Q: O Problem: Determine the blood sugar result for a patient from a Random Blood Sugar Test
A: Ans. Random Blood Sugar Test: This measures your blood sugar at the time you’re tested. You can take…
Q: Give IUPAC names for the following alkyl halides:
A:
Q: Calculate the molality of a 20.0% by mass ammonium sulfate (NH),SO solution. The density of the…
A:
Q: 24. A sample of 0.6760 g of an unknown compound containing barium ions (Ba?") is dissolved in water…
A: Mass of Unknown = 0.6705 Mass of BaSO4 = 0.4105
Q: Instruction: Calculate the number of each particle in an atom that is missing in the table. SHOW…
A:
Q: 2. Consider the five esters below with formula C 6H 12 O 2: (b) (c) (d) (i) Whic h is/are made from…
A: Since you have posted a question with multiple sub-parts, we will solve the first three subparts for…
Q: normality
A:
Q: Courtn 415 Theoretical yield of chemical reactions Gaseous butane (CH,(CH,),CH,) reacts with gaseous…
A:
Q: Refer to the chemical reaction below: BaCrOs) Ba + CrO 10 Ksp = 2.10 x 10 Calculate the molar…
A: Given : Ksp = 2.10 x 10-10
Q: Consider the following gas-phase reaction: NO2(9) + N20(g)=3 NO(g) Using data from Appendix C of…
A: Given Reaction : NO2(g) + N2O(g) ⇌ 3NO (g)
Q: Solve: Given the following equations and AH° values ΔΗ' (kJ) 164.1 [1] 2 N(e) + O2(e) →2 N,0(s) O…
A: Given equations are : Aim equation is : Calculate change in enthalpy of the aim equation = ΔH = ?…
Q: Benzoic acid Naphthalene Anthracene ОН M = 128.17 g/mol 122.12 g/mol 178.93 g/mol
A: Here we have to find the MM of an unknown solid using the freezing point depression. Here 0.5 gram…
Q: A compound of cycloalkene can be prepared through dehydration of 2-methylcyclohexanol. a) Outline…
A:
Q: What is the mass of 0.75 moles of (NH4)3PO4? O 111.75 g O 121.75 g O 131.75 g 101.75 g
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: bands to its corresponding interpretation. Choices: 1. Band A C=C conjugated 2. Band B N-H amide…
A: IR spectroscopy helps us to find the functional groups of the compounds.
Q: Given the following reactions Fe2O3 (s) + 3CO (s) - 2Fe (s) + 3CO2 (g) AH = -28.0 kJ 3Fe (s) + 4CO2…
A:
Q: What are the chemical properties of bis(benzene) molybdenum and tris(butadiene) molybdenum?
A:
Q: Find the moles of 2-butanol that must be dissolved in 0.145 kg of ethanol in order to produce a 10.0…
A: Given : molal concentration of 2- butanol solution = 10.0 m Mass of ethanol (solvent) = 0.145 kg…
Q: Fe,0.t 3 Ha ca) a) AH" a AG at 230°C
A: The reaction given is, => Fe2O3 (s) + 3 H2 (g) → 2 Fe (s) + 3 H2O (g)
Q: Explain the complications in the dispersion of spheroidal particles in the Mie theory. Please focus…
A: Gaustav Mie provided the mathematical description for spectral dependence of scattering by spehrical…
Q: quantity
A:
Q: solute 0.147 g C6H7NaO6 molar mass 198.11, dissolves in 50.00 mL, absorbance of solution 0.862,…
A: Given that - Mass of Solute, C6H7NaO6 = 0.147 g Molar mass of Solute, C6H7NaO6 = 198.11 g/mol…
Q: Draw the electron pushing reaction mechanism for the hydrolysis of the artificial sweetener…
A: Aspartame on hydrolysis gives aspartic acid, phenyl alanine and methanol under acidic, basic and…
Q: Draw the major product of this reaction. Ignore inorganic byproducts. CI AICI3 +
A: Benzene can give Friedel craft alkylation reaction which is electrophilic aromatic substitution…
