mass of Mg required to react…
A:
Q: Using the following balanced equation, determine how many moles of hydrogen gas should be produced…
A: Magnesium is strong alkali metal when it reacts with any acid reaction is vigorously flammable…
Q: When NaHCO3 is heated above 270°C, it decomposes to Na2CO3 (s), H2O (g) and CO2(g) a) Write a…
A: (a)Sodium bicarbonate decomposition reaction is given. Its balanced chemical equation is given…
Q: How many grams of NaOH is produced from 120 grams of Na2O?
A: Since you have posted multiple questions, we are entitled to answer the first only. Please repost…
Q: Write a balanced chemical equation for the decomposition of CaCO3 on heating and explain what the…
A: ✓CaCO3(s)=CaO(s)+CO2(g) ✓ following are two important sources of carbon dioxide when this Reaction…
Q: How many moles of oxygen gas react to yield 0.100 mole water with C5 h-12 + O2 CO2 + H2O
A:
Q: What is the enthalpy change when 49.4 mL of 0.430 M sulfuric acid reacts with 23.3 mL of 0.309 M…
A: A chemical reaction is symbolic representation of the conversion of substances to new substances. In…
Q: how many grams of carbon are needed to react completely with 4.75g of fe2o3 ?
A: Number of moles = mass/molar mass Molar mass of Fe2O3 = 159.688 g/mol Mass of C = moles of C ×…
Q: Sulfur dioxide is a toxic sulfur compound. List one natural source and one industrial source that…
A: Sulfur dioxide (SO2) is toxic sulfur compound.
Q: How many grams of carbon dioxide are produced from the combustion of 1.0kg of propane? Write a…
A: Combustion is the burning in the presence of oxygen.
Q: How many grams of H2 will result from the reaction of 100 g of CaH2 with excess H2O?
A: The Reaction is CaH2+2 H2o- Ca(oH) 2+ 2H2 1 mole of CaH2 will give 2 miles of H2
Q: The number of moles and the mass of Mg required to react with 5.00 g of HCl and produce MgCl2and…
A: Since you have posted sub parts question. We have solved First 3 sub parts for you. If want any…
Q: 1.ln the preparation of aspirin, 6.0 grams of salicylic acid was made to react with acetic anhydride…
A: Interpretation- To calculate the percentage yield of the asprin when 6g of salicyclic acid was made…
Q: PĄO10 is one of the most effective drying agents, having the ability to extract water from other…
A: Balanced chemical equation of a reaction is written according to law of conservation of mass.…
Q: How many moles of water are produced from the combustion of 3.5 mol of hydrogen and 5.2 mol of…
A: To solve this problem first we have to write the balanced chemical equation then according to the…
Q: How many grams of H2 are needed to produce 10.14g of NH3?
A: Given:Mass of Ammonia= 10.14 gWe need to calculate grams of H2 are needed to produce 10.14g of…
Q: If 42.1 g of C4H10 0 react with 73.0 g of ccording to the reaction ation : C4H10 0 + 02 CO2 + H20 a)…
A: Reaction organic compound with oxygen gas is called as "combustion reaction" and products formed in…
Q: What mass of water is produced from the complete combustion of 2.40×10−3 g of methane?
A: The mass of water produced from the complete combustion of 2.40×10−3 g of methane is calculated as,
Q: What type of reaction is… C6H14 (l) + O2 (g) →
A: C6H14 (l) + O2 (g) → CO2(g) + H2O(l) Hexane is reacting with oxygen to produce CO2 and water.
Q: If cobalt(II) sulfate is heated too strongly, the following reaction will occur CoSO4(s) à CoO(s) +…
A: If cobalt (II) sulfate is heated too strongly, the following reaction will occur: CoSO4(s) →…
Q: What mass of nitrogen is contained in the ammonium nitrate prepared by treatment of 6.69g of aqueous…
A: According to the balanced chemical equation,
Q: If 75.0 kg of hydrazine are reacted with 75.0 kg of oxygen, which is the limiting reagent?
A: Hydrazine is represented as N2H4. Limiting reagent is the reactant species which determines the…
Q: Write a balanced equation to show the reaction of gaseous ethane with gaseous oxygen to form carbon…
A: The given options are,
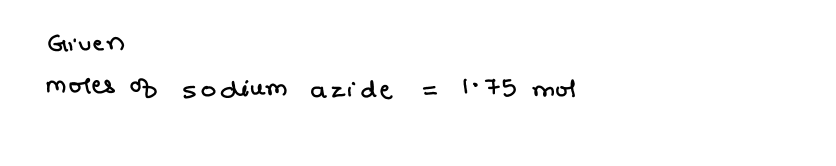
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- How many grams of H2 are needed to produce 10.14g of NH3?What mass of nitrogen gas is produced by the reaction of 8.0g of ammonium perchlorate?Write the balanced equation, then outline the steps necessary to determine the information requested in each of the following:(a) The number of moles and the mass of Mg required to react with 5.00 g of HCl and produce MgCl2 and H2.(b) The number of moles and the mass of oxygen formed by the decomposition of 1.252 g of silver(I) oxide.(c) The number of moles and the mass of magnesium carbonate, MgCO3, required to produce 283 g of carbon dioxide. (MgO is the other product.)(d) The number of moles and the mass of water formed by the combustion of 20.0 kg of acetylene, C2H2, in an excess of oxygen.(e) The number of moles and the mass of barium peroxide, BaO2, needed to produce 2.500 kg of barium oxide, BaO (O2 is the other product.)(f) See image
- How many moles of CaO will be formed from the given amount of V2O5?how many grams of oxygen are required to react with 10.0 g of C2H6Write the balanced equation, then outline the steps necessary to determine the information requested in each ofthe following:(a) The number of moles and the mass of Mg required to react with 5.00 g of HCl and produce MgCl2and H2.(b) The number of moles and the mass of oxygen formed by the decomposition of 1.252 g of silver(I) oxide.(c) The number of moles and the mass of magnesium carbonate, MgCO3, required to produce 283 g of carbondioxide. (MgO is the other product.)(d) The number of moles and the mass of water formed by the combustion of 20.0 kg of acetylene, C2H2, in anexcess of oxygen.(e) The number of moles and the mass of barium peroxide, BaO2, needed to produce 2.500 kg of barium oxide, BaO(O2is the other product.)

