I Review Rank the solutions in order of decreasing H30*]. Rank solutions from largest to smallest hydronium ion concentration. To rank items as equivalent, overlap them. MISSED THIS? Watch KCV 16.5; Read Section 16.5. You can • View Available Hint(s) click on the Review link to access the section in your e Text. Name Formula Ka Reset Help Acetic HC2H3O2 1.8 x 10-5 Benzoic HC,H;O2 | 6.5 x 10-5 Chloric HC103 >1 Chlorous HC1O2 1.1 x 10-2 0.10 M HCIO 0.10 М HF 0.1о м НCI 0.10 M HCN Hydrochloric HCI >1 Hydrocyanic HCN 4.9 × 10-10 Hydrobromic HBr >1 Hydrofluoric HF | 6.8 × 10-4 Largest concentration Smallest concentration Hydroiodic HI > 1 Hypochlorous HC1O 2.9 x 10-8 Nitric HNO3 >1 Nitrous HNO2 4.6 x 10-4 Perchloric HC1O4 > 1
I Review Rank the solutions in order of decreasing H30*]. Rank solutions from largest to smallest hydronium ion concentration. To rank items as equivalent, overlap them. MISSED THIS? Watch KCV 16.5; Read Section 16.5. You can • View Available Hint(s) click on the Review link to access the section in your e Text. Name Formula Ka Reset Help Acetic HC2H3O2 1.8 x 10-5 Benzoic HC,H;O2 | 6.5 x 10-5 Chloric HC103 >1 Chlorous HC1O2 1.1 x 10-2 0.10 M HCIO 0.10 М HF 0.1о м НCI 0.10 M HCN Hydrochloric HCI >1 Hydrocyanic HCN 4.9 × 10-10 Hydrobromic HBr >1 Hydrofluoric HF | 6.8 × 10-4 Largest concentration Smallest concentration Hydroiodic HI > 1 Hypochlorous HC1O 2.9 x 10-8 Nitric HNO3 >1 Nitrous HNO2 4.6 x 10-4 Perchloric HC1O4 > 1
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter14: Acid-base Equilibria
Section: Chapter Questions
Problem 113E: Draw a curve for a series of solutions of HF. Plot [H3O+]total on the vertical axis and the total...
Related questions
Question
7.
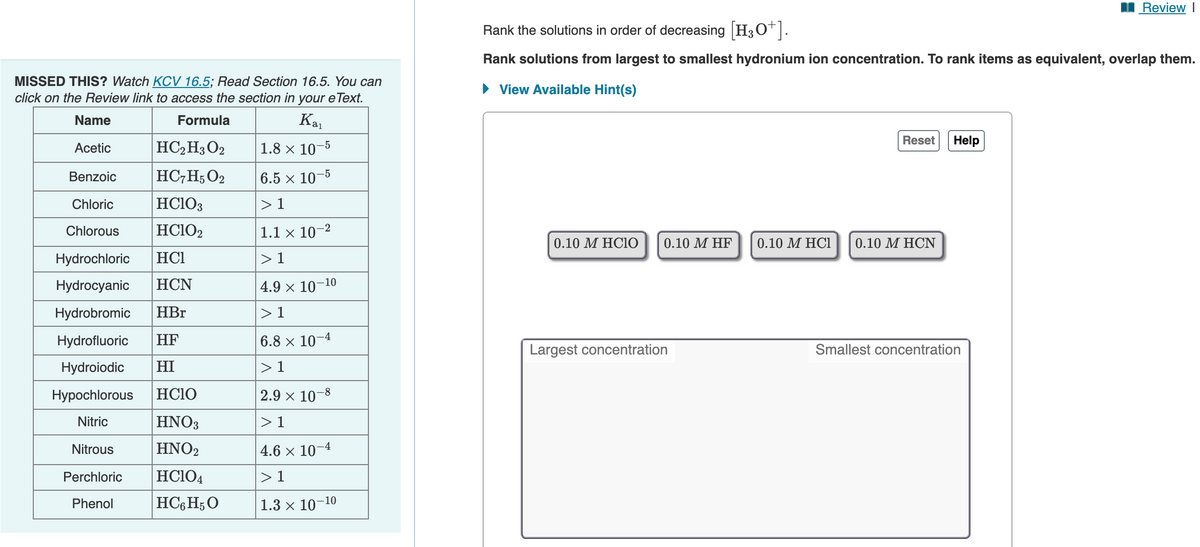
Transcribed Image Text:Review I
Rank the solutions in order of decreasing H3O*.
Rank solutions from largest to smallest hydronium ion concentration. To rank items as equivalent, overlap them.
MISSED THIS? Watch KCV 16.5; Read Section 16.5. You can
click on the Review link to access the section in your e Text.
• View Available Hint(s)
Name
Kai
Formula
Reset
Help
Acetic
HC2H3 O2
1.8 х 10-5
HC,H; O2
Benzoic
6.5 x 10¬
-5
Chloric
HCIO3
> 1
Chlorous
HCIO2
1.1 x 10–2
0.10 Μ HCIO
0.10 M HF
0.10 M HC1
0.10 M HCN
Hydrochloric
HC1
> 1
Hydrocyanic
HCN
4.9 x 10-1
-10
Hydrobromic
HBr
> 1
Hydrofluoric
HF
6.8 x 10–4
Largest concentration
Smallest concentration
Hydroiodic
HI
> 1
Нуроchlorous
HC1O
2.9 x 10-8
Nitric
HNO3
> 1
Nitrous
ΗΝΟ
4.6 х 10
-4
Perchloric
HC104
> 1
HC6H;O
Phenol
1.3 x 10-
-10
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax
