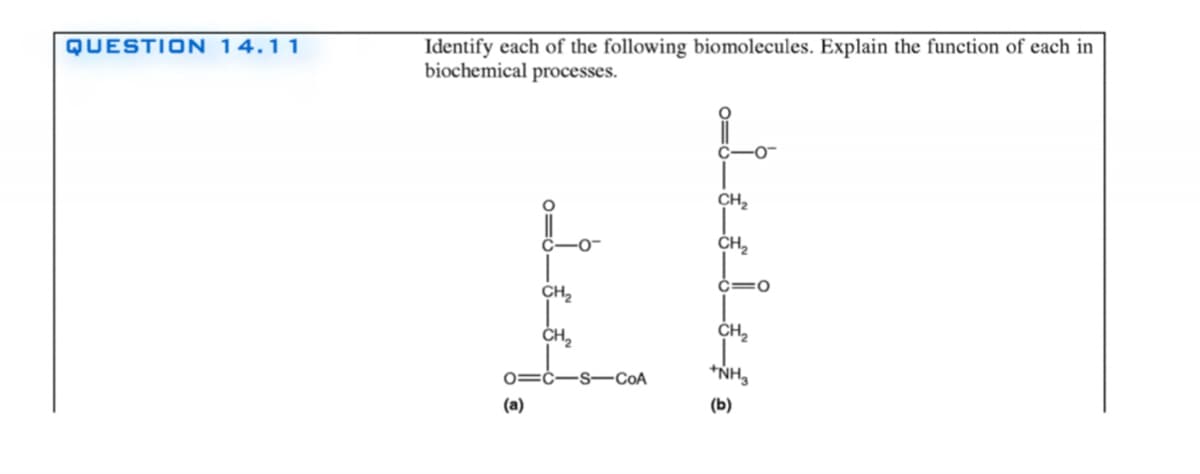
Identify each of the following biomolecules. Explain the function of each in biochemical processes. -0- CH, ČH, CH2 CH, CH, *NH, 0=ċ-s-CoA (a) (b)
Q: Give the full biochemical name for a D-aldopentose with the following pattern of chiral centers:…
A: Monosaccharides are the simplest carbohydrates and they are polyhydroxy aldehydes or ketones with a…
Q: What is unique about TPP that makes it useful in decarboxylation reactions?
A: TPP acts as a coenzyme in several decarboxylation reactions, such as pyruvate conversion to…
Q: Draw Haworth projection formulas for the b-anomer of monosaccharides with each of the following…
A: a. A contains a six carbon chain thus the Haworth projection may either be a six membered ring or a…
Q: Identify the IUPAC name of the 2nd or middle fatty acid.(lower case format) * CH,-0-C(CH,),„CH;…
A: Fatty acids are the building blocks of lipids which consists of hydrocarbons in their structure.…
Q: write the structure of the following disaccharides
A: Biomolecules are organic molecules made up of mainly carbon and hydrogen but there are other…
Q: What is the ratio of [isocitrate] to [citrate] under cellular conditions at 37°C?
A: The physiological free energy changes of a reaction determine its fate in the metabolic reaction.…
Q: Draw the structure and give the name of the epimers of the following monosaccharides ALDOTRIOSE I…
A:
Q: Give at least 5 examples of biological compounds having a ketone functional group and identify the…
A: Ketones have a carbonyl group (C=O) as a functional group. A ketone has two alkyl or aryl groups…
Q: Draw the following sugars using Haworth projections:a. b-d-galactopyranose b. a-d-tagatopyranose c.…
A: A Haworth projection represents the cyclic shape of the carbohydrate. For sugars with a ring…
Q: What are the four necessary conditions that define the biochemical standard state?
