Q: The Gouy balance is working by making use of the interaction between the magnetic fiel and
A: A Gouy balance is used to determine the magnetic susceptibility of a substance. A strong magnet is…
Q: Initial Initial Rate of oncentration Formation ofZ mmol/litre.s of Y 0.20 M 0.06 0.40 M 0.24 0.40 M…
A: General form for rate law is : Rate = k [X]m [Y]n
Q: THE SPECIFIC POWER BACTERICIDAL LAMP CLOSED TYPE, SHIELDED BE LESS THAN (W / M ³) 1. 1 2. 2-2,5 3.4…
A: The correct answer is given below
Q: E) LOCH3 1. AlCl3 2. H20
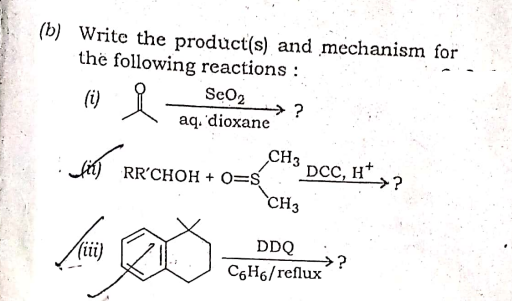
A: This is an example of friedel Craft acylation reaction in which anhydride react with lewis acid .
Q: Good hand written answer Asap Just mechanism
A: see below
Q: show-all-working-explaining-detailly-each-step
A: Fe+3 will get reduced because Fe+3 has high reduction potential while Cu will oxidised because…
Q: In case of SEC method, the proteins’ adsorption could be a result of overall ionic interactions…
A: In the size exclusion chromatography (SEC), the factor the that differentiates one type of molecule…
Q: show-all-working-explaining-detailly-each-step Answer should be typewritten using a computer…
A: To find: The monomer from which the polyester Terylene to be produced
Q: plied with a certain force, reverts to its original form. With stress given at 3.0 GPa and strain he…
A: Given data set: Stress = 3.0 GPa Strain = 0.230 We need to determine the Young's modulus of the rod.…
Q: show-all-working-explaining-detailly-each-step Answer should be typewritten using a computer…
A: Raoult's law: If the vapour pressure of the mixture is lower than as expected from the Raoult's law…
Q: show-all-working-explaining-detailly-each-step. Answer should be typewritten using a computer…
A: BaCl2 gives apple green color in flame test. C option is correct. BaCl2 The reaction of barium…
Q: Please answer with full mechanism ASAP
A: Given compound has ether functional group, to synthesize ether alcohol precursor is needed…
Q: problem
A:
Q: liquid-liquid extraction(LLE)Principles
A: Liquid–liquid extraction (LLE), also known as solvent extraction and partitioning, is a method to…
Q: show-all-working-explaining-detailly-each-step
A: In industrial area, we need to prepared high amount of yield of sulfuric acid ( H2SO4 ) in short…
Q: (Drag and drop into the appropriate area) Most 0.060 M H2SO3 (diprotic) 0.075 M HNO2 (monoprotic)…
A: In this determine the concentration of H+ ions from each acid. More is the concentration of H+ more…
Q: show-all-working-explaining-detailly-each-step
A: When sodium ( Na ) metal react with water then it's produces Sodium hydroxide by releasing of…
Q: A cylindrical metal specimen having an original diameter of 12.8 mm and gauge length of 50.80 mm is…
A:
Q: show-all-working-explaining-detailly-each-step
A: Diagonal relationship of Be can be shown that element which have similar properties of Be.
Q: The cool plasma tail needs to be removed in regular ICP-OES analysis using the axial view. Provide…
A: A question about spectroscopy, which is to be accomplished.
Q: Pls help ASAP. Pls show all work.
A: The Hess's law states that the enthalpy change in a chemical process is equal to the sum of the…
Q: See problem below Alfred was tasked to synthesize at least 250.0 ppm of his novel gold…
A: Given the absorbance of the blank solution = 0.200 First, we have to find the concentration (in…
Q: Melting point: 122-123 experimental product melting point: 120 mass of starting material: 1.04…
A: A numerical problem based on yield, which is to be accomplished.
Q: 1) LIAIH, ? 2) HO
A: Lithium aluminium hydride LiAlH4 is an hydride based reducing agent which reduces the given ester to…
Q: Polarographic cells are used to measure the partial pressure of gases only in static gas monitoring…
A: Please see explanation
Q: The solvent in a collodial system is also known as the [?] phase. dispersion dispersed particle…
A: Colloidal system is heterogeneous and contains two phases : dispersed phase and dispersion medium.
Q: Show-all-working-explaining-detailly-each-step. Answer should be typewritten using a computer…
A: A option is correct. Ethanamide Different carboxylic acid derivatives have very different…
Q: О (3 Can you please provide name and solve? Please apply arrow please & thank you 0 О :) Na ome Me…
A:
Q: Data for undiluted bleach solution: [NaOCI] used in trial (after addition of dye) 25.0mL Which graph…
A: Chemical kinetics is the branch of chemistry which deals with study of the rate of reaction. Rate of…
Q: C + Lo |||
A: 1.Ethyne is alkylated using NaNH2 and methyl iodide. Methyl acetylene is subjected to hydroboration…
Q: Please assist with sub part C.
A: As already calculated form part (a) and (b), the density of metal is 1.8 g/mL. Now, this density has…
Q: Answer with mechanism ASAP
A: see below
Q: show-all-working-explaining-detailly-each-step Answer should be
A: Option C is correct answer. pH = 0.5
Q: Show the mechanism and steps. Show all the work.
A:
Q: show-all-working-explaining-detailly-each-step Answer should be typewritten with a computer…
A: D Option is Correct. V is the alkali metal.
Q: please correct this asap
A:
Q: show-all-working-explaining-detailly-each-step. Answer should be typewritten using a computer…
A: Option C is Correct Answer. I.e., electric field in mass spectrometer is to deviate the charges.
Q: show-all-working-explaining-detailly-each-step
A: Catalyst is added to increase the rate of reaction.
Q: Question: How do I get the calibration curve from this data?
A: The dissolution of the solute in a suitable solvent produces the solution. If the amount of solute…
Q: 1. OsO, ? 2. HIO,
A: Given a compound of benzene Alkene and ketone group present. That's a reaction of compound is…
Q: Write three main methods which are included in this process.
A: Chemical reaction is a process in which two or more reactants combine to give a product.
Q: has a bulk density of 1.1 g/cm3. The soil core is placed in a p iratod The ov on drv soil and watcr…
A: According to the question, a 500 cm3 oven dry core has a bulk density of 1.1 g/cm3. This oven dry…
Q: Why are some mechanism considered as exact straight mechanism?
A: the solution is shown below.
Q: ETHANOL Sp.Gr.- 0.791 A cylindrical unknown solid material with a diameter of 10cm and a height of…
A: Given: Height of unknown cylinder = 30 cm = 0.3 m Diameter of unknown cylinder = 10 cm = 0.1 m We…
Q: There's the information to go off please show work and if more than onw problem is done label so I…
A: Given:Mass of NaCl + dish after second drying =37.323 g Mass of evaporating dish=35.452 g Mass of…
Q: Show-all-working-explaining-detailly-each-step
A: When two liquid Compound ( which have strongest hydrogen bonding ) are mixed to each other then…
Q: The viscosity of methane at 20 °C and 1 atm pressure is 1.09x10-4 Calculate the average collision…
A: Please open the attachment.
Q: 3.Write in detail about working of any one hydraulic system application in construction area with…
A: Hydraulic lifts are very important equipment used at the construction site, Especially construction…
Q: Show the arrow push mechanism or the electron flow mechanism
A: We have given the organic reaction and we have to Show the arrow push mechanism or the electron flow…
Step by step
Solved in 2 steps with 1 images

- Acid-catalyzed dehydration of neopentyl alcohol, (CH3)3CCH2OH, yields 2-methyl-2- butene as the major product. Outline a mechanism showing all steps in its formation.(ii) The elimination reaction between 2-bromobutane and NaOCH2CH3 gives two organic products. Draw a mechanism for the reaction which produces the major organic elimination product and provide a rationale as to why that is the major product.Explain and detail the reaction mechanism: NaBr + H2O + n-butyl alcohol = ?
- For the base-catalysed hydrolysis of 3-bromo-3-methylhexane (i.e. reaction with the nucleophile OH-): For the reaction intermediate, draw its structure and give the VSEPR description of the geometry at the reaction centre Give the names and draw the structures of the two reaction products. Explain your conclusions.(ii) The elimination reaction between 2-bromobutane and NaOCH2CH3 gives two organic products. Draw a mechanism for the reaction which produces the major organic elimination product and provide a rationale as to why that is the major product.(with explanation)Draw the reaction mechanism of conversion of compound 4a to 4b. Indicate what type of reaction mechanism. Write conditions and reagents used if there are any. Provide brief background in every step of the reaction mechanism.
- A) Provide the reagent and reaction mechanism to show how the reactants and products in the following reaction can interconvert B) under what conditions would the reaction I) favour reactants, II) favour the products and III) why?(i) Name, draw and describe the organic product of the reaction between 2-methylbut-1-ene and H2O in the presence of H2SO4 and provide a clear rationale as to why this is the major product of the reaction. (ii) The elimination reaction between 2-bromobutane and NaOCH2CH3 gives two organic products. Draw a mechanism for the reaction which produces the major organic elimination product and provide a rationale as to why that is the major product.The rate law for addition of Br2 to an alkene is first orderin Br2 and first order in the alkene. Does this informationsuggest that the mechanism of addition of Br2 to analkene proceeds in the same manner as for addition of HBr?Explain.
- One possible way of determining the identity of an alkene, is to let itundergo an oxidative cleavage reaction in the presence of hot basicpotassium permanganate. You are given two containers said to containdifferent alkenes. Container A is marked as cis / trans‐2‐butene andcontainer B as 2‐methyl‐1‐butene. Explain by referring to the formation ofproducts, how you would verify the identity of the alkenes.When A is reacted with hot aqueous NaOH, a compound B of molecular formula C8H11NO is produced. With this information, write the correct structure of B and propose the reaction mechanism (step by step, with the correct use of arrows) to obtain B.In the reactions given below, write to which organic molecule Y, Z, T, A ‘belongs, and give information about the reaction types that take place.



