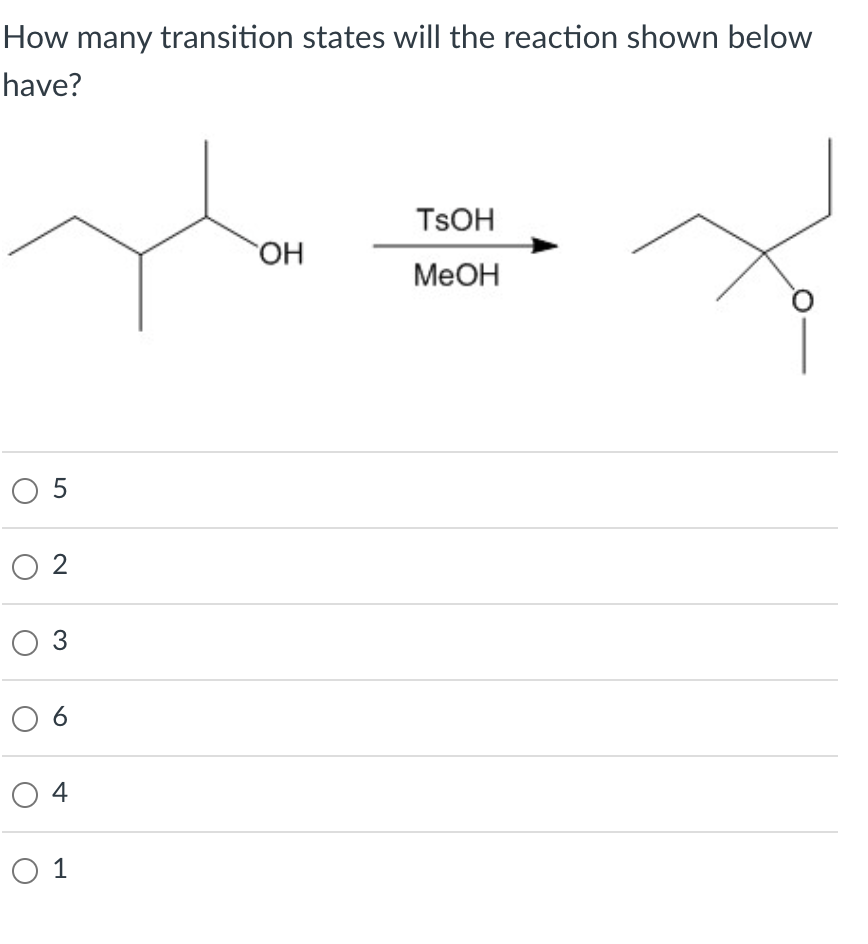
Q: Consider the carbon skeleton shown below. To form a primary alcohol, the functional group can be…
A:
Q: Both physical and chemical changes are associated with changes in energy. Compare the energy changes…
A: The energy difference between the products and reactants is known as the energy change of a…
Q: Identify the IUPAC name of the chemical structures
A: In this question we have to tell the IUPAC name of the compound.
Q: a. Sketch the apparatus that could be used to study the following reaction. Label anode, cathode,…
A: Galvanic cell :- A device used to convert the chemical energy of a spontaneous redox reaction into…
Q: What is the series of reactions for the following synthesis?
A:
Q: Alanine, abbreviated HAla, is an amino acid in protein. If 21.05 mL of 0.145 M NaOH neutralizes…
A:
Q: A gas has a density of 5.51 g/L at a pressure of 1.61 atm and a temperature of 298 K. What is the…
A:
Q: For the following reaction, AH° = - 92.5 k) I(9) + CsHio(g) : CHs(g) + 2HI(g) What effect does each…
A:
Q: (Q70) Using the tabulated values from the text (located in Appendix G, beginning at p. 1211)…
A:
Q: For the reaction 2IB1(g) I2(g) + Brz(g), K = 8.50 x 10-3 at 150 °C. If 0.300 mol of IBr is…
A:
Q: A mixture consisting of 0.2000 mol CO2, 0.1000 mol Hz and 0.1600 mol H20 is placed in a 2.00- L…
A:
Q: You wish to prepare a solution that is 12% sodium chloride by mass How much sodium chloride is…
A: Given :- % (w/w) of NaCl solution = 12 % Mass of solution = 2.0 kg To calculate :- Mass of NaCl…
Q: The heat of vaporization AH, of methanol (CH,OH) is 35.3 kJ/mol. Calculate the change in entropy AS…
A: We have to predict the change in entropy.
Q: LF ÓH
A:
Q: When a polar liquid displays a concave meniscus, what interparticle forces are acting? Compare and…
A: In concave meniscus adhesion forces are acting and in context Cohesion is acting .
Q: CI CO2ME H. CO,Me CO,Me MeO2C. CI 土 d) OH OH
A:
Q: Two coordination sphere isomers for MnCl3(CN)3
A:
Q: Propose a synthesis for each of the compounds below from benzene. It will take 4 or 5 steps, be sure…
A:
Q: 1.In the properties of a system listed below, which one is not state function A U B S С Р D Q
A: State functions in thermodynamics, depend on only initial and finals position and not depend on the…
Q: Predict the effect of adding more H,(g) to a constant-volume equilibrium mixture of N, H, and NH,.…
A: In this question, we will see the effect on equilibrium reaction after addition of hydrogen gas in…
Q: The following are the reasons why instrumental analysis methods are used, except for: a. Narrow…
A:
Q: Preparation of Acetone H3C CH3 a. Hydration b. Ozonolysis c. Oxidation с.
A:
Q: Use the appropriate standard reduction potentials in the appendix of the textbook to determine the…
A:
Q: 1) LDA NH 1) NaOH ww XX 2) 2)H H 2) OE 3) H,O
A: Detail mechanistic pathway is given below
Q: The decomposition reaction: SO,CL(g) S0,(g)+ Cl,(g) is first-order, and has a rate constant of 2.24…
A: For first order kinetics k = 2.303 log(A0/A)/t where k = rate constant A0 = initial concentration of…
Q: If a solid line represents a covalent bond and a dotted line represents intermolecular attraction,…
A: A hydrogen bond is a type of attractive (dipole-dipole) interaction between an electronegative atom…
Q: 4. Calculate (a) AG°, and (b) K, for the following equilibrium reaction at 25.0 °C. PCI,(g) =…
A: a.) ∆G° can be calculated using standard ∆G°f values of species involved in reaction. b.) KP can be…
Q: Consider the following equilibrium system at 661 K. coci, (9) Co(9) + Clz(9) If an equilibrium…
A: K is the ratio of product of concentration of products raised to their stoichiometric coefficient…
Q: Propose a synthesis for each of the compounds below from benzene. It will take 4 or 5 steps, be sure…
A:
Q: For the following hexapeptide shown below, answer the following questions. E-N-D-Y-K-L Draw the…
A: Hexapeptide given is: E-N-D-Y-K-L
Q: If a tree dies and the trunk remains undisturbed for 1.620 x 104 what percentage of the original 1*C…
A:
Q: You have a 36 inch diameter balloon that is currently at a pressure of 0.975 atm, a volume of 300.0…
A:
Q: Which of these can be rinsed down the sink? the left over liquid from a distillation of 1-butanol…
A: The correct answer is Option (4) None of them should go down the sink.
Q: can explain the energy changes as a chemical reaction. What is activation energy?
A:
Q: Outline a synthesis of acetylcholine iodide using dimethylamine, oxirane, iodomethane, and acetyl…
A: Oxiranes are potential intermediate for the synthesis of variety of functional groups. Oxiranes /…
Q: एफरकंच की पैंट कीनफं्कद ठेसळ नी १पवानपलल लपतता के्ूक ( ,1, ता, 5 ) ड क0क एexntsाrbie o हाटाशवाक…
A:
Q: 5) Determine the resulting pl, to 3 decimal places, when 9.18 mg of strontium hydroxide, Sr(OH)2,…
A:
Q: Use the References to access important values if needed for this question. The mass of copper(II)…
A:
Q: A solution of NaOH has a pH of 9.52. What is its pOH, [H3O'], and [OH-] at 25?
A:
Q: ahfference Furctional group Wave number com") HN Oc=C stretch e sp? c-H stretch 3 C-N Stretch ON -H…
A: The organic compound given is
Q: write lown adetaled mechanism fer the fallowing?
