I need to write about the mechanism and how it happens in this lab please help me explain what happens and how in order for Reduction of 4-Nitrobenzaldehyde to 4-Nitrobenzyl Alcohol and Thin Layer Chromatography
I need to write about the mechanism and how it happens in this lab please help me explain what happens and how in order for Reduction of 4-Nitrobenzaldehyde to 4-Nitrobenzyl Alcohol and Thin Layer Chromatography
Chapter5: Chromatography
Section: Chapter Questions
Problem 1Q
Related questions
Question
I need to write about the mechanism and how it happens in this lab please help me explain what happens and how in order for Reduction of 4-Nitrobenzaldehyde to 4-Nitrobenzyl Alcohol and Thin Layer Chromatography
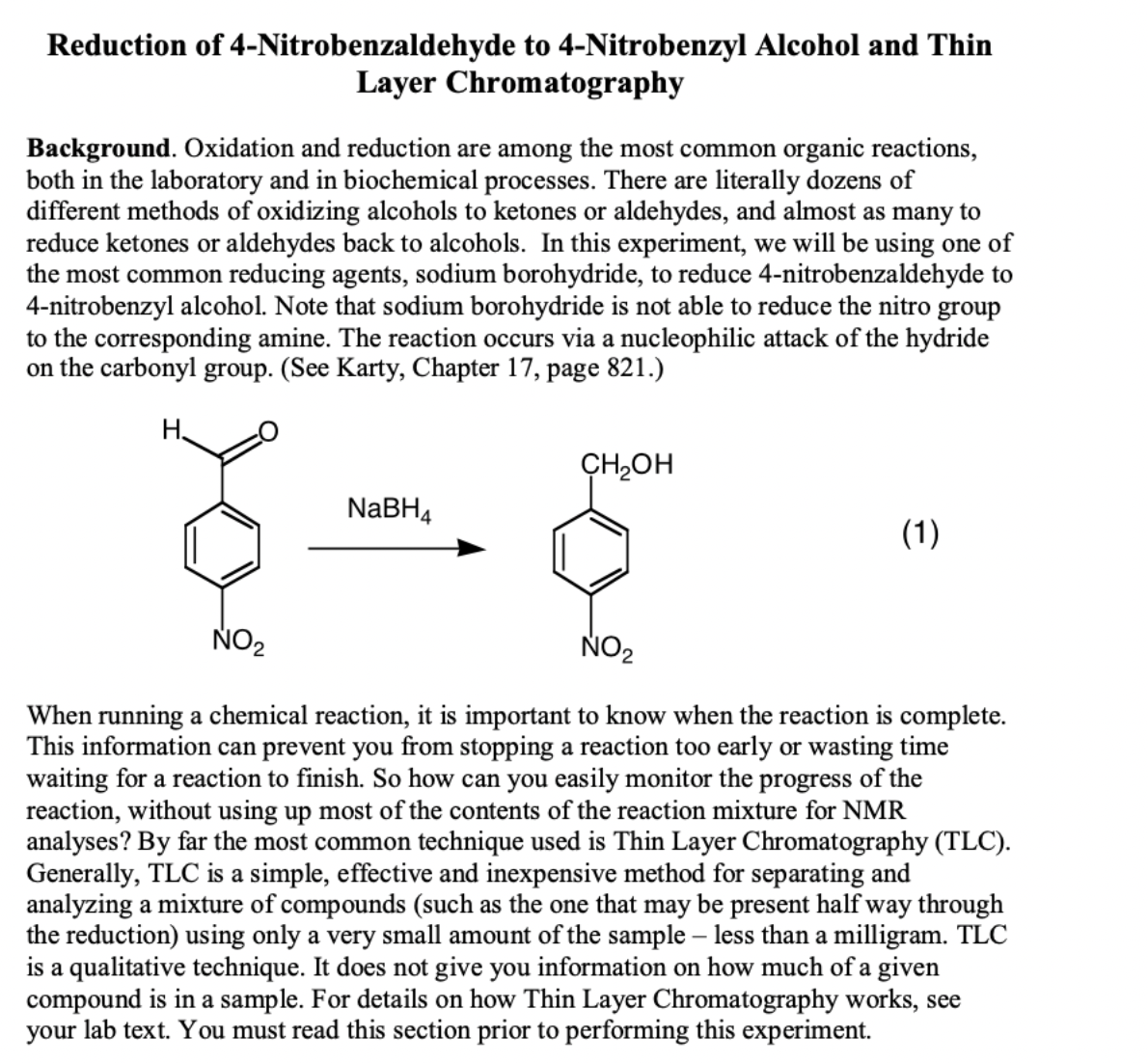
Transcribed Image Text:Reduction of 4-Nitrobenzaldehyde to 4-Nitrobenzyl Alcohol and Thin
Layer Chromatography
Background. Oxidation and reduction are among the most common organic reactions,
both in the laboratory and in biochemical processes. There are literally dozens of
different methods of oxidizing alcohols to ketones or aldehydes, and almost as many to
reduce ketones or aldehydes back to alcohols. In this experiment, we will be using one of
the most common reducing agents, sodium borohydride, to reduce 4-nitrobenzaldehyde to
4-nitrobenzyl alcohol. Note that sodium borohydride is not able to reduce the nitro group
to the corresponding amine. The reaction occurs via a nucleophilic attack of the hydride
on the carbonyl group. (See Karty, Chapter 17, page 821.)
H.
CH2OH
NaBH4
(1)
NO2
When running a chemical reaction, it is important to know when the reaction is complete.
This information can prevent you from stopping a reaction too early or wasting time
waiting for a reaction to finish. So how can you easily monitor the progress of the
reaction, without using up most of the contents of the reaction mixture for NMR
analyses? By far the most common technique used is Thin Layer Chromatography (TLC).
Generally, TLC is a simple, effective and inexpensive method for separating and
analyzing a mixture of compounds (such as the one that may be present half way through
the reduction) using only a very small amount of the sample – less than a milligram. TLC
is a qualitative technique. It does not give you information on how much of a given
compound is in a sample. For details on how Thin Layer Chromatography works, see
your lab text. You must read this section prior to performing this experiment.

Transcribed Image Text:After the reaction has been completed, add about 20 mL of methylene chloride
(dichloromethane/ DCM), and pour the solution into a separatory funnel. Add 20 mL of
water to the separatory funnel. Shake and let the aqueous and organic layers separate.
Place the organic layer (which layer is that?) with your product in an Erlenmeyer flask.
Repeat with another portion of methylene chloride. Combine the organic layers and add a
drying agent. Gravity filter the solution into 100 mL pre-weighed round-bottom flask and
wash the flask and drying agent with an additional 5mL of methylene chloride, also
filtered into your round bottom flask. Evaporate the solvent on a steam bath in the hood
(Erlenmeyer), or use the rotary evaporator (round bottom flask). Weigh the flask again
and calculate the crude yield.
(Be sure all solvent has been removed!)
Purification by recrystallization. Take a melting point of your crude product. Read the
section on recrystallization in your lab text before you come to class. Recrystallization is
a technique for purification of a solid compound. In general, the compound (and any
impurities) is dissolved in a minimal volume of hot solvent (or solvent mixture). This hot
solution is left to cool slowly to room temperature, and then placed in an ice bath. The
solubility of compounds is less in colder solutions, so crystals of compound should form
slowly from the saturated solution. (Since the impurity should be present in small
quantities, the impurity will stay in solution while the compound of interest is
crystallizing). If crystal formation is slow enough, the crystal should be pure compound.
The purified compound is physically separated from solution by filtration. However, if
this crystal formation is fast, impurities may be trapped in the rapidly forming lattice of
the solid crystal. Therefore, it is important to be patient while performing a
recrystallization.
It may help you to see pictures of a movie of the
process.
Recrystallize the product from methylene chloride, and determine the weight percentage
recovery for this purification step once this purified solid has completely dried. If your
TLC of CP1 or CP2 contained multiple spots, take a TLC of the purified compound by
dissolving a few crystals in methylene chloride and eluting with the same solvent as
above (30% ethyl acetate; 70% petroleum ether). Mark this purified product PP.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT