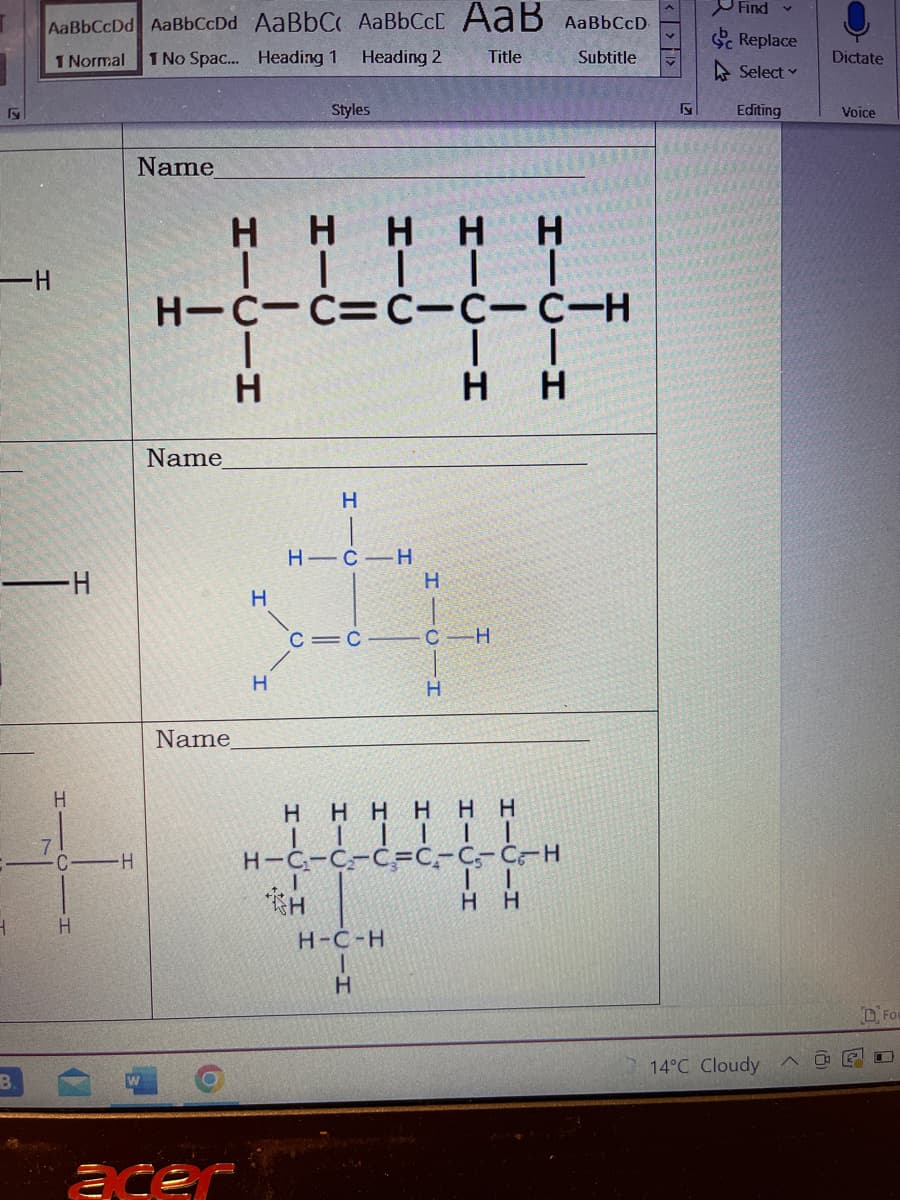
I нннн н | | | | | н-c-c=С-С-С-Н I | | н н Name Т. Name H-C-H c = c — с н / нннннн | | | | | | H-C-C-C=C-C-C-H | | TH н нн н-с-н н I H1C H 0-т I-о-І
I нннн н | | | | | н-c-c=С-С-С-Н I | | н н Name Т. Name H-C-H c = c — с н / нннннн | | | | | | H-C-C-C=C-C-C-H | | TH н нн н-с-н н I H1C H 0-т I-о-І
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.17QAP
Related questions
Question
100%
Transcribed Image Text:AaBbCcDd AaBbCcDd AaBbC AaBbCct AaB AaBbCcD
1 Normal 1 No Spac... Heading 1 Heading 2
Title
Subtitle
1
Styles
Name
HHHHH
|||||
H-C-C=C-C-C-H
|
H
||
HH
Name
H
H-C-H
C=C
F
-H
—H
H
C
1
B.
H
H
Name
H
H
H
H1C
C
H
Η Η Η Η Η Η
| | | | | |
H-C₁-C₂-C₂-C-C-C-H
II
HH
H
H-C-H
H
-H
y
Find ✓
Replace
Select
Editing
14°C Cloudy
A
Dictate
Voice
F
Foo
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