Q: Which of the following can be a Bronsted acid? А. OH" В. HCI С. Co32- D. NaCI
A: Bronsted-Lowry acid-base theory: The Bronsted-Lowry acid-base theory states that the acid is a…
Q: Magnesium sulfate, MgSO4, is added to 72.0 mL of 9.50 x 102 M sodium hydroxide, NaOH, until…
A:
Q: 12:02 1 Question 18 of 25 Submit Draw the major product of this reaction. Ignore inorganic…
A:
Q: Chemistry A 20.55 mL sample of .63 M HCL is titrated with .63 M NaOH. Calculate the pH after the…
A: Neutralisation reaction is reaction where 1 mole of acid react with 1 mole of base to form salt and…
Q: Given that you have 10% solution of methyl parahydroxybenzoate and you use 0,1% how many grams of…
A: Given, 10 % solution of methyl parahydroxybenzoate Used : 0.1 % Required, Grams…
Q: ecipitate Another Solid cobalt(II) acetate is slowly added to 75.0 mL of a 0.0317 M potassium…
A: Given, [KOH] = 0.0317 M Volume of KOH in mL = 75.0 mL Now, Solubility product of Co(OH)2 = Ksp…
Q: Which of the following shows the increasing boiling point oh halogens? A. F2 < I2 < Br2 < Cl2 B. I2…
A: Intermoleular forces can be defined as the forces that exist between two or more than two molecules.…
Q: 1. Do all of the following for each molecule or ion. a. Draw the Lewis Dot structure that minimizes…
A:
Q: Predict whether or not these substances will mix. Justify your answer. 1. Paint thinner and oil 2.…
A:
Q: 2. Pt* forms a complex with two ammonia molecules, one SCN and a Cl ion. a) What is the formula and…
A: Answer: To form a complex, ligands donate their lone pairs in the vacant orbitals of central metal…
Q: Example 3. Calculate the magnetic field streagth required to observe the NMR frequency for fluorine…
A: NMR spectroscopy is a technique used to study the properties of organic compounds. It is a…
Q: A galvanic cell is powered by the following redox reaction: Br,(1) + H,(g) + 2 OH (aq) 2 Br (aq) + 2…
A:
Q: I. Using Markovnikov's rule, predict the predominant product in each of the following addition…
A:
Q: A gas mixture with a total pressure of 2900 torrtorr is used by a scuba diver. If the mixture…
A: Numbers of moles is calculated by dividing the given mass with molecular mass of compound.
Q: Calculate the reaction quotient and determine the direction in which each system will proceed to…
A: Given- 2SO3(g)<->2SO2(g)+O2(g) Kp=6.5atm P(SO2)=1.00atm P(O2)=1.130atm P(SO3)=0.25atm
Q: a 22.1 ml sample of 0.506M formic acid is titrated with 0.506M NaOH. Calculate the pH after the…
A:
Q: 1. A 15.00 g of NaCI is dissolved in 5.00 L of water. Find the molarity of the solution? Show your…
A:
Q: The letter labels closest to the points marked with a dot refer specifically to those points. The…
A:
Q: using ELECTROPHILIC SUBSTITUTION solve this problem
A:
Q: B3. Write a detailed mechanism for the following Knorr Synthesis 1) (BUO-NO (0.5 equiv) , HCI A…
A: Preparation of pyrrole compound by using beta amino keto-ester and beta-keto ester is called Knorr…
Q: The rate constant k1 = 4.60 x 10^-4 s^-1 at 350 Celsius. If Ea = 104 kJ/mol. Calculate the T2 in…
A:
Q: Which of the following statements is incorrect about this reaction: CH3-CH=CH2 + HBr -->…
A:
Q: the References to access important values if needed for this question. Draw a structural formula for…
A: The reaction given is,
Q: 3. How many molecules of ethylene diamine will react with Ni(H,O),*? Explain your answer.
