Q: Practice Exercise Give the IUPAC names of the following structures: a. CH,-CH=C-CH,-CH, C,H, C,H; CH...
A:
Q: What is the work which can be accomplished by the following reaction: (s) + 2K - 2 K* + 21 Calculate...
A: The reaction given is, => I2 (s) + 2 K → 2 K+ + 2 I-
Q: Predict the major product of each of the following reactions. Show the stereochemistry of the produc...
A: 1. Sn1 reaction occurs via carbocation formation and then nucleophilic attack is observed. 2. In pr...
Q: The ground state election configuration for the Gallium (lll) ion is
A:
Q: The Arrhenius equation is k = Ae-Ea/RT The slope of a plot of In k vs. 1/T is equal to -Ea/R Ok Ea O...
A:
Q: (a) 2 Mg(s) + O2(g) → 2 MgO(s) kJ (b) 2 SO2(g) + 02(g) → 2 SO3(g) -139.9: kJ (c) 2 C,H2(g) + 5 02(g)...
A:
Q: 12. Lightly scented oils, such as jojoba oil, can be used as carrier oils for strongly fragrant esse...
A:
Q: A generic Lewis structure is given where Y represents the central atom and the outer atoms are repre...
A: We have to predict the correct compound for given structure.
Q: Silicic Acid structure is this: The pK,s are: 9.83 and 13.1 он но - si- он OH Draw each acid reactio...
A: a.) Given compound is a weak acid with high values of pKa. To write acid reactions , we show it ion...
Q: ed Total Equation (molecular equation) oC2(aq) + AGNO3(aq) → of reactants and products (write below ...
A:
Q: Sodium chloride (chemical formula NaCI) has different properties than the elements (Na & CI) that it...
A: Concept: The physical properties of an compound are often quite different to the physical properties...
Q: Refer to the (unbalanced) equation CS2+ CaO –→ CO2 + CaS. How many grams of CaS are produced if 53 g...
A:
Q: a 0.55 M solution of a monobasic compound B has a pH of 11.32, what is the Kb value of the base B
A:
Q: net ionic equation
A:
Q: For two reactions, whose entropies of activation are equal, the activation energies are 50.00 and 60...
A:
Q: Titrations are generally both more accurate and more precise the smaller the concentration of titran...
A:
Q: E H;PO2 HNO3 Sn, HCI A NANO2 B CUCN D H,SO4 HCl
A: Nitration of benzene is aromatic electrophilic substitution reaction where benzene acts as nucleophi...
Q: What ratio do we need to prepare a solution with pH = 4.15 using formic acid and its conjugate base,...
A:
Q: Fill up the empty boxes with the correct chemical structures. CN OH Ph
A:
Q: minimum number of steps; from one to six are required. List reagents by letter in the order that the...
A: The first conversion involves formation of olefin from a carboxylic moiety and the second conversion...
Q: The ?a of a weak monoprotic acid is2.72×10−5. What is the pH of a 0.0751 M solution of this acid?
A:
Q: Calculate the H3O* ion concentration in an aqueous solution with a pH of 2.24 at 25°C. Report your a...
A:
Q: Five grams of vinegar solution was given to you to analyze for the percent acetic acid present in th...
A: To solve this problem we have to calculate the percentage by mass of acetic acid.
Q: Two (2) grams of vinegar solution was given to you to analyze for the percent acetic acid present in...
A:
Q: Consider an analyte solution of 50.0mL of 0.050M hydrochloric acid, HCl, titrated against 0.10 M so...
A: “Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for y...
Q: Draw structures for the carbonyl electrophile and enolate nucleophile that react to give the enone b...
A: Aldol condensation: The alpha hydrogen of carbonyl compounds is acidic in nature and it is removed a...
Q: Predict the sign of the entropy change, AS°, for each of the reaction displayed. Drag the appropriat...
A:
Q: How would I draw C9H19NO with this Hmnr data 4.6(s,2H) 3.3(s,3H) 2.2(q,2H) 1.6 (s,9H) 1.3 (t,3H)
A: First we have to determine the total number of Double Bond Equivalent of the given molecular formula...
Q: Part B If the volume of the original sample in Part A(P = 412 torr , V1 = 10.0 L ) changes to 64.0 L...
A: Given, Initially Pressure (P1) = 412 torr Volume (V1) = 10.0 L Later Pressure (P2) = ? Volume (V2) ...
Q: Consider the substitution reaction shown below. By what mechanism will the reaction proceed? Br HOCH...
A: There occur substitution reaction.
Q: pH
A:
Q: Assign the correct stereochemical configurations to each of the chiral centers in 1-bromo-2-chlorocy...
A: Assign stereochemistry of the given structure--
Q: Emission lines from the PASCHEN SERIES would appear in the __________________ region of the electrom...
A: In this question, Emission lines from the Paschen series would appear in the __________________ regi...
Q: what is the antimicrobial compound that is found in table salt?
A: Answer - According to the question - Antimicrobial compound - An antimicrobial agent is defined as ...
Q: wavelength
A:
Q: write five reaction of aldol by chemical e quation ?
A: Aldol reaction: Aldol is an word use for aldehyde and alcohol. When the enolate of an aldehyde or a ...
Q: Which of the following phosphate salts are insoluble in water? A) (NH4)3PO4 B) Li¿PO4 C) Na3PO4 D) K...
A: Which of the following phosphate salts are insoluble in water.
Q: What is the equilibrium constant (K) at 25°C for the following cell reaction? Co(s) +Ni2+→Ni(s)+ Co...
A:
Q: Question 4 If a reaction has a AG° of -16.0 kJ/mol, will that reaction favor the starting materials ...
A: 4) Case- I When ∆G° < 0 [∆G° = (-)], reaction is spontaneous, (products are more stable than reac...
Q: temperature? What mass of solute will dissolve in 100 g water at the following temperatures? a. NaNO...
A: 6.) To answer these values we would check the curve of given solute and temperature, then look at y...
Q: () Complete the following reactions. Show the stereochemistry where applicable. F - Na CN Aceton (le...
A: Since there are more than 3 question in the given problem so as per the Bartley rules and regulation...
Q: (molecular equation)
A:
Q: In a combustion reaction, the most common fuel that burns contains compounds called hydrocarbons. Th...
A: In this question, combustion of methane is represented by the equation: CH4 + 2O2 → CO2 + 2H2O 1. In...
Q: Current Attempt in Progress Chargers for cell phones get warm while they are being used. Some of the...
A: Concept: Total energy required for charging= (energy wasted as heat + energy required to charge the...
Q: Calculate the pH of the following: - 0.10 M alanine hydrochloride
A:
Q: Use the References to access important values if needed for this question. Draw the structure of the...
A:
Q: CH30
A:
Q: Calculate the amount of heat (in kJ) required to raise the temperature of a 86.0 g sample of ethanol...
A: Heat is required when heating of substance is observed. It can be determined using the following for...
Q: What is the oxidation number (ON) of Cl in the following compound? Cl2O5 What is the oxidation num...
A: Given :- Chemical formula of compounds : Cl2O5 N2O7 To find :- oxidation no. of Cl in Cl2O5 an...
Q: Lithium metal reacts with nitrogen gas to form lithium nitride. Identify the balanced reaction that ...
A:
Step by step
Solved in 2 steps with 2 images

- Suppose a glass of wine is exposed in air, it tends to become sour due to reduction of C2H5O formation of C2H5NH2 dissolution of CO2 oxidation of C2H5OHwhat type of reaction occurs when sugar (C12H22O11) is heated? The (unbalanced) reaction again is C12H22O11 ----> C + H2O A) Combustion B) DecompositionIdentify the reactant that gets oxidized in the following reaction. 2CIO3- (aq) + C2O42- (aq) + 2H2O(l) → 2ClO2(g) + 2CO2(g) + 4OH- (aq)
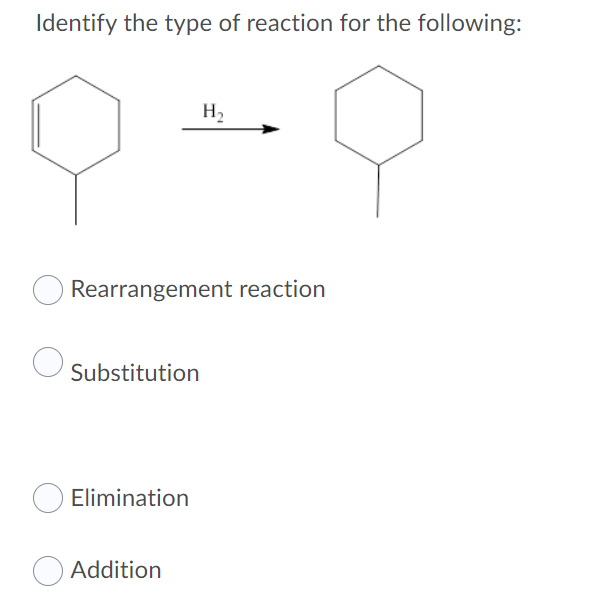
- Which of the following is a concerted reaction with a ΔG < 0? (Hint: Exergonic reaction)Identify the reactant that gets oxidized in the following reaction 5C2O4^2- (aq)+ 2MnO4^-(aq)+16H+(aq)---> 2Mn^2+(aq) +8H2O(l) +10CO2(g)Determine the organic reaction involved in the following mechanisms: a. oxidation b. substitution c. elimination d. addition
- 22. Complex compounds that have high activation energy are complex compounds that are... A. inertB. is not stableC. stableD. labileName the two groups into which phenomenon of catalysis can be divided. Give an example of each group with the chemical equation involved.Consider the reaction in producing wine: C6H12O6(aq)——-> 2CO2(aq). When zymase is added to glucose, the production of ethanol and carbon dioxide increases in a shorter period of time. What is the role of zymase in this reaction? a. Reactant b. Enthalpy c. Catalyst d. Product Please show your solution.
- Identify the strongest reducing agent from the given reaction: Choose among a.H2O2 b.NADH c.NAD+ d.O2 Given reaction: oxaloacetate + 2H+ +2e- ↔ malate Eo=+0.330 V NAD+ + H+ + 2 e- ↔ NADH Eo = - 0.105 V O2(g) + 2H+ + 2e- ↔ H2O2 Eo = +0.695 Vdissociation of H2CO3 to H20 and CO23-phosphoglycerate is converted to 2-phosphoglycerate by phosphoglycerate mutase within a cell (see reaction equation below). The delta G’^o of this reaction is -4.44 kJ/mole. If within the cell the concentration of 3-phosphoglycerate is 2 mM and the concentration of 2-phosphoglycerate is 100 mM, the actual delta G of this reaction at 25 °C is ___________________ kJ/mol. (Please use 3 significant figures in answer) Reaction: 3-phosphoglycerate to 2-phosphoglycerate (R = 8.315 J/mol · K)

