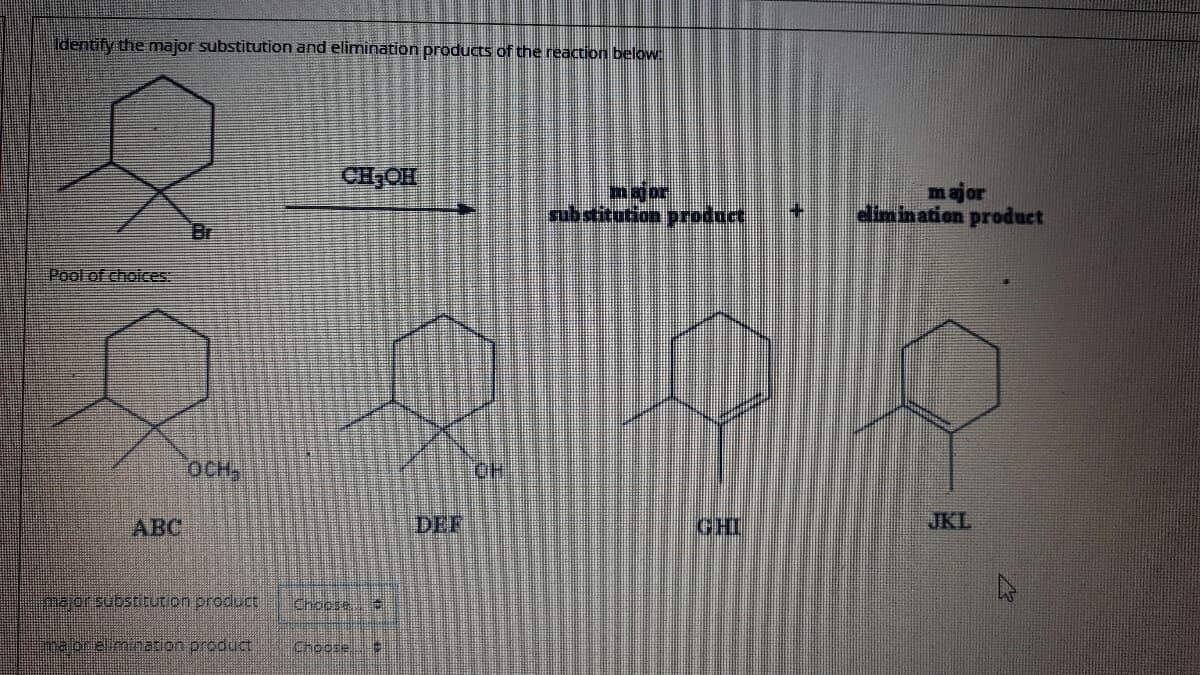
Identify the major substitution and elimination products of the reaction below: CHOH major edimin ation product pubstitution product Br Pool of choices: OCH, OH ABC DEF CHI JKL Imajonsubstitution product Chocse
Q: Which arrow-pushing mechanism best explains the bromination of the following alkene shown below?©GMU…
A: As the question given in the second image is incomplete, we will solve the question given in the…
Q: Br .S.
A: SOLUTIONS: Step 1: The above compound is a secondary alkyl halide. The secondary alkyl halide when…
Q: Select the expected major product of the reaction shown. JDHDMC но OH Lof 17 388
A:
Q: 4. Predict the major product of the following reactions (Consider regioselectivity and…
A: Nucleophiles are nucleus loving species which tend to donate electron pairs to electrophiles whereas…
Q: 6. The two pentene isomers react with mCPBA, followed by an aqueous workup with NaOH/H2O. Draw the…
A: In presence of m-CPBA, alkene is oxidised to epoxide.
Q: What is the major elimination product expected from the following reaction? Br. NaOMe MeOH Select…
A:
Q: uestion 34 Consider the mechanism for the conversion below and identify the intermediates that lead…
A: Topic- This reaction is called intramolecular aldol reaction.
Q: cise 25. Determine the mechanism of nucleophilic substitution of each reaction. Draw the products,…
A: Unimolecular nucleophilic substitution reaction, SN1 is favored in presence of a polar protic…
Q: Identify the following reactions as additions, eliminations, substitutions, or rearrangements. OH…
A: a) Elimination b) Elimination c) Substitution
Q: 5. a) When the molecule below is treated with HCI, thế curved arrow formalism to draw a mechanism…
A: As you have asked multiple question. We will answer the a) part. If you want any specific answer…
Q: 23. Treatment of alkene A with HBr gives the alkyl halide B as one of the products. Draw the…
A:
Q: Hz O heat
A: The given reaction is the nucleophilic substitution reaction. H2O is the nucleophile that will…
Q: Which of the following alkyl halides yields the product shown as the only possible product of an…
A: E2 reaction means elimination reaction of 2nd order. In such reaction, the loss of leaving group and…
Q: Predict whether the following reactions will proceed via substitution (SN1 or SN2), or elimination…
A: We have to predict type of reaction and major product.
Q: What is the product of the SN2 reaction shown below? Nat SH ? DMSO HS- HS.. SH HS b. c. d. a. Which…
A:
Q: Ans . SUl fenation oF benzene involves an electrophilic substitution mechanisem, what would Be the…
A: Solution The sulfur oxide electrophile arises in one in every of 2 ways in which looking on which…
Q: For the following reactions, determine which mechanism they will follow, explain why the…
A: From given Both possible products is drawn and from that major product is determined and finally…
Q: 17. Draw the mechanism for the following SN1 processes. OH Br OH HBr HBr Br (a) (Ь) OEt H20 ELOH (c)…
A: The mechanism of given SN1 reactions are given below -
Q: 3.2 7 Show the polarity on each haloalkane / alkyl halide. Show and name the mechanism of how the…
A:
Q: What is/are the major product(s) of the following substitution reaction? Br CH₂OH (substitution) Vam…
A: 39) Tertiary alkyl bromides undergo nucleophilic substitution reaction in SN1 reaction mechanism.In…
Q: OH H2N. OH D A В Select BOTH products of this reaction: + H2O ZI
A: It is breaking of carbon-nitrogen bond of amide and gives carboxylic acid and amine
Q: Write the expected substitution product(s) for each reaction and predict the mechanism by which each…
A: The expected substitution product has to be written. The mechanism by which each product is formed…
Q: Br (a) OH OTs (d) NBS |HOCI HO- он (g) (h) + enantiomer HO,
A: The reaction of substrate with a particular reagent leads to the formation of specific product.…
Q: Complete the following reactions by identifying the major product(s) or the missing reaction…
A: The given problems are based on the functional group inter conversions by using different reagents.
Q: 2. (a) Provide the structure of the product of the following reaction for preparation of an alcohol.…
A:
Q: Q 15.20: Predict the major product of the following reaction. AICI, II II IV A IV ID II
A: Answer:- This question is answered by using the simple concept of chemical reaction of benzene to…
Q: H14.35 - Level 2 Unanswered • 3 attempts left What is the major product formed in the following…
A:
Q: Select the expected major product of the reaction shown 1. LDA, -78 C 3-methyl-4-hexanone 2. CH3CH2I…
A: LDA is a good base, so it takes hydrogen atom from carbon, now an enolate is formed. This reaction…
Q: Select the best method to synthesize the following alkene. H2SO4 CH,O Na* В. А. heat CH3OH он Br…
A: Best method formation for alkene is witting reaction because in E1 reaction Rearrangement possible…
Q: What is the expected major product of the following reaction sequence? 1. t-BUOK, t-BUOH CI 2a. Og,…
A: Step 1 - Hoffmann elimination Elimination reaction of alkyl halide with bulky base like tertiary…
Q: Provide the missing major product, reactant, or reagent and the mechanism (e.g. SN1 or SN for each…
A: We need to provide the product, reactant and reagent for the following reactions along with suitable…
Q: In the following SN1/E1 reaction three elimination products are formed. Which El product do you…
A: E1 products goes through carbocation intermidiate. So the yield of product depends mostly on…
Q: Ph,CuLi NO 2 OMe 1. nBuLi 2. PHCHO OMe 1. nBuLi 2. PHCHO Br Br LOME | Pd(PPH3)4
A: The details solution for this reaction is provided below in attach image.
Q: Draw the coupling product of the Pd(0)-catalyzed Suzuki reaction between the compounds shown. Show…
A: coupling reactions : When two substrates are combined in the presence of a metal catalyst,…
Q: What is the major substitution product to the following reaction? H2S ll
A: The reaction taking place is given as,
Q: In the following reaction, the racemic product shown was only a minor product. An achiral product…
A:
Q: 4. Prodiet the productis) of the lollowing reactions, including stercochemistry when necessury und…
A: Nucleophilic substitution reaction
Q: (iii) Draw the organic products of the following Pd-catalysed reactions, paying particular attention…
A:
Q: :Br: NH2 A G :o:
A: Electrophilic centers are those which has electron deficiency. Nucleophilic centers are those which…
Q: Which of the following is the expected product for the Pinacol-Pinacolone rearrangement reaction: но…
A:
Q: Which mechanism does the following reaction fall into? CI 1) NaNH2, NH3 (liq) 2) H3O+ SNAr…
A: Given reaction is : Options are : a. SNAr (sometimes called addition-elimination) b. None of…
Q: B. Draw structures for the products in the following reactions. CN CN CN Jo 70⁰ 1. PrMgBr 2. H₂O* 1.…
A: PrMgBr is the nucleophile here LiAlH4 is reducing agent I2, NaOH,∆=> iodoform reagent.
