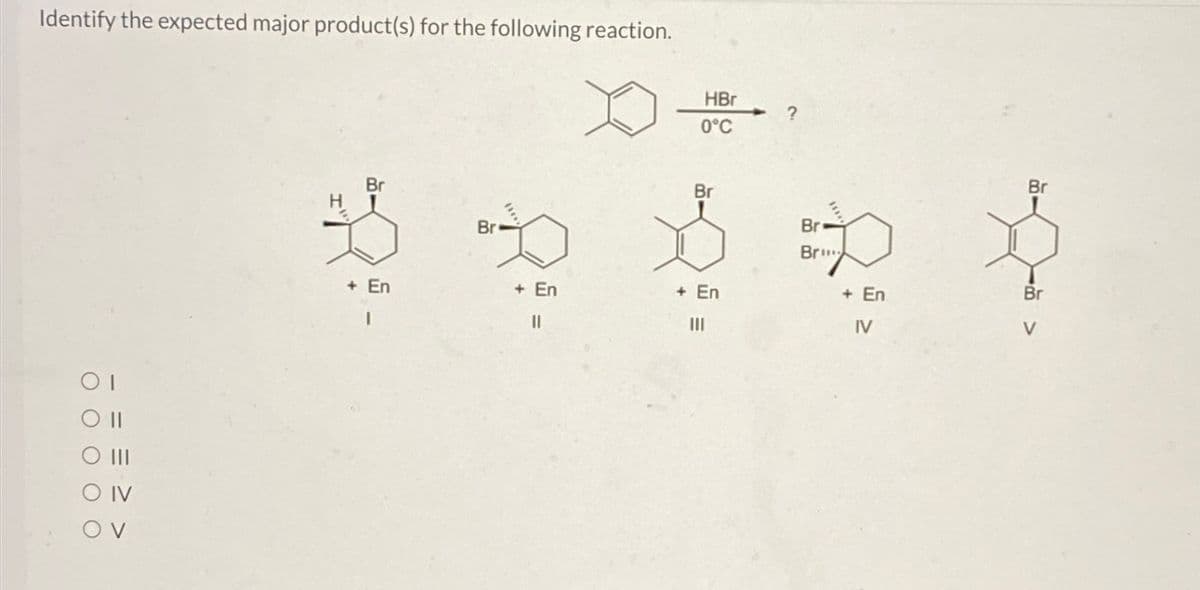
Identify the expected major product(s) for the following reaction. ΟΙ O III O IV OV HBr ? 0°C H Br Br Br Br Br Bri + En + En + En + En Br I III IV V
Q: For the reaction at equilibrium: 2 H2O (g) +2 Cl2 (g) + energy 4 HCI (g) + O2(g), If the temperature…
A: In the endothermic reaction, if the temperature of the reaction is increased, the equilibrium…
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product…
A: We have to predict the mechanism.
Q: Macmillan Learning H&C. CH2 H&C CH HC Consider alkene 2. CH3 ΤΟ =CH2 H2 H₂C 생 Consider alkene 3. H₂C…
A: The objective of the question is to correct the given names of the alkenes. The names of the alkenes…
Q: how to calculate the enthalpy and entropy for experiment " The Thermodynamics of a Galvanic Cell".…
A: Cell Equation:According to Nernst equation, the reaction entropy is related to cell potential by the…
Q: give Iupac name of this product.
A: IUPAC Rules for the nomenclature of the branched organic compounds..First we should select the…
Q: Nitesh
A: The objective of the question is to predict the major product from the reaction of CHO (aldehyde)…
Q: Choose the best reagents to complete the reaction shown below. OH Q
A: Organic reaction mechanisms describe the step-by-step processes by which organic molecules undergo…
Q: Draw an outer electron box diagram for a Mn 4+ cation. 1 し
A: The detailed answer is shown below Explanation:I hope this helps.Thankyou
Q: Please don't provide handwritten solution ..
A: The given reaction is a chemical reaction between an aldehyde (CHO) and an alcohol (HOCH2CH2OH) in…
Q: Curved arrows are used to illustrate the flow of electrons. Use the reaction conditions provided and…
A: We have to predict the intermediate and the product.
Q: You have 50.0mL of 0.122M HCl. You add 10.0mL of 0.25 M NaOH What is the pH of the solution now?
A: The objective of this question is to calculate the pH of a solution after the addition of a base…
Q: What is the concentration of sodium ions in 0.300 M Na₂SO₄?