A: Answer: Ethylene diamine is a bidentate ligand and water is a monodentate ligand.
Q: The vapour pressure of a pure substance at various temperatures is presented below T (°C) Pvap…
A: The vapour pressure v/s temperature data given is, T (°C) Pvap (mmHg) 0.0 16.0 15.0 31.1…
Q: Write the IUPAC names for these alcohols. a. но но
A: The naming of an organic compound can be done with help of rules of international union of pure and…
Step by step
Solved in 2 steps with 1 images

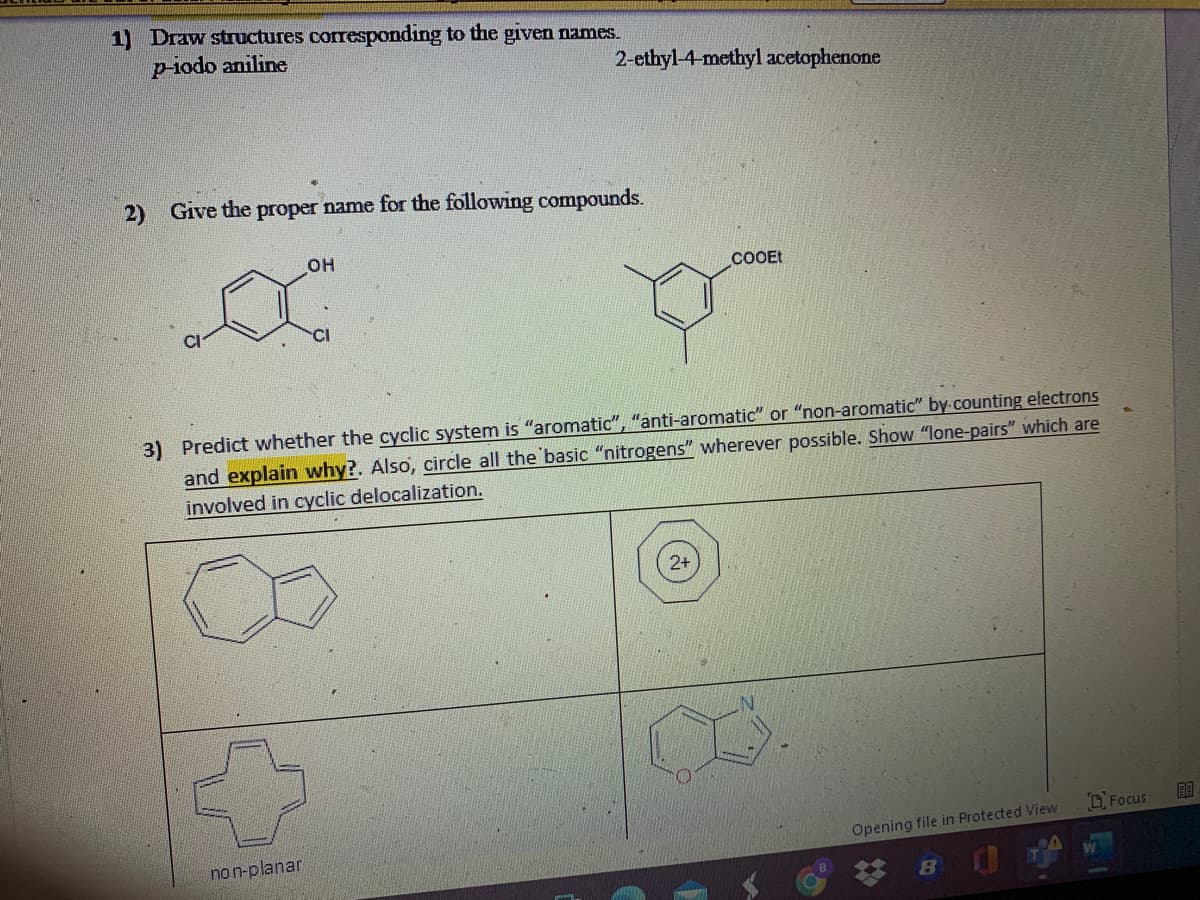
- Predict whether the cyclic system is aromatic , anti-aromatic, or non-aromatic by counting electrons and explain why ? Also circle all the basic nitrogen’s wherever possible . Show all lone pairs which are involved in cyclic delocalization?Aside from the multiple ring systems shown in the table, Naphthalene, Anthracene,Phenanthrene Chrysene,Pyrene, and Benzopyrene. Search for 1 multiple ring aromatic compound and list down its properties, sources, and uses or applications.Please provide bond energy dissociation accurately for these 3 bonds for the below structure (with reference must be from a book or an article which I can find please): between carbon - carbon within the aromatic ring; between carbon - carbon between two aromatic rings; and between carbon - hydrogen within the aromatic ring. Thanks so much for your help!
