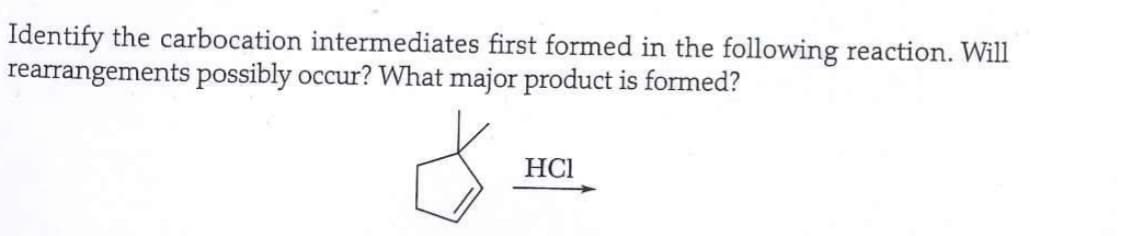
Identify the carbocation intermediates first formed in the following reaction. Will rearrangements possibly occur? What major product is formed? 8 HCI
Q: talk about specific ruthenium-phosphine reaction with oxygen
A: Asking is, Reaction of Ru with oxygen? Reaction of phosphene with oxygen?
Q: Consider the equilibrium system described by the chemical reaction below. Determine the…
A: Given: Kc = 2.4×10-3 Equilibrium concentration of H2O = 0.11 M Equilibrium concentration of H2 =…
Q: 4. Suppose you live in a different universe where a different set of quantum numbers is required to…
A: N is the principal quantum number which gives information about the main shell or main energy level…
Q: 2Hg²+ (aq) + 2e Hg₂²+ (aq) + 2e¯ calculate AG Hg₂+ (aq) Mac Hg₂ 2+ (aq) 2Hg(1) = 0.92 V Eº = 0.85 V…
A:
Q: Calculate the pCa vs. EDTA titration curve for 50.0 mL of 0.0060 M Ca2+ being titrated with 0.0150 M…
A:
Q: Complete the table by calculating the amounts of colored after and distilled water required to make…
A: We need to complete the table by calculating the amounts of colored after and distilled water…
Q: Diels-Alder Product CN CN CO₂Me CO₂Me
A:
Q: Propose a mechanism for the following reaction using only the reagents given. Make sure to clearly…
A: In presence of heat these compounds undergoes pericyclic reaction involving pi electron density.
Q: 17) Chemical Formulae: C₁2H18 8 4H, Doublet Triplet Triplet Doublet 7 6 5 4 PPM Quartet, 2H 3…
A: 1H-nmr provides the information of hydrogen environment of the unknown compound.
Q: 10 Draw a structural formula for the cycloalkene with the molecular formula CH₁0 reacts with Cl, to…
A:
Q: galvanic cell runs for 1.0 minute with a current of 0.65 A. How much charge passed through the cell…
A: Well, we know charge is basically the multiplication of current(A) and time(sec). Data are already…
Q: For this experiment you used 0.25 g of sodium bicarbonate, which is a relatively small mass of…
A: A chemical reaction always mantains the law of conservation of mass So when we Increases the…
Q: the reaction of 1.88 g of salicylic acid (MM = 138.122 g/mol) with 1.09 g of acetic anhydride (MM =…
A:
Q: he free energy for a reaction can be related to the equilibrium constant through the formula below.…
A: For the dissociation of borax reaction, standard enthalpy change, ∆Ho = 188.69 kJ/mol standard…
Q: Complete the table below by deciding whether a precipitate forms when aqueous solutions A and B are…
A: Here, we have to find whether a precipitate will form or not when the given solutions are mixed.
Q: Suppose a 500. mL flask is filled with 0.70 mol of NO3 and 1.0 mol of NO. The following reaction…
A:
Q: Use the following equation to answer the question below. 3 CaO + 2 NF3 → 3 CaF2 + N2O3 What is…
A:
Q: Macmillan Learning If a gaseous mixture is made by combining 4.66 g Ar and 4.71 g Kr in an evacuated…
A:
Q: Q13) 0.35 g of sodium hydroxide (NaOH) pellets are dissolved in water to make 8.0 L of solution.…
A: “Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: Which hydrogen is most acidic? (A) H. (B) H H (C) (D)
A:
Q: Macmillan Learning Hydrogen produced from a hydrolysis reaction was collected over water. The data…
A: Given: Total volume of H2 collected = 93.84 mL Temperature = 23.0°C Barometer pressure = 737 mm Hg…
Q: Phosphorus reacts with oxygen to form diphosphorus pentoxide, P₂O5. 4 P(s) + 50₂(g) 2P₂05 (s) How…
A:
Q: (d) 2.73 kg of CuSO4 · 7 H₂O in 309 L of solution 4.0 M
A: Answer:- This question is answered by using the simple concept of calculation of molarity using the…
Q: Question 1 If an octahedral iron(II) complex is paramagnetic, which of the following sets of…
A: Given: An octahedral iron (II) complex is paramagnetic. We have to find the correct condition to…
Q: At high temperatures, iodine molecules can dissociate into iodine atoms. For the reaction I₂(g) ⇌ 2…
A:
Q: What is the IUPAC name for the compound below? A) 4-ethyl-5-ethyl-5-methylheptane B)…
A: Rule of IUPAC- 1) Choose the longest carbon chain as parent chain. 2) Numbering start from those…
Q: Question 14 of 34 Write the complete ground-state electron configuration of F-. For multi-digit…
A: The Atomic number of fluorine is 9 as it contains 9 electron in the atom of it. When the fluorine…
Q: structure of the molecule. As you solve the structure, keep the following questions in nd: What is…
A:
Q: When a 1.05 g sample of CuSO4.5 H₂O(s) was heated so that the waters of hydration were driven off,…
A: we have to calculate percentage of water in the hydrate
Q: Write a balanced chemical equation and the net ionic equation for the process where aqueous calcium…
A:
Q: What is the change in entropy when I mole of water at 0 °C is heated to form steam at 110°C at…
A:
Q: Draw the MO diagram for Trihydridoboron. Which orbital on boron is completely nonbonding? Explain…
