Q: The hybridization of C in the CH4 molecule is sp2 . True False
A:
Q: How many sp3 hybridized carbon atoms are present in the following molecule? s, OA6 OB, 12 OD.8
A: The sp3 hybridised C atoms are given below -
Q: The hybridization of the central atom in Clo, ion is O sp sp2 O sp3 dsp3
A: The number of valence electrons in chlorine is 7. In ClO3-, there is one negative charge which means…
Q: What is the correct hybridization for the oxygen atom in the following molecule: CH3CH2CH2OH ? sp3 O…
A: Hybridization of oxygen in propanol:
Q: Identify all of the carbon atoms that are sp2-hybridized in the following molecule: H :ci: :či: :0:…
A:
Q: Consider the following structure: H. H-C-C -C-CEN H. H. What is the hybridization of the carbon atom…
A: The structure given is
Q: State the hybrid orbitals for C1,C2,O1 and O2
A: hybridization is the concept of mixing of atomic orbitals to form new hybrid orbitals.
Q: Which element does the following hybridization picture belong to? 2s 2 Px 2p, 2p2 sp unhybridized…
A: Here we have to predict the element having the following given hybridization picture.
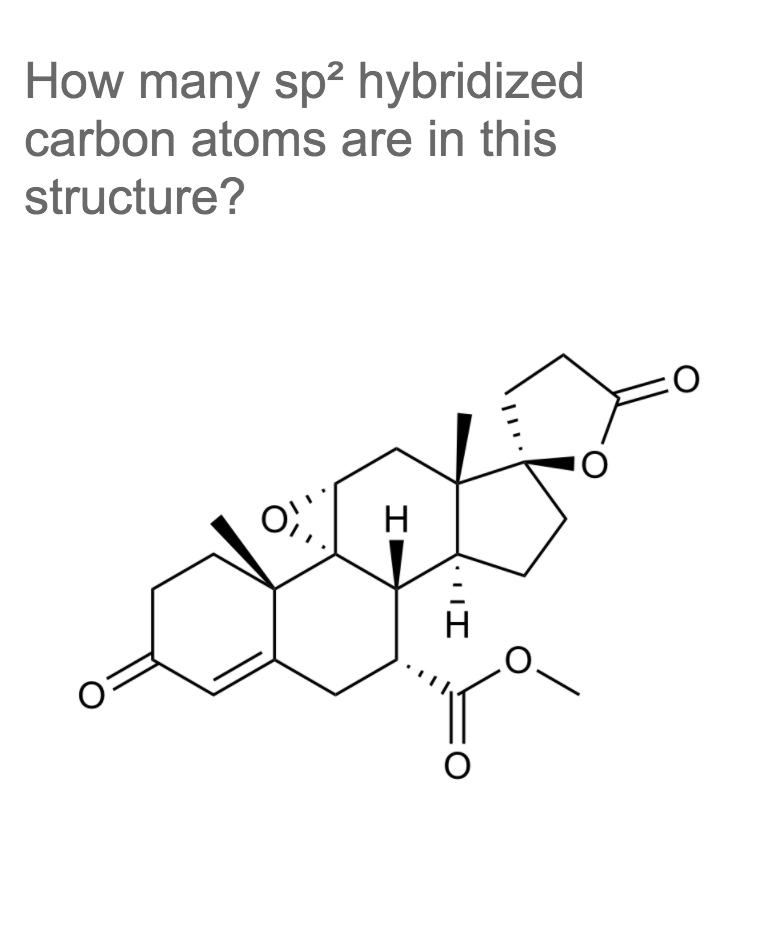
Q: What is the number of sp2 hybridized carbon atoms on the given structure?
A: The sp2 hybridization is the mixing of 1s atomic orbital and 2p atomic orbitals in which there is…
Q: H2N HO IZ
A: Hybridization: The atomic orbitals that have comparable energy and different shape are mixed to give…
Q: The molecule NF3 has which bond hybridization? O sp O sp2 O sp'd O sp'd O sp
A:
Q: If a molecule has (trigonal bipyramidal) geometry, then the hybridization of central atom is * O sp3…
A: Given Geometry = Trigonal bipyramidal Hybridization = to be determined
Q: What are the hybridizations of each carbon atom in the following molecules?
A:
Q: 1. Determine the hybridization of carbon orbitals as sp^3, sp^2, or sp in the C atoms in the…
A: To determine the hybridization of the C atoms in the given compound, we have to calculate the steric…
Q: What is the hybridization of the indicated nitrogen atoms? O A is sp; B is sp² O A is sp²; B is sp³…
A: What is the hybridization of the indicated nitrogen atoms ?
Q: Question attached
A: Hybridisation is defined as the formation of new hybrid orbitals from unhybrid atomic orbitals by…
Q: Provide the answer. Urgent
A:
Q: How many sp³ hybridized carbon atoms are in this structure? ОН Н N. НО НО エ-Z
A: We have to identify the number of sp3 hybridized carbons.
Q: Complete the electron-dot structure of caffeine, showing all lone-pair electrons, and identify the…
A: The compound given is,
Q: Molecular Polar Draw Lewis Structure Hybridization of Formula Center Atom Y/N? N3 co,2- BF4
A: Note - Since you have asked a question with multiple sub-parts, we will do the first three sub-parts…
Q: H H -C-C H C-H H H d. H H е. NH2
A: The hybridization of different carbon atoms in the different molecules is as follows:
Q: Determine the hybridization of the labeled atom in attached species ?