Q: 23) Which reaction will yield the following alkene as only elimination product? NaOEt A) Br E-3. Br…
A:
Q: what IS the major organk Product obtaines fiom the following lealtion? A) OH B) OH C) Hz' D) PC HO-…
A: “Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: Elimination reactions of cis-and trans-1-bromo-2-methylcyclohexanes with NaOEt in E1OH can give the…
A: We have to predict the main product for the following given reactions as follows in step 2:
Q: What is the 1, 4 - addition product in the reaction equation below? A.D B.A OC.E OD.C + HC1- B C D E
A: For the formation 1,4-addition product the carbocation has to undergo resonance stability to form…
Q: dict the product of the following reactions. 1. NaOr-Bu t-BUOH 2. КОН, НССI, Draw the molecule on…
A: To find: product formed by the following reaction:
Q: T A TOpice TReferences) OH 1. H,PO, H20 MeO 80 Meo 2. HI a - Proton transfer g- SyI Nucleophilic…
A: 1) This reaction is basically elimination reaction and it is going in acidic medium. 2) This…
Q: 1. For each cof the following pairs of SNa reactions, indicate which reaction accurs Fester: a)…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: 1. Draw the products for the following base-catalyzed E2-elimination. What are their relative…
A: The reaction given are,
Step by step
Solved in 3 steps with 2 images

- 1. SN1 or SN2 2. Draw major product withs steorchemistry 3. include reaction mechanismI need an explanation on the mechanism, linked bellow 1 paragraph explanation on the steps and mechanism. and what the final product is calledExplain how and why rearrangements occurduring Friedel-Crafts alkylation reactions formingmore than 1 product. Also illustrate therearrangement reaction from the aboveexample.
- Plastic photochromic sunglasses are based on the following reversible rearrangement of a dye inside the lenses that occurs when the lenses are exposed to sunlight. The original dye absorbs UV light but not visible light and is thus colorless, while the rearrangement product absorbs visible light and is thus darkened. (a) Show the mechanism of the rearrangement. (b) Why does the rearrangement product absorb at a longer wavelength (visible light) than the original dye (UV)?Predict the product(s) and provide the complete mechanism for each reaction below.There are two isomeric cyclohexa-1,4-diene products when benzoic acid undergoes the Birch reduction (see Problem 25.24). (a) Draw the mechanism that leads tothe formation of the major product. (b) Will the Birchreduction of benzoic acid occur faster or slower than theBirch reduction of benzene itself? Hint: Is –CO2H anelectron-donating or an electron-withdrawing group?
- Draw the mechanism ffrom benzaldehyde to this using: i)NaBH4 ii)TsCl, py iii)NaCN iiii)H+, H2OIn addition to organic halides, alkyl tosylates (R'OTs. Section 9.13) also react with organocuprates (R2CuLi) to form coupling products R – R'. When 2° alkyl tosylatee are used as starting materials (R2CHOTs), inversion of the configuration at a stereogenic center results. Keeping this in mind, draw the product formed when each compound is treated with (CH3)2CuLi.1. SN1 or SN2 2. Include major product and stereochemistry 3. Add detailed reaction mechanisms
- Provide the curved arrow mechanism for the reaction of the compound shown below treated with Cl2/FeCl3. Include transition states and the major product. I apprecitate the helpIn addition to organic halides, alkyl tosylates (R'OTs. Section 9.13) also react with organocuprates (R2CuLi) to form coupling products R – R'. When 2° alkyl tosylatee are used as starting materials (R2CHOTs), inversion of the conguration at a stereogenic center results. Keeping this in mind, draw the product formed when each compound is treated with (CH3)2CuLi.1.Draw the structures of all 3 resonance hybrid forms of the benzenonium intermediatefor both o and p attack for the case of activating groups an indicate which one in eachcase is the most stable form.Draw the structure of all 3 resonance hybrid forms of the benzenonium intermediatefor m attack in the case involving a deactivating group. 2.Halogens are deactivating groups but direct electrophilic attack at the o and ppositions. Why is this the case?

