Q: What is the difference between ferrite powder and ferrous fluid? Compare tabularly
A:
Q: 1. City Cifra bor Consider the following reaction: PCI, (g)+ Cl₂ (g) PCI, (g) K = 4.30 x 10-6…
A:
Q: Use the data given in the table below to draw the S(T)-diagram for the heating up of ethanol from…
A: The table shows the values of Cp and enthalpy of phase changePhase TransitionsCp/J mol-1K-1s(Heating…
Q: 6. A student makes measurements from which she calculates the enthalpy of neutralization as 58.5357…
A: The given outcome for the enthalpy of balance is 58.5357 kJ mol¹. The understudy gauges that her…
Q: Based on the balanced chemical equation shown below, determine the concentration (M) of Fe2+(aq) if…
A: Volume of Fe2+ = 40.00 ml = V1Volume of BrO3- = 43.47 ml = V2Molarity of BrO3- = 0.125 M = M2
Q: A certain metal M forms a soluble sulfate salt M₂(SO4)3. Suppose the left half cell of a galvanic…
A:
Q: Using standard reduction potentials from the ALEKS Data tab, calculate the standard reaction free…
A: Recall the given redox reaction, We have to calculate the standard…
Q: One gram of activated charcoal has a surface area of 1000 m². If complete surface coverage is…
A:
Q: what volume will a sample of f2 gas occupy in a syringe at 5.57 bar is the f2 has a volume of 25.0…
A:
Q: Question 34 How many grams of oxygen (O) are present in a 6.35 g sample of potassium nitrate (KNO3)?…
A:
Q: What reagents are needed to obtain cyclohexyl acetate from 1-cyclohexylethan-1-one
A: To obtain cyclohexyl acetate from 1-cyclohexylethan-1-one, you would typically need the following…
Q: 3. Circle the compound that matches the spectrum provided. Note that the peak at 1.5 ppm exchanges…
A: NMR spectroscopy is an analytical technique for structure elucidation of a compound. 1H NMR is used…
Q: N2(g) +202(g) 2 NO2(g) Initially, 0.45 mol/L of nitrogen and 0.68 mol/L of oxygen are introduced…
A:
Q: 10:28 PM Fr Jan 2 Determine if the molecule shown below is chiral. here or pull up for additional…
A:
Q: Give only typing answer with explanation and conclusion Draw out the dipole moments occurring in…
A: Para-hydroxyacetophenone and ortho-hydroxyacetophenone are two isomeric compounds with different…
Q: Question 2 A mixture of cyclohexane (b.p 81 °C) and toluene (b.p 110.8 °C) need to be separated and…
A: Given that, a mixture of cyclohexane (b.p 81 °C) and toluene (b.p 110.8 °C) needs to be separated…
Q: 35.0 mL of 0.255 M HNO3 is added to 45.0 mL of 0.660 M Mg(NO3)2. What is the concentration of…
A: Volume of HNO3 = V1 = 35.0 mlMolarity of HNO3 = M1 = 0.255 MVolume of Mg(NO3)2 = V2 = 45.0…
Q: Classify the following unbalanced chemical equations according to the six types of chemical…
A: A. Combination reaction;-two or more compounds combine to form one compound. B. Decomposition:- a…
Q: Calculate the reaction entropy of the reduction of sulfur dioxide to hydrogen sulfide at a…
A: Given Reaction: Reduction of sulfur dioxide to hydrogen sulfideData:To Determine:Entropy for…
Q: What mass in grams of precipitate theoretically forms when 0.500L of 0.100M calcium hydroxide is…
A:
Q: Consider the following equilibrium: N₂(g) + 3H₂(g)2NH3(g) AG = -34. KJ Now suppose a reaction vessel…
A: Because, initial amount of H2 gas is not given in atm, so equilibrium of the reaction will shift…
Q: What is the molarity of C6H12O6 in an aqueous solution that is prepared by dissolving a 18.05 g…
A:
Q: Question 3 A mixture of four fictitious organic liquids are added to a flask, and a student attempts…
A: Simple distillation is a process to separate the mixture of two or more liquids based on the…
Q: Each of the Lewis structures shown contains one polar covalent bond. Identify the bond, and use the…
A: Polar covalent are the covalent bonds which have higher electronegativity difference between the…
Q: A chemist dissolves 575. mg of pure hydrochloric acid in enough water to make up 380. mL of…
A:
Q: Determine the shape and bond angle of these oxynitrogen ions. Linear 180° NO Trigonal planar 120°…
A: Shapes of molecules can be found based on VSEPR theory
Q: 1. In which example does oxygen have a 1- charge? a. Sc₂03 b. Li₂O c. H₂O2 d. CO2
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: What is the pH of a bleach solution that has a [OH-] = 1.0 x 10-³ M?
A: The concentration of the hydroxide ion iswe have to calculate the pH of the solution that is
Q: What volume of 0.181 MNa3PO4 solution is necessary to completely react with 87.9 mL of 0.105 MCuCl2
A: The balanced reaction between Na3PO4 and CuCl2 is
Q: Complete the Avogadro's Statement for the element hydrogen: 1 mole of hydrogen hydrogen atoms =…
A: A mole is defined as that amount of the substance which has mass equal to gram atomic mass if the…
Q: Spell out the full name of the compound. Submit Request Answer Part E Spell out the full name of the…
A: To spell out the full name of the compound.According to IUPAC nomenclature,…
Q: In the following E2 reaction step, identify the reactant that contributes the HOMO and the reactant…
A: During reaction electron is transferred from HOMO of one reactant to LUMO of other
Q: Select the true statements. BF3 has a trigonal planar shape. Trigonal planar molecules have three…
A: To identify the true statements from given question.a) BF3 has trigonal planar shape.Since in BF3,…
Q: CH3 B CI CH3 H Br H3C P CH3 H Br H H H3C CH3 H H・・・> Н H Br H Br H CH3 CI
A: Chiral Molecule: A Molecule which has non superimposable mirror image. A chiral molecule contains a…
Q: determine the vaopur pressure of a solution at 28 degree celsius that contains 62.5 g of fructose in…
A:
Q: 5. Draw the structure of (S)-1-bromo-1-chloropropane. Take care to indicate the three- dimensional…
A: For the three dimensional orientation of the molecule having S- configuration at carbon number 1.…
Q: Select the molecules that are polar. CH₂Cl2 IOF5 CF4 ASF3
A: A bond is polar when there is electronegative difference between two two bonded atoms. The one…
Q: 3. Assume that you prepare a buffer by mixing 42 ml of 0.26 M acetic acid and 56 ml of 0.15 M sodium…
A:
Q: Complete the following reaction by drawing a structural formula for the product. CH₂CH3 + H₂ Ni or…
A: The given reactant is:When a ketone is reduced by H2 in the presence of Ni or Pt, a secondary…
Q: Based on the Lewis structure and your knowledge of VSEPR theory, approximate the smallest bond angle…
A: Since,According to the VSEPR theory, steric number (which represent hybridization) is the…
Q: dentify the species oxidized, the species reduced, the oxidizing agent and the reducing agent in the…
A:
Q: There are three different possible structures (known as isomers) of a dibromoethene molecule,…
A: Two chemical compounds which have same molecular formula are categorized as isomers. The structure…
Q: A galvanic cell at a temperature of 25.0 °C is powered by the following redox reaction: 2MnO4 (aq) +…
A:
Q: Predict the product and draw the mechanism of its formation. Include lone pairs and charges in your…
A:
Q: Why SnI4 formed under AcOH/ Ac2O condition and SnI2 formed under 2M HCl and nitrogen flow? Please be…
A: The formation of SnI4 or SnI2 depends on the reaction conditions and the oxidation state of tin…
Q: 3. a. Suggest plausible structures and reagents to complete the scheme shown below. Reagent SiMe3 Li…
A: Alkyl lithium is generally prepared by treating alkylhalides with lithium.In this case,…
Q: Classify the following as constitutional isomers, enantiomers, the same compound, or not isomers.…
A: The above 2 compounds are constitutional isomers.
Q: 6. Draw the products of each reaction. a. b. C. HO COOH COOH + NaOCH3 OH OH H₂SO4 H₂SO4
A:
Q: 1. propose a mechanism for the following transformation. 1) Excess LAH HO 2) H₂O OH
A: The given reaction is .We have to draw the mechanism of the above given reaction.Type of reaction:…
Q: 5. To balance a redox reaction in an acidic or basic environment, you would use the following…
A: Following two questions given - (5) Which method used to balance redox reaction in an acidic or…
Step by step
Solved in 3 steps with 3 images

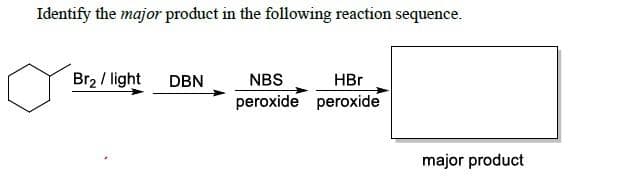
- From 1 being the least reactive and 4 being the most reactive rank the following alkyk chlorides in terms of increasing reactivity via SN2 reaction mechanism.In which compound is the halogen substituted most rapidly by aq hydroxide ions? a. CH3CH2CH2CH2Cl b. (CH3)3CI c. (CH3)3CCl d. CH3CH2CH2CH2IDraw the three likely products of the following reaction. Identify the major product.