A: A typical situation is an irrational situation set for something in order to create a standard…
Q: If mevalonate labeled with 14C in the carboxyl carbon were administered to rats, which carbons of…
A: The mevalonate pathway, or the isoprenoid pathway, also termed the 3-hydroxy-3-methylglutaryl-CoA…
Q: Given the following reaction, identify the class and subclass of the enzyme involved. CH2OH C=O…
A: Enzymes catalyse the reaction either by making or breaking the bond. They might form double bonds or…
Q: Draw the isomers for each aldotetrose and ketopentose in the figure below and designate each isomer…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: In solution, glucose exists predominantly in the cyclic hemiacetal form, which does not contain an…
A: Glucose is the carbohydrate source. The glucose can be hemiacetal or hemiketal. They are formed when…
Q: isomaltose is a disaccharide which can be obtained by enzymatic hydrolysis of amylopectin. Deduce…
A: Disaccharide sugars are the ones that are formed when two monosaccharides are joined by glycosidic…
Q: What is the natural source of jasmonic acid compound and its synthesis pathway?
A: Introduction Jasmonic acid is a compound of plant origin. Some natural sources are the leaf of Fagus…
Q: What are the immediate biochemical sources for the two nitrogen atoms in urea?
A: Introduction: Urea is formed as a by-product during the ornithine cycle which takes place in the…
Q: Draw a Fischer projection formula for the enantiomer of each of the following monosaccharides. а.…
A: The type of isomerism where isomers have the same molecular formula but different orientations in…
Q: If thymine nucleotides are degraded by the same enzymes as those catabolizing uridine nucleotides,…
A: The cleavage of the ring structures metabolizes thymine and uracil nucleotides. The usual end…
Q: two chemical structures of aldehyde or ketone
A:
Q: Following are Fischer projections for a group of five-carbon sugars, all of which are aldopentoses.…
A: Chiral molecules can exist in the configurations that are non-superimposable mirror images of each…
Q: Draw the keto or enol tautomer for each of the following compounds when they were treated with…
A: 1. Propanol on being treated with traces of acids or bases, its structure is converted to Propanone.…
Q: Name, draw and describe the organic product of the reaction between 2-methylbut-1-ene and H2O in the…
A:
Q: COOH H- H- -OH HO -H H- -OH ČOOH е.
A: ANSWER;- e.) Galactaric Acid.
Q: Draw the structure of the polyketide you would expect to obtain from one unit of acetyl coenzyme A…
A: Polyketides: These belong to a very large class of molecules that includes molecules that are…
Q: Draw the enediolate intermediate of the ribulose-5-phosphate isomerasereaction (Ru5P → R5P).
A: An enediol is an alkene enol with a hydroxyl group attached to both carbon atoms of the carbon…
Q: Draw a Fischer projection formula for the enantiomer of cach of the following monosaccharides a.…
A: Monosaccharides are carbohydrates with a single monomeric unit. There are different types of…
Q: what is the conjugate acid for the following reaction? HC2HO4 + H2O H3O+ + C2HO4- A)C2HO4- B)…
A: Conjugate acid is a compound, which donates protons or hydrogen in the chemical reaction.
Q: The chemical structure of Coenzyme A contains the following EXCEPT— a β-mercaptoethylamine…
A: The correct answer is LIPOIC ACID RESIDUE
Q: Considering the monosaccharide transformation given below, what is the name of the depicted…
A: Introduction: Carbohydrates are large biomolecules that are mainly synthesized during the process of…
Q: Give at least 5 examples of biological compounds having an ester functional group and identify the…
A: Esters are the class of organic compounds that are formed due to the reaction between an alcohol and…
Q: Draw the structure of two different aldohexoses that yield the followingaldaric acid when oxidized…
A: Aldohexose are 6 carbon containing compound which had an aldehyde group attached to it. Aldaric acid…
Q: Is phenylalanine hydrophilic or hydrophobic? Explain why.
A: Hydrophilic : It is a molecule or other molecular entity that is attracted to water molecules and…
Q: 1. aarachidnonic acid is the amjor starting material for eicosanoids 2.both gylycoholic and…
A: Arachidonic acid is a polyunsaturated omega-6-fatty acid present in the phospholipids. Eicosanoids…
Q: What is the structural basis for enzyme specificity?
A: Enzymes are essentially proteins that act as a catalyst for biological reactions. They act by…
Q: What is catalase and why do some microorganisms have it?
A: Catalase is a tetramer of 4 polypeptide chains, each over 500 amino acids long. It contains 4…
Q: The following structure is sorbose Answer the following questions and briefly explain Draw…
A: Carbohydrates are the major source of energy for most organisms present on the earth. Carbohydrates…
Q: Which of the following differentiates erythrose with erythrulose? Erythrose has 2 chiral carbon…
A: Erythrose and erythrulose are monosaccharide sugars that consist of four carbon atoms. They differ…
Q: Describe the function implied by the name of each of the following enzymes Citrate decarboxylase…
A: The enzymes are essentially the biocatalysts present in all living systems. Each enzyme catalyzes a…
Q: Considering the monosaccharide transformation given below, and given that the reactant is a-D-…
A: As the substrate given is alpha - D- mannose which undergoes oxidation and form carboxylic acid…
Q: Explain the process of polyacrylamide reaction, and what are the two reagents are crucial for this…
A: Polyacrylamide is defined as a mesh like matrix that is suitable for the separation of proteins of…
Q: Which step in lipid metabolism would you expect to be affected by 3,4-dihydroxybutyl-1-phosphonic…
A: Lipid metabolism is the synthesis and degradation of lipids in cells, involving the breakdown or…
Q: Saccharomyces cerevisiae is used in the production of wine and beer, while Lactobacillus acidophilus…
A: Introduction :- Saccharomyces cerevisiae is an eukaryotic organism which is used in the production…
Q: Identify all the possible products when the tripeptide Ala-Val-Ala undergoes partial hydrolysis
A: Proteolytic enzymes hydrolyze proteins at optimum temperature and pH and cleaves bonds between them.…
Q: Refer to the lipid structure below. CH, O-C-(CH,),,CH, CH-O-C-(CH,),,CH, NH,+ CH O-P-O-CH,-CH-COO-…
A: Lipids are biomolecules which serves a variety of functions in lifeforms as it undergoes specific…
Q: 4.) C-3 epimer of Fructose
A: Stereoisomers are molecules that have the same chemical constitution but with different…
Q: What is the pI value of phosphoglucomutase?
A: Phosphoglucomutase is a chemical that moves a phosphate bunch on an α-D-glucose monomer from the 1'…
Q: Give the most common monosaccharide derivatives?
A: Monosaccharides are sugars that cannot be further hydrolyzed into simpler carbohydrate compounds.…
Step by step
Solved in 3 steps

- Mevalonate is a component of which biochemical pathway?This is a conjectural question: If the reactive part of coenzyme A is the thioester, why is the molecule socomplicated?Looking at the structure of alpha linolenic acid, how many rounds of beta oxidation will occur to fully oxidize the fatty acid into acetyl CoA molecules? Please provide explanations
- name the oxidation/reduction product for the given monosaccharideIn Bacillus subtilis, threonine is metabolized by the following sequence of reactions: (a) oxidation; (b) decarboxylation;(c) transamination; and (d) oxidation to produce pyruvate. Outline this sequence of steps, show the structures of thesubstances, use abbreviations for the cofactors, and show any enzyme-bound coenzymes. What type of enzyme bound intermediate is likely for reaction (d)?If the w-carbon atom of a fatty acyl CoA molecule with a C18 chain is labeled with C14, how many rounds of β-oxidation are required to produce a radioactively labeled acetyl CoA? a. four b. five c. six d. seven e. eight f. nine
- label all the carbon atoms in glutamine (1-5) and nitrogen (1-2) and show how the molecule is metabolized, which enzymes and cofactors are needed to do so and what metabolic pathway do their products lead into?How many more acetyl CoA are generated from stearic acid than from linoleic acid during beta oxidation?Why is oxidative decarboxylation important?

