How much energy in Joules is needed by a 10 grams of ice at -30°C to become steam at 120°C? Express your final answer in whole number and do not type the unit in your answer. * ; Csteam -2.01 L₂ = 2260 ½; L = 334; Cice = 2.100 Cwater = 4.186 ; =
How much energy in Joules is needed by a 10 grams of ice at -30°C to become steam at 120°C? Express your final answer in whole number and do not type the unit in your answer. * ; Csteam -2.01 L₂ = 2260 ½; L = 334; Cice = 2.100 Cwater = 4.186 ; =
Physics for Scientists and Engineers, Technology Update (No access codes included)
9th Edition
ISBN:9781305116399
Author:Raymond A. Serway, John W. Jewett
Publisher:Raymond A. Serway, John W. Jewett
Chapter20: The First Law Of Thermodynamics
Section: Chapter Questions
Problem 20.10OQ: A 100-g piece of copper, initially at 95.0C, is dropped into 200 g of water contained in a 280-g...
Related questions
Question
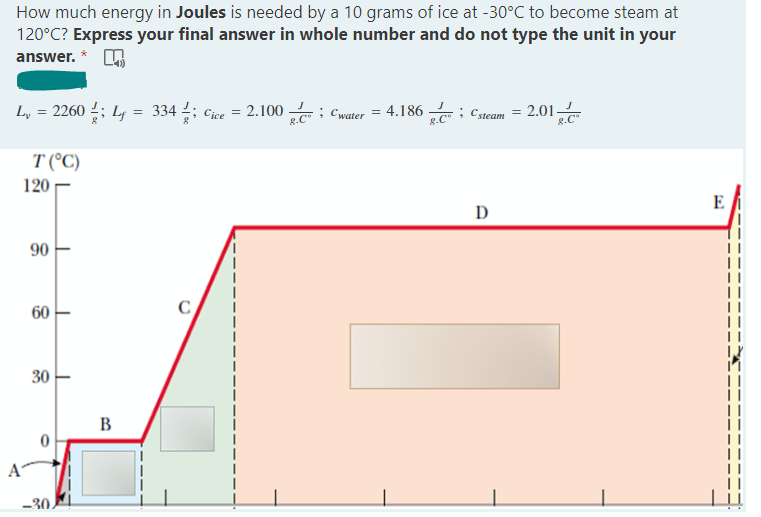
Transcribed Image Text:How much energy in Joules is needed by a 10 grams of ice at -30°C to become steam at
120°C? Express your final answer in whole number and do not type the unit in your
answer. *
L₁ = 2260 ½; Lf = 334; Cice = = 2.100
; Cwater = 4.186
; C steam 2.01-
=
8.C
T (°C)
D
E
120
90
60
30
-30
B
8.C
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you

Physics for Scientists and Engineers, Technology …
Physics
ISBN:
9781305116399
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning


Physics for Scientists and Engineers, Technology …
Physics
ISBN:
9781305116399
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning


College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College