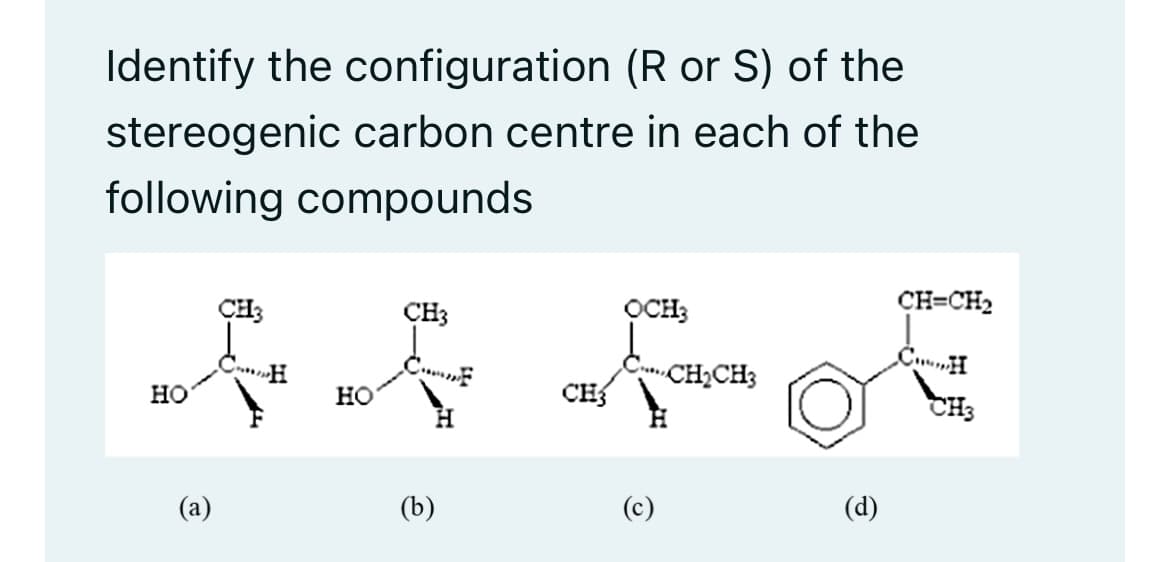
Identify the configuration (R or S) of the stereogenic carbon centre in each of the following compounds HO @ CH3 HO CH3 H (b) CH3 OCH₂ C-CH₂CH3 (d) CH=CH₂ CH CH3
Q: What is the units of the following constant: a Acid value b. Saponification value c. Ester value d.…
A: Fatty acid is a carboxylic acid with an aliphatic chain.The carbon chain may be saturated or…
Q: How many solid ounces of methylcellulose are in 7 pints of 2 % (w/v) methylcellulose solution? Round…
A: First check the amount of methyl cellulose in given solution and then convert it in ounce.
Q: An analytical chemist is titrating 222.2 mL of a 0.3600M solution of methylamine (CH3NH₂) with a…
A:
Q: Write a balanced net ionic equation to show why the solubility of CoCO3 (s) increases in the…
A: We will Write a balanced net ionic equation to show why the solubility of CoCO3 (s) increases in the…
Q: 17. Draw the molecular orbital diagrams for a photochemical (hv) reaction between two molecules of…
A: Here we have to predict the product for the 2nd question and the mechanism and for the first…
Q: in fractional distillation, how do you tell which liquid is in which fraction and which fraction has…
A: FRACTIONAL DISTILLATION is a separation technique in which two or more miscible liquids or gases are…
Q: Write formulas/charges as appropriate for each of the following monatomic ions. C-4
A: Write formulas/charges as appropriate for each of the following monatomic ions.
Q: 18. 19. 20. a. b. C. d. a. b. C. What is the major product of the following reaction? Br Br T T H H…
A:
Q: An analytical chemist is titrating 123.4 mL of a 0.2500M solution of propylamine (C3H₂NH₂) with a…
A: Given Values -> Volume of C3H7NH2 = 123.4 ml Molarity of C3H7NH2 = 0.2500 M Volume of HNO3 = 15.4…
Q: A slice of pizza contains 28g of carbohydrate, 13g of protein and fat. If the pizza contains 243…
A: A slice of pizza contains ; Carbohydrates = 28g Proteins = 13g and fat. The caloric values are ;…
Q: Which C-H bond has the lowest BDE?
A: Given : structure of molecules This is based on stability of radical.
Q: Q10. Coulomb interactions in an atom with 8 electrons is obtained by adding contributions from pairs…
A: Dear student as all questions are not relatable so I can solve only 1 question according to bartleby…
Q: Use the interactive to observe the temperature change that results from burning various fuels in a…
A:
Q: CI O H Classify the compound shown here. MacBook Air A) aldehyde B) ketone C) alcohol D) ester E)…
A: In the above question we have to find out the classification of compound on the basis of functional…
Q: The production capacity for acrylonitrile (C₂H₂N) in the United States exceeds 2 million pounds per…
A:
Q: What is the major organic product of the reaction below? 0.0 OA B H₂C CH₂C N+CH₂MgBr NH₂ CH A CH₂…
A:
Q: Grams of MgCl2 to prepare 3.25 L of a 0.950 M MgCl2 solution
A:
Q: A NH3 HO NH B C What is the name of component A of sphingomyelin (shown in pink)? What did the…
A:
Q: dicate which atoms in the following compound are chiral centers. a CO₂H 16 HC OH CH₂ [References]…
A:
Q: For the reaction sequence below, select the expected major product. 1) Br₂ O 3-methyl-2-hexyne O…
A:
Q: What is the functional group of the product of the following reaction? alcohol O aldehyde OH Oketone…
A: -> Grignard reagent has basic character also nucleophilic character hence it can give…
Q: 4.a. Calculate the % ionization: 0.10 M CH3NH2 ; pKa = 10.657 * Your answer 4.b. Calculate the %…
A: First I am going to calculate the Kb value from the given pka value then by writing balanced…
Q: Ten liters of a gas at 396 K and a pressure of 1.12 atm. The pressure is decreased to 0.52 atm…
A:
Q: Name the following compounds 1. 2. 3. sh
A:
Q: < Ethambutol is a chiral drug used to treat tuberculosis. Determine the number of chiral centers and…
A:
Q: calculate the mole fraction of nacl in a solution prepared by dissolving 117 g nacl in 3.45 kg H20
A:
Q: Which structure is trans-3-hexene? OCH₂ H CH3. H OCH₂CH₂ H c=c OCH3CH₂ H FC. FC FC. H CH₂CH₂CH3…
A: Which structure is trans-3-hexene ?
Q: Draw the structure of 2-bromo-4-methylaniline in the window below.
A: Here we are required to structure of 2-bromo-4-methylaniline.
Q: Organic chemistry is the study of: O I have no idea at this point Carbon containing compounds…
A: Organic chemistry is the study of ?
Q: Are the following Ionic Compounds, Covalent Compounds or Acids? Write formulas as appropriate for…
A: Ionic compound: Ionic compound composed of cation which is positively charged (+charge) and an anion…
Q: ving 51. Draw a structural formula for each of the following unsaturated hydrocarbons: (1.2) KUC (a)…
A:
Q: A 35.0 mt sample of 0.225 M HBr was dituted to 42.3 ml, what is the Corrc. of diluted Her ?
A: Concentration of diluted HBr can be calculated using dilution formula.
Q: ra ↑ (1) 503 / H₂ 50g (Oore.:) 2 C/₁₂/FeCl3 Ⓒ H₂0+ / heat not work as am which of the following…
A:
Q: B. Supply the missing arrows to complete the reaction mechanisms below. For each of the reactions,…
A: Here we are required to draw the curly arrow to complete the reaction
Q: Quinine 0.07 0.26 0.19 Codeine 0.16 0.53 0.38 Cocaine 0.65 0.90 0.73 Strychnine 0.28…
A: Rf value: It is the ratio of distance travelled by solute to distance travelled by solvent.…
Q: Write formulas/charges as appropriate for each of the following monatomic ions. a. Calcium ion b.…
A: a) Calcium is Group-II element and it has [Ar]2s2 electronic configuration. •It donate to electron…
Q: hat is the functional group of the major organic product of the reaction below? LIAIH4 H₂C ketone…
A: -> LiAlH4 is reducing agent which can reduce cyanide to primary amine .
Q: How much heat would need to be removed to cool 29.5 g of water from 25.6°C to -10.7°C? Csolid =…
A: We have to predict the amount of heat removed from the water to have given temperature change.
Q: OH A) methanone B) methanoic acid ethanal D) ethanol Jtm E) ethanoic acid
A:
Q: Which of the following could be used to complete the following reaction? ol O CH₂CH3MgBr/ether O…
A:
Q: Convert this pressure to pascals.
A:
Q: 1) Do the following synthesis: (e) Benzene to 1,3-dipropylbenzene (f) Benzene to p-nitrobenzoic…
A: To answer: 1) Do the following synthesis: (e) Benzene to 1,3-dipropylbenzene (f) Benzene to…
Q: Completely answer the following, I will give a good feedback after. Draw structures for each of the…
A: The general formula of acyclic alkane is CnH2n+2 The general formula for cyclic alkane is CnH2n
Q: A 628 mL sample of gas contains 0.0370 moles and kept at 368 K. What is the pressure of the gas in…
A: Ideal gas law is gas law which obeys gas laws under all condition of temperature and pressure.
Q: The rate law for 2NO(g) + O₂(g) → 2NO2(g) is Rate = K[NO]²[0₂] The following mechanisms have been…
A: A question based chemical kinetics that is to be accomplished.
Q: Question 12 Points 3 Which of the following points is/are true for plasma? O It is a very good…
A: Plasma is a very good electrical conductor. Plasma is readily influenced by electric and magnetic…
Q: Suppose that unbeknownst to you, your thermometer consistently reads 2.10 °C too high. Would this…
A: To answer: If temperature reads consistently high, how it'll affect the molar mass of any compound.
Q: At 2300°C, Kc = 2.5 x 10¹2 for the reaction: 203(g) 30₂(g) ← Determine Kp for this reaction.
A:
Q: ch letter is near the most acidic protons in the molecule below. A В с OD А H3C B
A: Hydrogen which bonded with more electronegative atom is most acidic Hydrogen.
Q: Which of the following best describes the reaction below. H₂O*, heat H₂C NHCH, H₂C 'OH O The…
A: Given reaction is an example of hydrolysis of amide in an acidic medium.
Step by step
Solved in 2 steps with 1 images

- When attached halohydrin is treated with NaH, a product of molecularformula C4H8O is formed. Draw the structure of the product and indicateits stereochemistry.When attached halohydrin is treated with NaH, a product of molecular formula C4H8O is formed. Draw the structure of the product and indicateits stereochemistry.a. Draw a three-dimensional structure for the following steroid. b. What is the structure of the single stereoisomer formed by reduction of this ketone wrth H2, Pd-C? Explain why only one stereoisomer is formed.
- 1. What is the relationship between the following two compounds? a.stereoisomersb.identicalc.constitutional isomers 2. Assign the absolute configuration of the chirality center as R or S.Draw compounds that fit the following descriptions: (a) A chiral alcohol with four carbons (b) A chiral carboxylic acid with the formula C5H10O2 (c) A compound with two chirality centers (d) A chiral aldehyde with the formula C3H5BrOa. Idêntify thè cònfigûràtiòn of the dôublè bônd. b. Rèdraw the strùctùre and lábel the chîrál cèntérs usîng an ásterisk. c. Rèdràw the strùctüre with ALL thê chirál cèntèrs in S Cònfigûratîon. d. Dráw the ènantiômer of the strûcture dräwn in letter C. e. identify the number of diastereomers that this compound can have.
- An acid–base reaction of (R)-sec-butylamine with a racemic mixture of 2phenylpropanoic acid forms two products having different melting points and somewhat different solubilities. Draw the structure of these two products. Assign R and S to any stereogenic centers in the products. How are the two products related? Choose from enantiomers, diastereomers, constitutional isomers, or not isomers of each other.Betamethasone is a synthetic anti-inammatory steroid used as a topical cream for itching. Betamethasone is derived from cortisol, with the following structural additions: a C=C between C1 and C2, a uorine at C9, and a methyl group at C16. The configuration at C9 is R, and the configuration at C16 is S. Draw the structure of betamethasone.An unknown compound (whose molecular formula is C5H8O) shows IR absorption at 3600 and 3300 cm^-1. It’s ^1H-NMR spectrum contained singlets at delta 1.5, 2.2, and 2.9 (broad) ppm, in the ratio 6:1:1. Draw the structure for the unknown compound.