- FUNCTIONAL GROUPS The research team on Natural Products Chemistry from De La Salle University-Dasmariñas under Mr. Gab has discovered a novel compound from an endemic Philippine nudibranch, Chromodoris cavitensis, whichinhabits the coral reef system near Caylabne Bay in Ternate, Cavite. Exhaustive analysis has revealed acomplex structure as shown below which they aptly named as “chromocavitenin”. Initial tests revealed that thiscompound has potent anti-carcinogenic properties. Encircle and identify the 13 distinct functional groups that arepresent in chromocavitenin.The global consumption of synthetic dyes is increasing due to increase in demand invarious processing/manufacturing industries. Analyze why among the large number of organic molecules, only relatively few moleculesare considered as dyes. Select any one dye which is used in industry and give justificationof type of the dye, its application in the industry and related reactions.Please write down the resulting products. How can I know which of the two substituents already present is more strongly conducting in the case of double-substituted benzene? Is generally ortho preferred to para? In the example of OH and NH2 both otho,para conducting. So are there three possible products?
- Naphthalene is a white volatile, solid polycyclic hydrocarbon used in moth balls and repellents. Looking at the structure of naphthalene given below, determine if it is aromatic or not. Please explain the reasoningTheoretical mass of P-Coumaric acid is 164.16, in LCMS analysis it observed 146.9 why this is so? & same issue with other phenolics, Please Answer.?The name of the parent six-membered sulfur-containing heterocycle is thiane. It is numbered beginning at sulfur. Multiple incorporation of sulfur in the ring is indicated by the prefixes di-, tri-, and so on. (a) How many methyl-substituted thianes are there? Which ones are chiral? (b) Write structural formulas for 1,4-dithiane and 1,3,5-trithiane. (c) Which dithiane isomer (1,2-, 1,3-, or 1,4-) is a disulfide?(d) Draw the two most stable conformations of the sulfoxide derived from thiane.
- Aspirin, or 2-acetoxybenzoic acid, (C9H8O4) is often synthesised from salicylic acid.(i) Sketch and discuss any changes in the number of possible structural conformations ofaspirin relative to those of salicylic acid. (ii) Re-draw the structure predicted to be the lowest energy conformation of aspirin,indicating any expected stabilising and destabilising interactions. Justify your choice.Thoroughly describe what is meant when a compound is considered aromatic. In your answer, provide examples and be sure to discuss how Huckel's Rule applies, and what it means to be anti-aromatic versus non-aromatic. When providing specific examples to demonstrate the requirements that you list, describe the delocalized pi electrons and sp2 hybridization. Lastly, why is benzene so special compared to alkenes like cyclohexene....both are 6-carbon atom cycles so are they chemically similar or not really similar at all?Please advise if these models resemble the cis-2-bromocyclohexanol & trans-2-bromocyclohexanol. Can you please advise on the priority numbers of each substituent. I am confused from the reading. Is the proproty a single element, the group of elements, the left and right carbon atoms? Please advise and give commentary on Br. & Methyl group oriantation related to the doubl carbon bond plain. And if you can provide a visual that would be even better for the futre, the more detail the better my study notes will be. Thank you. Justin

