Q: None
A:
Q: For the reaction 2 H2(g) + O2(g) → 2 H₂O(g) which equation would be used to determine the entropy…
A: Step 1: Step 2: entropy change can be determined by calculating the change in number of gaseous…
Q: How to calculate the pH of a buffer solution 1) Calculate the pH of a solution prepared by…
A: Step 1: Step 2: Step 3: Step 4:
Q: None
A: Thank you.
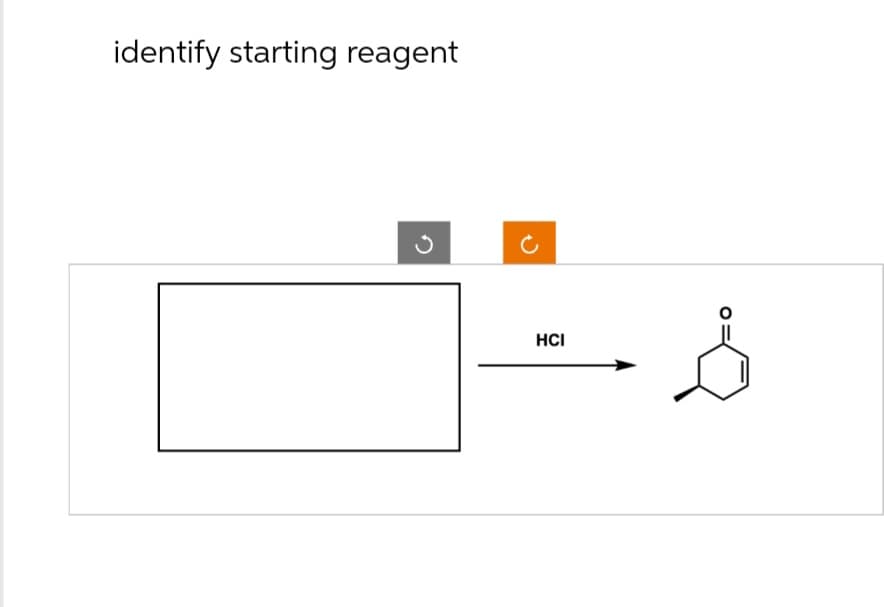
Q: 12) How would you synthesize the following molecule using a Robinson annulation? H3C -CH3 CH3 CH3
A: EXPLANATION:Robinson annulation = Michael addition + Aldol condensationThe backward synthesis has…
Q: For the reaction CO(g) + 3H2(g) → CH4(g) + H2O(g) AH = -206.1 kJ and AS° = -214.7 J/K The standard…
A: The objective of this question is to calculate the standard free energy change (ΔG°) for the given…
Q: b write a mechanism for the reaction using curved arrows to show electron reorganization.…
A: Step 1: Step 2: Step 3: Step 4:
Q: Please fill in the blanks with appropriate, reactants, products, or reagents. CI ? NaSH ? DMF Br KCN…
A:
Q: None
A:
Q: Sodium and water react according to the equation 2Na(s)+ 2H2O(l) → 2NaOH(aq) + H2(g) What number of…
A: Thank you.
Q: 1) Which of the following is NOT part of the kinetic theory of gases? A) A gas is composed of very…
A: 1. Option B is not part of Kinetic theory of gases. According to the theory, particles are tiny and…
Q: Please answer this question with steps thanks.
A:
Q: 6. Starting with benzyl bromide, show how you would synthesize each of the following: (a) (b) OH (c)…
A: a: To synthesize phenyl-2-propanol from benzyl bromide, the following steps can be…
Q: At the end of the experiment you will be assessing the purity of your aspirin by measuring its…
A: Step :
Q: Calculate the volume percent of solute in each of the solutions. A solution made by adding 20.1 mL…
A: The objective of the question is to calculate the volume percent of solute in the second solution.…
Q: Determine the ΔG° in KJ for a voltaic cell made of a Ag strip and an Al strip immersed in 1.0 M…
A: 1. ExplanationOxidation half reaction Pb(s) <-------> Pb2+(aq) + 2e- E°red =…
Q: 6. Which compound has the greater ? (300 nm is a greater than 250 m.) .0 and "NH 10 and b. NH, 01-0…
A:
Q: How to solve for polydispersity if a. Using Mass and Molecular Weight b. using Mole Fraction and…
A: The distribution of molecular weights in a polymer sample is referred to as polydispersity. Several…
Q: The wow expert Hand written solution is not allowed.
A: Step 1:v Step 2: Step 3: Step 4:
Q: Classify each of the following organic reactions in the table below. w-་-c“w,-་ H- -CH2-CH₂-CH₂…
A: 1: In this process an ethyl group is removed from the main group, which is known as elimination.2:In…
Q: The chemical formulae of some acids are listed in the first column of the table below, and in the…
A: 1.) HNO3 (Strong Acid): When nitric acid (HNO3) is dissolved in a liter of water, it dissociates…
Q: i need solution please help me my question answer.
A: The objective of this question is to calculate the molar concentration of glucose in a solution…
Q: 2. The following table gives the values of the preferential adsorption (Qe) and respective…
A: Ce (mol/L)Qe (mol/g-charcoal)0.01814.660 x 10-40.88272.479 x 10-3The concentration of the…
Q: 127. Write a plausible reaction sequence of 4 steps starting from A, B, C, or D. You may use any…
A: The objective of the question is to create a plausible reaction sequence of 4 steps starting from…
Q: None
A: Eugenol is a naturally occurring compound found in essential oils of cloves, nutmeg, cinnamon, and…
Q: None
A: To solve this problem, we need to use the Arrhenius equation, which relates the rate constant (k) of…
Q: A mixture of NaHCO3 and Na2CO3 has a mass of 2.52 g. When treated with HCl(aq). 661 mL of CO2 gas is…
A: Given:…
Q: Hoping for solutions since I’m having a hard time with this. Pls skip if unsure or not willing to…
A: The objective of the question is toIdentify which weak acid system is best to use in preparing…
Q: None
A: Answer well explained above
Q: 3. For each of the following alkenes, provide a reasonable phosporus ylide reagent. CH2CH3
A: The objective of the question is to identify a suitable phosphorus ylide reagent for the given…
Q: CONCEPT QUESTION 1. Retrosynthesis reaction. For the product given below, show the retrosynthesis…
A: Step 1:Step 2:The retrosynthesis and the forward synthesis of the target molecule are given. Step 3:…
Q: 1. Propose reagents for the following reaction. (Hint: Only one of the two ways of adding a carbon…
A: Here's the step-by-step mechanism for the conversion of a 1,4-halohydrin to a lactone. In your case,…
Q: You are performing a titration of 25.00 mL of 0.0100 M Sn2+ in 1 M HCl with 0.0200 M Fe3+ resulting…
A: “Since you have posted a question with multiple sub parts, we will provide the solution only to the…
Q: Draw an alkyl halide and choose the best reaction conditions that would undergo an SN2 reaction to…
A: Step 1: Step 2: Step 3: Step 4:
Q: None
A: Avogadro's Law describes how gases behave at Standard Temperature and Pressure (STP), which is…
Q: Good expert don't upload any image give me the answer with proper explanation please.
A: Step 1:To determine the exact configuration of the molecules, the R/ S configuration theory is…
Q: Please color the answer
A: If you further need clarification, comment please Thanks
Q: A buffer is prepared by adding 5.00 g sodium acetate (MW = 82.0343 g/mol) and 10.0 mL of 5.00 %…
A: The objective of this question is to determine the amount of strong base that can be added to a…
Q: For each of the following compounds, decide whether the compound's solubility in aqueous solution…
A: The solubility of a compound in water can depend on the pH of the solution, especially if the…
Q: 2) Complete the following multi-step synthesis. Protecting groups will probably be required. (6 pts)…
A: Step 1: Step 2: Step 3: Step 4:
Q: pH i got from previous question which was calculate the pH of a 1.00L buffer solution that is 0.100M…
A: To calculate the pH of the buffer solution after the addition of NaOH, we need to consider the…
Q: Thinking 16. Find the quadratic function such that f(4) = -26, f'(4) = -19 and f "(4) = -6. [ 17.…
A: Step 1:solution (16) Given f(4) = −26, f′(4) = −19, f″(4) = −6The objective of the problem is to…
Q: Draw bond line structures for the following (a.) 4-Isopropyldecane (b.) 2,2-dichloropropane (c.)…
A:
Q: For the next compounds/ions, draw two (2) appropriate Lewis structures. One structure must obey the…
A: Lewis structures represent atoms by showing how they share electrons when they form a molecule.…
Q: (7 points) Calculate the silver ion concentration, [Ag*], of a solution prepared by dissolving 8.25…
A: Step 1:To find the silver ion concentration [Ag+],in the solution after dissolving 8.15g of AgNO3…
Q: Show the mechanism and all products formed when benzoic anhydride is treated with the nucleophile…
A: The objective of the question is to understand the reaction mechanism and identify the products when…
Q: X INCORRECT; see section 18.1 A piece of a wooden tool found at a Northwest Indian fishing village…
A:
Q: Assign 1H NMR chemical shifts (ppm values) to the hydrogens indicated in…
A: In the given question, we have to assign the 1H NMR chemical shifts to the protons associated with…
Q: 3. Provide a mechanism for the following reaction. OMe a) MeLi (2 equiv) b) H3O+ HO Me Me
A:
Q: Consider the following reaction: 2 C2H6(1) + 702(g ) à 4 CO2(g) + 6 H2O(1) DH = 367.4 KJ Calculate…
A: The objective of the question is to calculate the enthalpy change (ΔHrxn) for the formation of 849 g…
Step by step
Solved in 2 steps with 5 images

- Label compounds B–D as stereoisomers, conformations, or constitutional isomers of A.Draw the products formed when p-methylaniline (p-CH3C6H4NH2) is treated with each reagent.a. HClb. CH3COClc. (CH3CO)2Od. excess CH3Ie. (CH3)2C = Of. CH3COCl, AlCl3g. CH3CO2Hh. NaNO2, HCli. Part (b), then CH3COCl, AlCl3j. CH3CHO, NaBH3CNRank the following groups in order of decreasing priority. −C≡CH, −CH(CH3)2, −CH2CH3, −CH=CH2
- Captopril is a drug used to treat high blood pressure and congestiveheart failure.Designate each stereogenic center as R or S.hi! i need answers on letters d to f. The instructions says give the IUPAC name of each compound. Thank u!Draw the products formed when each alkene is treated with BH3 followed by H2O2,HO−. Include the stereochemistry at all stereogenic centers.

