If you will measure 90µl of liquid, what pipet calibration should you use appropriately? O 10µl - 100µl O 2ul - 10µl O 100ul - 1000µl O 100ul - 200µl
If you will measure 90µl of liquid, what pipet calibration should you use appropriately? O 10µl - 100µl O 2ul - 10µl O 100ul - 1000µl O 100ul - 200µl
Chapter8: Sampling, Standardization, And Calibration
Section: Chapter Questions
Problem 8.1QAP
Related questions
Question
100%
Q2 and q5

Transcribed Image Text:If you will measure 90µl of liquid, what pipet calibration should you use
appropriately?
10µl - 100µl
2ul - 10ul
O 100µl - 1000ul
100ul - 200pl
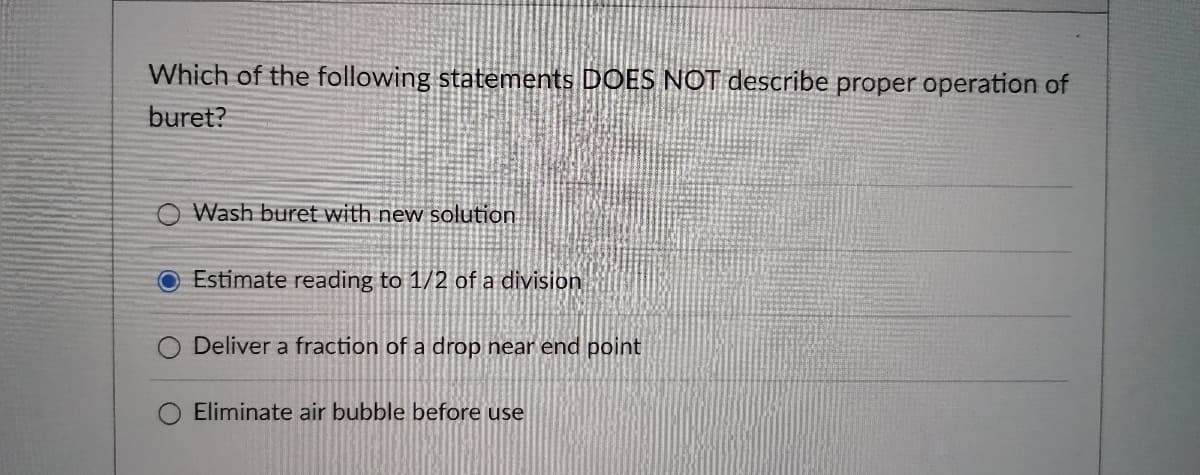
Transcribed Image Text:Which of the following statements DOES NOT describe proper operation of
buret?
O Wash buret with new solution
Estimate reading to 1/2 of a division
Deliver a fraction of a drop near end point
Eliminate air bubble before use
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 5 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