A: The objective of this question is to find the concentration of sodium ions in a 0.300 M solution of…
Q: The uncertainty in the position of an electron moving with a velocity of 3.0×102 m/s accurate upto…
A:
Q: D.32 Write the IUPAC name for each of the following compounds, including stereochemical…
A: Compounds IUPAC Name: need to be determine
Q: Macmillan Learning H&C. CH2 H&C CH HC Consider alkene 2. CH3 ΤΟ =CH2 H2 H₂C 생 Consider alkene 3. H₂C…
A: The objective of the question is to correct the names of the given alkenes. The names of organic…
Q: 18. Which of the following claims about a binary compound in which the bonding is To most likely to…
A: A binary ionic compound is formed by the electrostatic force of attraction between a cation and…
Q: What is the ionization reaction of HNO3(aq)?
A: The objective of the question is to determine the ionization reaction of nitric acid (HNO3) in…
Q: An unknown weak base with a concentration of 0.0910 M has a pH of 12.10. What is the Kb of this…
A: The Kb of the base is approximately 2.0x10-3.Explanation:Step 1: Let B be the unknown weak base. The…
Q: 8) Devise a Synthesis for the following conversion
A: Given is organic synthesis reaction. The given starting compound is 1-methylcyclohexane.The given…
Q: Consider the two proposed modes for fragmentation of a ketone: + CH2CH3 + CH3 (a) Identify which…
A: The objective of the question is to identify which fragmentation of a ketone, either CH2CH3 or CH3,…
Q: Reaction in an ideal gas system A2(g) + 3 B2(g) → 2 AB3(g) takes place at a temperature of 860 K in…
A: see attachmentExplanation:Step 1: Step 2: Step 3: Step 4:
Q: The rate constant for the decay of a radioactive element is 1.95 × 10⁻³ day⁻¹. What is the half-life…
A: The objective of this question is to calculate the half-life of a radioactive element given its rate…
Q: Use your calculator to find the log of the following numbers.(a) 10−9(b) 1 × 10−11(c) 7.4 × 103(d)…
A: (a). −9 (b). −11 (c). 2+log10(74) Or approximately 3.86922 (d). 5 (e). 0 Explanation:
Q: Calculate the pOH of a 0.0251M HCl solution
A: The objective of this question is to calculate the pOH of a 0.0251M HCl solution. The pOH of a…
Q: Show how the following compound can be synthesized from the indicated carbon sources (you may use…
A: In first step Allylic bromination takes place using NBS to form allyl bromide.The alkyl halide…
Q: Provide analytical techniques use for characterizing products. 1) Provide advantages and…
A: Analytical techniques used for characterizing products include spectroscopy (like UV-Visible,…
Q: Consider the gas-phase reaction: H2(g) + 12(g) → 2 HI(g) The reaction was experimentally determined…
A: The objective of the question is to draw the energy diagrams for the given mechanisms and then show…
Q: Write the rate expression in terms of reactants and products for the following reaction. What is the…
A: Rate of a reaction is defined as the decrease in concentration of reactant in unit time (Or)…
Q: Macmillan L For the given SN2 reaction, draw the organic and inorganic products of the reaction, and…
A: Organic SN2 reaction is given.We have to draw the organic and inorganic products.
Q: 1. a) Draw α-D-glucopyranose (1->6) a-D-fructofuranose
A: 1. a) Draw a-D-glucopyranose (1-6) a-D-fructofuranoseExplanation:
Q: How many grams of sulfuric acid (H2SO4) will be required to make 100.0 mL of 5.0 M solution?
A: The objective of this question is to calculate the amount of sulfuric acid (H2SO4) in grams required…
Q: How many grams of copper sulfate would you need to make 300.0 mL of solution that has a sulfate ion…
A: Volume of copper sulfate (CuSO4) solution = 300.0 mLConcentration of sulfate ion (SO42-) = 35.0…
Q: Q2: Consider a molecular interaction denoted as X-HY, where "-" signifies a covalent bond and "..."…
A: The objective of the question is to determine the expected energies for the covalent structures…
Q: Predict the products of the following reactions. (a) + HONH, H+ H+ + H₂NNH₂ (c) (d) H+ H+ Ph-C-Ph +…
A: I hope this helps you.Explanation:Step 1:Step 2: Step 3: Step 4:
Q: 2. Show a reaction sequence to convert oil of wintergreen (methyl salicylate) into Aspirin (acetyl…
A: For details answer please find the explanation section.Explanation:
Q: iii.Draw the structures of the major organic product(s) or condition(s) denoted by A to E in the…
A: The given reaction is shown below.We have to predict the structure of the major product of the given…
Q: 17. What is the reaction below? A. an addition B. an elimination C. a substitution D. a…
A: A cheletropic reaction is one type of pericyclic reaction in which one atom on one of the reagents…
Q: Click the "draw structure" button to launch the drawing utility. Draw the structure of the acyclic…
A: Carbonyl compound (aldehyde and ketone) reacts with alcohol to form hemiacetal in the presence of an…
Q: Phenol (PhOH, MM = 94.11 g/mol) is a weak acid (pKa = 10.000). a) 0.515 grams of phenol is dissolved…
A: pKa of phenol = 10.00molar mass of phenol = 94.11 g/molmass of phenol = 0.515 gmolarity of KOH =…
Q: Which of the following nuclides are most likely to decay via beta decay? O F-18 OI-131 O CI-37…
A: The objective of the question is to identify the which of the given nuclides are most likely to…
Q: For the reaction at equilibrium: 2 H2O (g) +2 Cl2 (g) + energy4 HCI (g) + O2(g), If the [02] is…
A:
Q: 10. What is the acid dissociation constant for H2PO4* if the pKы1, pKb2, and pKыз for phosphoric…
A: Acidity is defined as the ability of a molecule to release protons.If a compound easily donates…
Q: You need to prepare a carbonic acid/bicarbonate buffer with a pH of 7.350 for an experiment. To…
A:
Q: how many litres of NO can be produced when 25 L of O2 are reacted with 25L of NH3. All gases are at…
A: The objective of the question is to determine the volume of Nitric Oxide (NO) that can be produced…
Q: infrared spectroscopy. the ving pairs of compounds by using
A: IR spectroscopy or infrared spectroscopy is a spectroscopic method for the determination of…
Q: A B C D Stress Strain Illustration B Refer to Illustration B which material is Hard and Strong? Q5.…
A: The first two questions are impossible to answer without the referred Illustration B. The…
Q: 1. 1 equivalent LDA 2. Br
A:
Q: Consider the molecule 1-Chlorobutane A) Draw all 7 Newman Projections for the molecule at C1-C2. B)…
A: The objective of this question is to explain the given conditions of
Q: a) b) What is the conjugate acid of NH3? What is the conjugate base of H2O?
A: (a) The conjugate acid of NH3 is (b) The conjugate base of H2O is Explanation:(a) The substance that…
Q: Calculate the cell potential of the concentration cell described by Ag(s)|Ag+(aq, 0.25 M)||Ag+(aq,…
A:
Step by step
Solved in 3 steps with 4 images

- Predict the products of the following reactions.(a) sec@butylmagnesium iodide + D2O ¡ (b) n@butyllithium + CH3CH2OH ¡ (c) isobutylmagnesium bromide + but@1@yne ¡Predict the major products of the following reactions. Include stereochemistry whereapplicable.(a) 1@methylcycloheptene + BH3 # THF, then H2O2, OHPredict the product of the following reaction and classify the reaction Pb+FeSO4---> PbSO4+ ______
- Predict the major products of the following reactions, including stereochemistry.(a) cyclohexene + KMnO4>H2O (cold, dilute)(b) cyclohexene + peroxyacetic acid in water(c) cis@pent@2@ene + OsO4>H2O2(d) cis@pent@2@ene + peroxyacetic acid in waterPredict the products of each of the following reactions. (a) MnCO3(s) + HI(aq) ⟶(b) CoO(s) + O2(g) ⟶(c) La(s) + O2(g) ⟶(d) V(s) + VCl4(s) ⟶(e) Co(s) + xsF2(g) ⟶(f) CrO3(s) + CsOH(aq) ⟶In which compound is the halogen substituted most rapidly by aq hydroxide ions? a. CH3CH2CH2CH2Cl b. (CH3)3CI c. (CH3)3CCl d. CH3CH2CH2CH2I
- Predict the major products of the following reactions, including stereochemistry.(a) cyclohexene + KMnO4>H2O (cold, dilute)(b) cyclohexene + peroxyacetic acid in waterPredict the products of the following reaction. Co(s) + xsF2(g)---->Give the major organic product(s) for each step in the following reactions (g,h,i,j)

