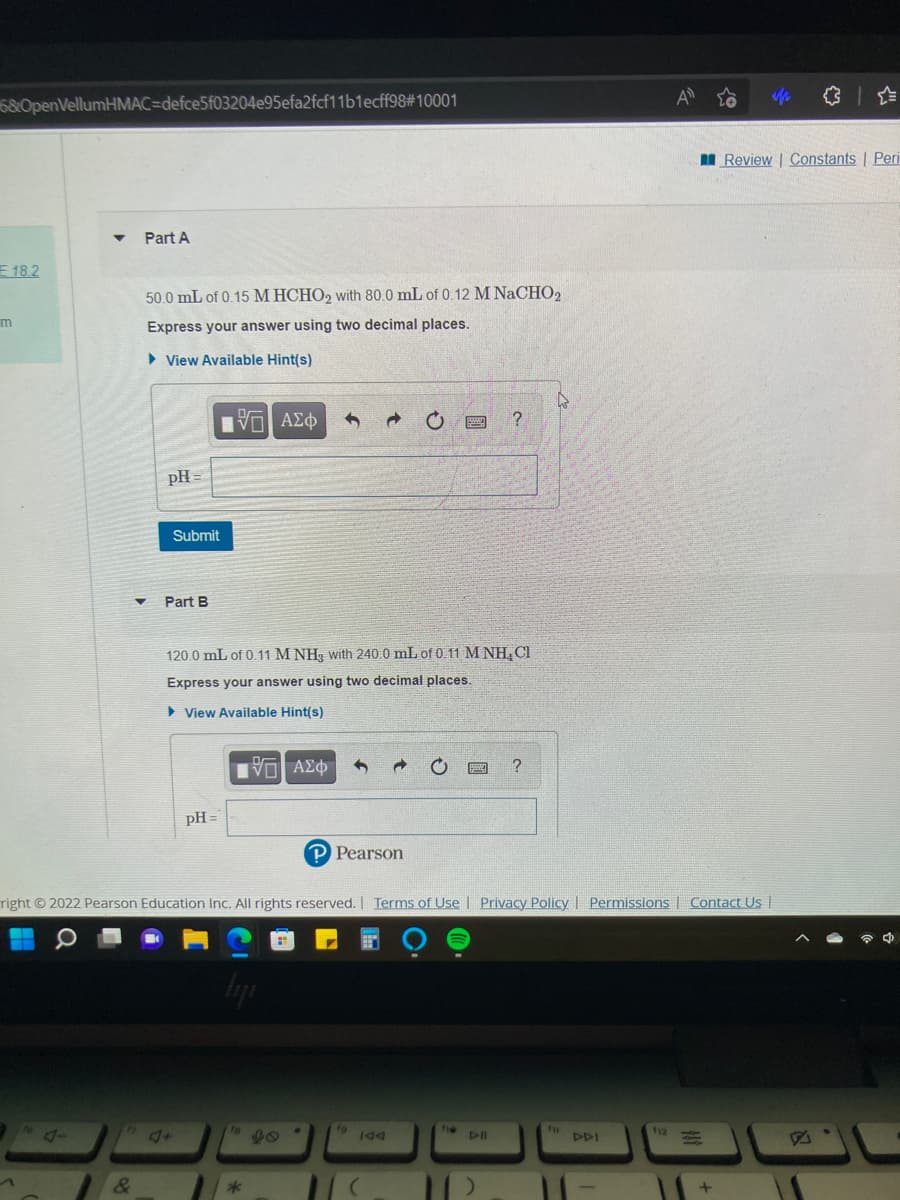
I Review | Constants | Part A 50.0 mL of 0.15 M HCHO2 with 80.0 mL of 0.12 M NaCHO2 Express your answer using two decimal places. > View Available Hint(s) Hνα ΑΣφ ? pH = Submit Part B 120.0 mL of 0.11 M NH3 with 240.0 mL of 0.11 M NH,CI Express your answer using two decimal places. > View Available Hint(s) ? pH = P Pearson
I Review | Constants | Part A 50.0 mL of 0.15 M HCHO2 with 80.0 mL of 0.12 M NaCHO2 Express your answer using two decimal places. > View Available Hint(s) Hνα ΑΣφ ? pH = Submit Part B 120.0 mL of 0.11 M NH3 with 240.0 mL of 0.11 M NH,CI Express your answer using two decimal places. > View Available Hint(s) ? pH = P Pearson
Chapter31: Introduction To Analytical Separations
Section: Chapter Questions
Problem 31.19QAP
Related questions
Question
100%
Transcribed Image Text:68OpenVellumHMAC=defce5f03204e95efa2fcf11b1ecff98#10001
I Review | Constants | Peri
Part A
E 18.2
50.0 mL of 0.15 M HCHO2 with 80.0 mL of 0.12 M NaCHO2
m
Express your answer using two decimal places.
• View Available Hint(s)
να ΑΣφ
pH =
Submit
Part B
120.0 mL of 0.11 M NH, with 240.0 mL of 0.11 M NH, CI
Express your answer using two decimal places.
> View Available Hint(s)
Πνα ΑΣφ
pH =
P Pearson
right 2022 Pearson Education Inc. All rights reserved. Terms of Use | Privacy Policy I Permissions | Contact Us |
f9
f12
4+
DDI
&
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning