Q: .CI НО + y lad fa Lah CI HO HO В E A
A: Acid chloride react with alcohols to form ester.
Q: 17.36 How would you carry out the following transformations? CO,H CO,H (a) CO,H CH,OH (b)
A:
Q: How do the mechanisms of the following reactions differ?
A:
Q: :0: :NH2 NH3 NH, CN" hydrolysis R `H R CN R a-amino acids can be prepared by the Strecker synthesis,…
A: - anion abstracts the most acidic proton - also hydrogen linked to nitrogen is also acidic proton
Q: 1. Identify the reagents for each of the following reactions: (a) он (b) (c) он (d) (e) OCH H.
A:
Q: 3. Using only cyclopentane and methanol as your carbon sources, synthesize the following compounds:…
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: 5. For each reaction, predict the product and draw a mechanism for its formation. HBr HCI (a) (b)…
A: Since we only answer up to 3 subparts. Please resubmit the question with other sub-parts (up to 3)…
Q: QI. Provide all of the necessary reagents for the following reactions. Me CO,H (a)
A:
Q: Identify the MAJOR product that would be obtained from the following reaction. A" `MgBr Me Me then…
A: Given : structure of reactant.
Q: Select the correct set of reagents to carry out the following transformations: G HO HO to OH CI A…
A:
Q: Which of the following carboxylic acid is the proper product of the reaction of 2- oxacyclobutanone…
A: We have to predict the product of given reaction.
Q: Pd (OAC) 2 ( CH) N PPh, HO Br Br
A: In benzene bromination followed by Friedel craft acylation gives p-bromoacetophenon product.…
Q: 11. Choose the major product. e (A) O... (B) meo 12. Choose the major product. E + C TEN mes hv sell…
A: This are the examples of pericyclic reaction. Pericyclic reactions are those reactions which occurs…
Q: Synthesize the following compounds from the indicated starting material. OH a) Br Ph OH b) Br CHCH3…
A: Given first two product we can get by grignard addition reaction. And last can be get by elimination…
Q: h) co,H CH,DH „CH;SH
A: The given transformation cab be carried out as: 3-phenyl-prop-2-en-1-oic acid…
Q: What is the major product formed in the following reaction? A A В в C C X D Your answer E E NaOCH3…
A: NaOCH3 with CH3OH can act as a strong nucleophile as well as base .It is not a bulky base , so the…
Q: Propose a mechanism for the following reaction and account for the stereochemical outcome. Br OH…
A: The reaction given is,