A: Hybridization refers to the intermixing of atomic orbitals which leads to the formation of new…
Q: What is the steric number and hybridization of the central atom? a. CH3NO2 b. CHCl3 c. CH3OH
A: Mixing of atomic orbitals of comparable energy is known as hybridization.
Q: Determine the hybridization of the marked N atom in tofacitinib, a drug used to treat rheumatoic…
A:
Q: Electron delocalization occurs when there is a(n) This is a conjugated system of p orbitals in…
A: pi bonds are defined as the bonds formed by the lateral overlap of two atomic orbitals.
Q: What is the hybridization of N in the following structure? O sp sp² O sp³ (+) EN-H
A:
Q: Do negative and positive charges count toward hybridization?
A: Hybridization is the process of intermixing of atomic orbitals of either same energy or nearly same…
Q: Use valence bond theory to predict the hybridization of the circled atom. A) p CH2 B) sp C) sp D) sp…
A: Hybridization = no of sigma bonds + no of lone pairs = 4 = sp3 Hybridization = 3= sp2.
Q: How many sp2 hybridized carbon atom exist in the following structure? HO. NH2 ZI
A: The sp2 hybridized carbon atoms are explained as those which have unsaturation and which contain a…
Q: How many sp³ hybridized carbon atoms are in this structure? ОН Н HO HO エ-N
A: Hybridization: The atomic orbitals that have comparable energy and different shape are mixed to give…
Q: H Н— С—С С—Н H In how many sigma bonds does the highlighted atom participate? In how many pi bonds…
A: The number of sigma bonds and pi bonds and hybridization for the above mention highlighted carbon as…
Q: Which atomic orbitals from carbon hybridize to form the bonds in CH4? А) 1s, 2рх, 2p_у, 2p_z В) 2s,…
A: In any molecule, only the valence shell orbitals participate in the hybridisation. Since the carbon…
Q: Predict the hybridization about the central atom in the given structure. X Y:
A: Arrangement of valance electron around the central atom determines the molecular geometries of the…
Q: 4. The hybridization of the central atom in ClF3 is A. sp2 B. sp3 C. sp3d D. sp3d
A:
Q: What is the hybridization of the carbon atoms numbered 1 and 2 respectively in the following…
A:
Q: What are the hybridizations of atoms 1 and 2 respectively in the following structure?
A: Hybridisation is the mixing of atomic orbitals of a atom to form a hybrid orbital.
Q: Which types of atomic orbitals of the central atom mix to form hybrid orbitals in (a) SiClH₃; (b)…
A: (a) SiClH3 Hybridization of Si = sp3 Types of atomic orbitals mixed are one s-orbital and three…
Q: What is the steric number and hybridization of the central atom? a) HNO2 b) HF c) F2
A:
Q: See the Attachment & Answer the following question? Ques: What is the hybridization of each N…
A: SOLUTION: Step 1: Nitrogen is quite different from carbon when it comes to the hybridization. It is…
Q: How many sp2 hybridized atoms exist on the following structure?
A:
Q: What is the hybridization state of Si in SiH4 and in H3Si – SiH3?
A:
Q: Which atomic orbitals from carbon hybridize to form the bonds in CH₄?
A: The given molecule is CH4 We have to find the atomic orbitals of carbon that hybridize to form the…
Q: CH H-N .C. H2N Angle: Hybridization: H2C Shape:
A: hybridization is the redistribution of atomic orbital to reach the maximum covalency of centre…
Q: What is the hybridization of the carbon atom indicated by the arrow in this structure? All atoms in…
A: Hybridization of carbon (C) in hydrocarbons: Alkane -C-C- in this case all bonds are single and the…
Q: Which of the following structures exhibits an sp³-hybridized nitrogen atom? N-H H3C H. ČH3
A: The structure which exhibits sp3 - hybridized nitrogen atom = ?
Q: Determine the hybridization of the marked N atom in tofacitinib, a drug used to treat rheumatoid…
A:
Q: Which hybridization scheme allows the formation of at least one pi bond? sp3, sp2, sp3d2
A: Formation of pi bond takes place by side wise overlap when two p orbitals are perpendicular to rest…
30
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Which of the following compounds contains the greatest number of sp2 hybridized carbon atoms?What kind of hybridization do you expect for each carbon atom in the following molecules? (a) Propane, CH3CH2CH3 (b) 2-Methylpropene, (c) But-l-en-3-yne, H2C=CHa€"Ca%ojCH (d) Acetic acid,O-C-N bond angle and orbital hybridization of C atom?
- In sp²-hybridized orbitals, how many p-orbitals remain to form multiple bonds?What are the hybridizations of the Carbons indicated by the arrows?Which of the following can be used to explain why all bond distances and angles in methane, CH4, are the same? (a) resonance; (b) delocalization of elec tron s; (c) bond polarities; (d) electron ega tivity; (e) orbit al hybridization.
- Fill in each blank sentence with numbers 0-10. An sp3-hybridized atom possess _____ s orbital(s), _______ p orbital(s), and _____ hybrid orbital(s) in its valence shell. Such an atom can form up to _____ sigma bonds and _____ pi bonds.Citric acid is responsible for the tartness of citrus fruits, especially lemons and limes. How many sp2 hybridized carbons are present?Based on the picture, How many sp2 hybridized atoms are in the following compound?