A: Trihydroboron is formed as a result of interaction of atomic orbitals of boron and three hydrogen…
Q: Why is the alkyl halide below not capable of undergoing an E2 reaction upon treatment with sodium…
A:
Q: Draw the major product from this reaction. Use wedge and dash bonds to indicate relative…
A: -> Alkene can be converted to cyclopropane in presence of CH2I2/Zn/Cu . -> It is Simmons Smith…
Q: For the chemical reaction shown, 2H₂O₂(1) + N₂H₂(1) - 4H₂O(g) + N₂(g) determine how many grams of N₂…
A:
Q: For many purposes we can treat dimethyl ether C2H6O as an ideal gas at temperatures above its…
A:
Q: consider the reaction 2 NO₂(g) → N₂O4(g). (a) Using AGF N₂O4(g) = 97.79 kJ/mol and AGF NO₂(g) = 51.3…
A:
Q: The vapour pressure of a condensed phase a. remains the same b. increases C. decreases as pressure…
A: Answer Vapour pressure of liquid varies with temperature only So…
Q: An unknown was treated with six drops of HCI. A precipitate was observed. Which of the following…
A: This is cation analysis problem. We are adding reagents to precipitate out cations. Hydrochloric…
Q: At which HCl addition was there an indication that all the sodium bicarbonate had been used up?…
A: Sodium bicarbonate consumption can be identified from the balanced reaction.
Q: 10. Compounds C and D form nearly ideal solutions. At 20°C, the vapor pressure of pure C and D are…
A: #10:(a) The phase diagram can be plotted by taking the mole fraction of any one of C and D on the…
Q: Draw structural formulas for the isomeric carbocation intermediates formed on treat- ment of each…
A: Carbocation (carbon + cation) is defined as the carbon having +ve charge along with the three bonds.…
Q: A buffer solution is prepared by mixing equal volumes of 1.0 M NH, and 1.0 M NH CI. How would adding…
A: Given : buffer solution Tip : use common ion concept
Q: Determine the pH if 50.0 mL of 0.50 M HI solution is added to 0.027 L of a 0.10 M KOH solution
A: Given : 50.0 mL of 0.50 M HI solution is added to 0.027 L of a 0.10 M KOH solution Tip : pH = -log…
Q: .Draw the # and molecular orbitals for the discrete and localized T bonds in the following…
A: “Since you have posted a question with multiple sub-parts, we will solve the first three subparts…
Q: how do we determine the ph of weak acid?
A:
Q: When a 1.49-g sample of solid sodium hydroxide dissolves in 31.0 g of water in a coffee-cup…
A: Given - Mass of NaOH = 1.49 g Mass of water = 31.0g Final temperature= 35.07°C Initial temperature=…
Q: Explain what is happening in the following situations in terms of solubility. Part A Fish die in a…
A: Solubility of gases in liquid are exothermic reaction. So increase in temperature decreases the…
Q: A student observes that the solution containing Br₂ is reddish, a solution of Cl₂ is faintly…
A: Given that A student mixes a cyclohexane solution containing Br2 with a halide ion solution…
Q: (b what is the energy, in Joules, of 1.0 mol of photons of green light? 215/mol Energy-
A:
Step by step
Solved in 2 steps with 2 images

- Identify the nucleophile that attacks the carbocation intermediate in the acid catalyzed hydration is shown A. HO- B. H2O C. H+ D. H3O+List the following carbocation intermediates in order from least stable to most stable. Explain why they were ranked this way.Which of the following set of conditions would lead to the Hofmann alkene as the major product (starting from the substrate below)? Options: H2SO4 LDA NaOEt, HOEt NaOH, H2O
- What products are formed when the following molecule reacts with catalytic H2SO4, H2O, and heat? Please show the mechanism for the reaction.What product(s) are formed in the following reaction. Indicate the proper stereochemistry of the product(s).Identify the mechanisms by which each reactions above proceeds from the choices listed.
- A carbocation intermediate is central to which of these reaction types? E1, E2, SN1, SN2 ? Choose 1Illustrate the carbocation pathway for the following reaction below.Identify the leaving group for a potential elimination of the following compounds. Compare the leaving group activities for A and K. Which of the following compounds cannot be subjected to elimination? Explain.
- What alkynes give each of the following ketones as the only product after hydration with H2O, H2SO4, and HgSO4?Answer the following questions about the mechanism for the acid-catalyzed hydration of an alkene: a. How many transition states are there? b. How many intermediates are there? c. Which step in the forward direction has the smallest rate constant?Determine if the carbocation below is expected to undergo a rearrangement. Write NR if norearrangement will occur. If a rearrangement is expected, write the type of shift above the arrow,show the mechanism, and draw the resulting structure from the expected rearrangement.