Q: OH H (b) H (c) OH (d) (e) OCH
A:
Q: CH20 L Et H2 Me e OMe sive the product L of the reaction. Draw a mechanism for the formation of L.…
A: Due to technical error ,unable to provide you the solution. Please resubmit the question once again
Q: 2. (J. A) .h). Show how to achieve the following transformations. ??? ??? OH
A:
Q: Using any alkene and any other reagents, how would you prepare the following compounds? a. CH₂CHCH₂…
A: We are authorised to solve only first three subparts of a question. Please post rest of the subparts…
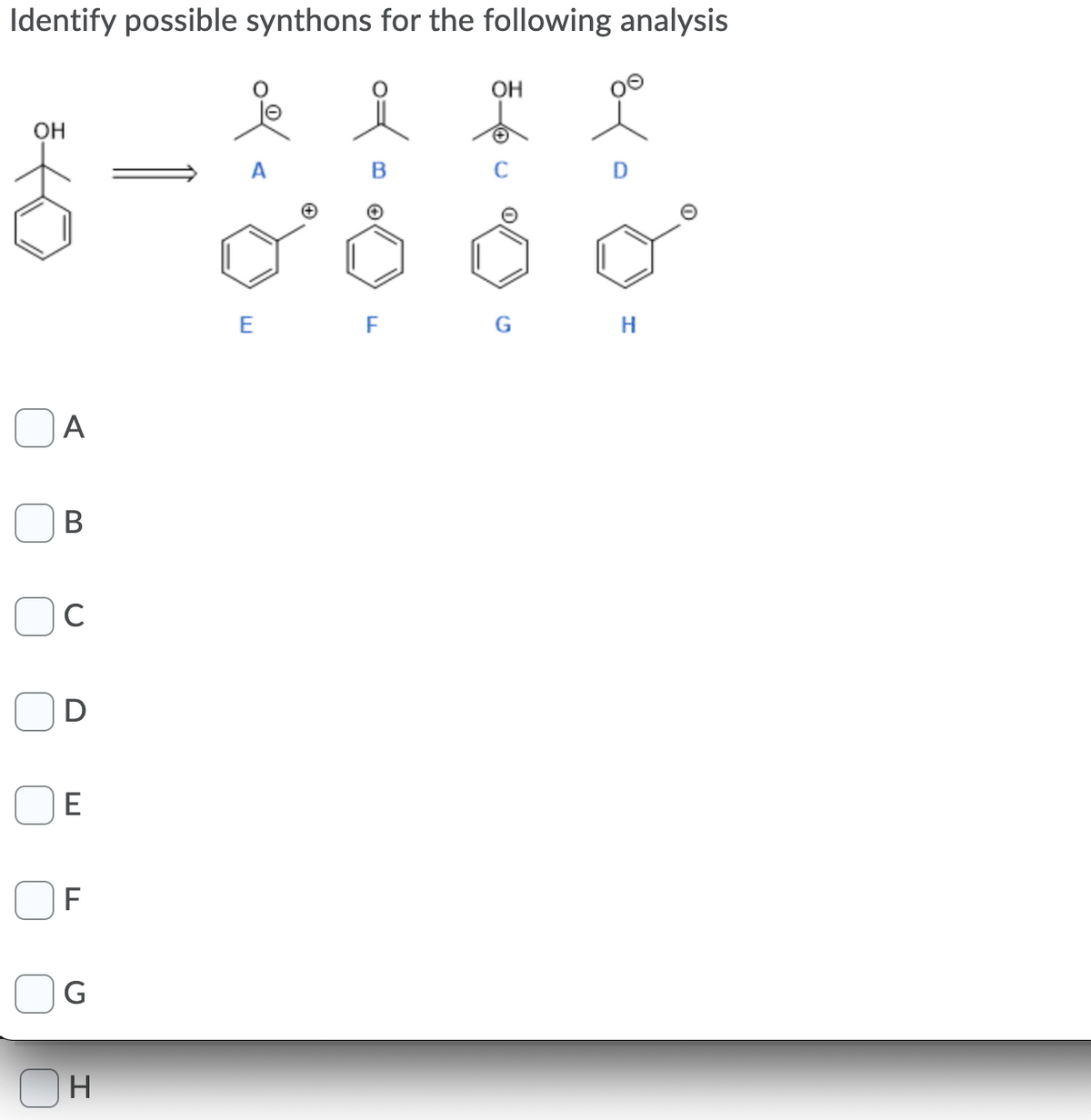
Q: Which of the following synthons represents the reagent below. R OH R CH2* .C. В. R A. R CH2* С. D.…
A:
Q: What is the major product of the following reaction? NH* CrO3CI HO, CH2C12 O a. butanone O b.…
A:
Q: How do the mechanisms of the following reactions differ?
A:
Q: What ester and what Grignard reagent might you start with to prepare the following alcohols? HO CH3…
A: The Grignard reagent reacts with the eater to form ketone initially. The ketone reacts with the…
Q: 11. What products would you expect from the reaction of 2-iodopentane with the following reagents:…
A:
Q: i.) j.) k.) Br Br Br NaOH NaOMe NaOEt
A:
Q: но он Please propose possible mechanisms for the following reactions. H HH HH H H (i) ROOR H CH3 H D…
A: Answer:- i) Natural peroxides are natural mixtures containing the peroxide useful gathering (ROOR′).…
Q: Choose the major product(s) resulting from the following reaction: H3O* НЕАТ A NH2 O NH3 ONHS NH2 HO…
A:
Q: Propose mechanisms and show the expected products of the following reactions.…
A: Solution: The reaction of 2,4-dimethyl chlorobenzene with sodium hydroxide is an example of aromatic…
Q: 10. What starting material would result in the following transformation? он но. A. H. C. OH но. D.…
A:
Q: I. Identify the reagents for the following transformations. Br HO OH OH он OH Br Br Но OH + en HO HO…
A: The reaction in which alkene converts to alcohol in presence of BH3.THF and H2O2 is known as…
Q: What is the product of the following set of reactions? 1. BH3-THF HOT ..... Me 2. SOC, ElN NEt2 HO…
A:
Q: Identify the MAJOR product that would be obtained from the following reaction. H,PO, You OH но
A:
Q: Which of the following reactions will not produce the major product given? A A B C C X D Your answer…
A: The correct option can be choosen by looking at each reaction & their mechanism.
Q: Addition of HBr to 1-phenylpropene yields only (1- bromopropyl) benzene. Propose a mechanism for the…
A: The reaction taking place is given as,
Q: 43. 5-Androstene-3,17-dione is isomerized to 4-androstene-3.17-dione by hydroxide ion. Propose a…
A: Hydroxide ion, OH-(aq) being a strong base, attacks the acidic proton at α-position to the carbonyl…
Q: Dichlorocarbene can also be generated by heating sodium trichloroacetate. Propose a mechanism for…
A: Please find below the pic for the reaction taking place.
Q: Which of the following compounds A-D would you expect as a product for the following reaction? If…
A:
Q: Draw the mechanism to obtain the following stereoisomers from the reaction of trans- 2-butene with…
A:
Q: 10). Which of the following reactions produces alcohol after treatment with H3O+? Answer A) B) D) E)…
A: We will discuss about the product of above reaction.
Q: 19. Identify the reagents that will accomplish each of the following transformations: (a) OMe (c) CN…
A:
Q: What is the product of the following reaction? Me H. 1) Br, H,0 H Me 2) NaOH A. B. C. D. E. но Br но…
A:
Q: ow might you prepare the following esters from the corresponding acids? (a) b) CHCHCHCH H C
A:
Q: select whether the mechanism involves a functional group conversion or a carbon skeleton…
A: Whether the given mechanism involves a functional group conversion or a carbon skeleton modification…
Q: 17. Choose the most suitable product for the following transformation. HO NH2 Single product но…
A:
Q: (a) OH (b) CN CI HO (c) но
A: Reactions are multisteps Reaction.
Q: What is the product of the following transformations? 1. HNO3, H2SO4 2. NaOMe 3. Fe/HCI OMe NH2 H2N…
A: The complete reaction for the above compound with given reagents is given below :
Q: What is the major organic product obtained from the following sequence of reactions? Mg H20 Br Et,0…
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- • Which of the following substrate should be used to synthesize 3-methylhexanal from 3-methylhexanol A.) KMnO₄ B.) F₂ C.) PCC D.) Both A and B E.) None of the given choicesShow how the synthetic scheme developed in Problem 23.67 can be modified to synthesize this triiodobenzoic acid X-ray contrast agent.Following is a synthesis for toremifene, a nonsteroidal estrogen antagonist whose structure is closely related to that of tamoxifen. (a) This synthesis makes use of two blocking groups, the benzyl (Bn) group and the tetrahydropyranyl (THP) group. Draw a structural formula of each group and describe the experimental conditions under which it is attached and removed. (b) Discuss the chemical logic behind the use of each blocking group in this synthesis. (c) Propose a mechanism for the conversion of D to E. (d) Propose a mechanism for the conversion of F to toremifene. (e) Is toremifene chiral? If so, which of the possible stereoisomers are formed in this synthesis?
- One step in the urea cycle for ridding the body of ammonia is the conversion of argininosuccinate to the amino acid arginine plus fumarate. Propose a mechanism for the reaction, and show the structure of arginine.Propose a synthesis of diethylstilbestrol (Problem 27-44) from phenol and any other organic compound required.Azlactones are important starting materials used in the synthesis of dehydro α-aminoacids. They react with aldehydes to form an intermediate that is hydrolyzed under acidic conditions to give the final amino acid product. Provide the structure of the intermediate and propose a mechanism for its formation.
- Amino acids can be prepared by reaction of alkyl halides with diethyl acetamidomalonate, followed by heating the initial alkylation product with aqueous HCl. Show how you would prepare alanine, CH3CH(NH2)CO2H, one of the twenty amino acids found in proteins, and propose a mechanism for acid-catalyzed conversion of the initial alkylation product to the amino acid.In the Ritter reaction, an alkene reacts with a nitrile in the presence of strong aqueous sulfuric acid to yield an amide. Propose a mechanism.Dihydropyran is synthesized by treating tetrahydrofurfuryl alcohol with an arenesulfonic acid, ArSO3H. Propose a mechanism for this conversion.
- In the iodoform reaction, a triiodomethyl ketone reacts with aqueous NaOH to yield a carboxylate ion and iodoform (triiodomethane). Propose a mechanism for this reaction.Treatment of 1-aminoadamantane, C10H17N, with methyl 2,4-dibromobutanoate in the presence of a nonnucleophilic base, R3N, involves two successive SN2 reactions and gives compound A. Propose a structural formula for compound A.Treatment of 2-cyclohexenone with HCN/KCN yields a saturated keto nitrile rather than an unsaturated cyanohydrin. Show the structure of the product, and propose a mechanism for the reaction.