A: 1) The compound is said to be acidic in nature only when its conjugate base is stabilized by…
Q: Complete the following nuclear equation and identify X. Chromium-53 + alpha particle ---> neutron +…
A:
Q: A photon of wavenumber 100 cm-1 has a wavelength of (5 points)
A:
Q: How many atoms of Al are needed to react with 34.5 grams of O2 gas?
A:
Q: 1. A solute is prepared by dissolving 15.0 g of table salt in 70.0 g of H2O. Find a) mass percent of…
A:
Q: cinunt Gyan Date Page Plese slove all sub.pasts of this quitim. oution calculate…
A: Since you have posted a question with multiple sub-parts,we will solve first three sub-parts for…
Q: CE264 students prepared 59.5 g of cement and 60mL of kerosene. They put the kerosene inside the le…
A: Here we are required to find the specific gravity of cement Density of water =1g/ml
Q: 1) Determine the pH for each of the following (show your calculations; answer to two decimal…
A:
Q: How does one use ΔH notation to calculate energy changes in a chemical reaction? Is the reaction…
A: ∆H is used to denote the enthalpy change of the reaction. Enthalpy is a measurement of energy in the…
Q: Fumaric acid is quite common in the plant world and is added to many foods due to its acidity and…
A: Given, C4H4O4(aq) + 2NaOH(aq) --> Na2C4H2O4(aq) +2 H2O(l) The molarity of NaOH = 0.09973 M…
Step by step
Solved in 3 steps with 3 images

- The following carbocation is an intermediate in the electrophilic addition reaction of HCl with two different alkenes. Identify both, and tell which C-H bonds in the carbocation are aligned for hyperconjugation with the vacant p orbital on the positively charged carbon.Explain the mechanism of the following reaction using photodissociation: HCOOCH3 + hv -> CO + CH3OHChemistry III. reactionsGive the main product and specify the mechanism that is favored.
- Given the following order of the trans-effect: CO > Br- > Cl- > NH3 suggest syntheses of any TWO of the three possible isomers starting from [PtCl4]2-. Make sure that you assign the correct charge to any intermediates which you draw.Chemistry 1-bromo-1-methylcyclopropane + H2O → 1-methyl cyclopropane + HBr bromocyclopropane + H2O → cyclopropane + HBr Can I get a detailed description of the mechanism of these two reactions? (Specify each specific step and talk about how it happened.).Which of the following reactions result in a positive ∆ Ssys? a. H2O(g) = H2O(l) b. H2(g) + I2(g) = 2 Hl(g) c. C2H2O2(g) = 2CO(g) + H2(g) d. H2O(g) + CO2 (g) = H2CO3(aq)
- Predict the products of the following reaction: HI + Cl2[Rh(PPh3)3Cl] is a precatalyst that can be activated by dissociation of a phosphine ligand to form an active catalyst, B, which is used in the hydrogenation of alkenes. The active catalyst can then undergo oxidative addition in the presence of H2 to form complex C. Propene coordinates to C to form complex D, which then undergoes a 1,2- insertion step to form E. Reductive elimination of propane from E regenerates the active catalyst B. Draw complexes B – E and hence provide a full catalytic cycle, including the activation step, for the hydrogenation of propene12.The products of the reaction KMnO4 + ZnCO3 KMnO4 + ZnCO3 A) KCO3 + ZnMnO4 B) KCO3 + Zn(MnO4)2 C) K2CO3 + Zn( MnO4)2 D) K2CO3 + ZnMnO4
- Which of the following two complexes should undergo the fastest exchange with 13CO: W(CO)6 or Ir(CO)Cl(PPh3)2?What is the heat of combustion of ethane, C2H6, in kilojoules per mole of ethane? Properties of C2H6 at 298K ∆Hf °kJ/mol = –84.0 ∆Gf°kJ/mol –32.0 S°J/(mol·K) = 229.2 Δ?∘rxn=___________kJ/mol ethanePredict the products of each of the following reactions. (a) MnCO3(s) + HI(aq) ⟶(b) CoO(s) + O2(g) ⟶(c) La(s) + O2(g) ⟶(d) V(s) + VCl4(s) ⟶(e) Co(s) + xsF2(g) ⟶(f) CrO3(s) + CsOH(aq) ⟶

