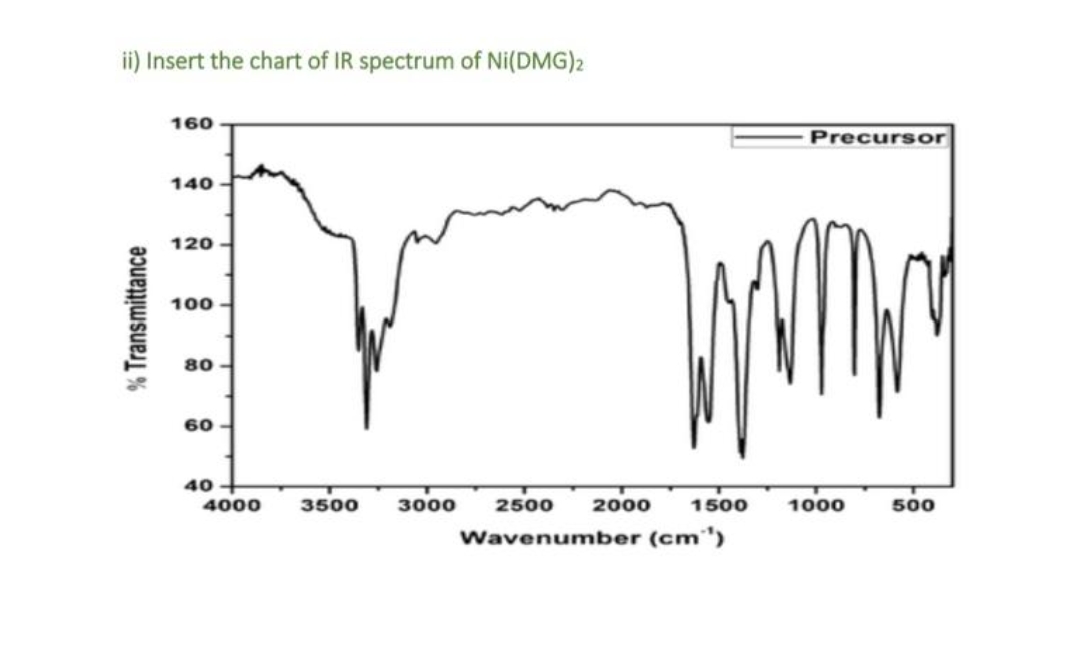
ii) Insert the chart of IR spectrum of Ni(DMG)2 160 Precursor 140 120 100 80 60 40 4000 3500 3000 2500 2000 1500 1000 500 Wavenumber (cm") % Transmittance
Q: Iron (III) can be separated from other metals, such as chromium, by extraction into ether from a str...
A: The ratio of concentrations of a solute (here it is iron) distributed between two phases( organic an...
Q: is solution is prepared by mixing 25 mL of 0.1 M HCHO2 with 25 mL of .2 M NaCHO2. The pH of the solu...
A: Given values-> Volume of HCHO2 = 25ml Molarity of HCHO2 = 0.1 M Volume of NaCHO2 = 25 ml Molarity...
Q: Determine the heat associated with a decrease in the temperature of 375g of water from 31.5°C to 24....
A: Heat associated with decreasing temperature is calculated using the equation, ...
Q: Question 4 - - For the following two compounds, the given names do not correctly follow IUPAC naming...
A:
Q: A chemist determines by measurements that 0.095 moles of bromine liquid participate in a chemical re...
A:
Q: For the following compound, the given name is not correctly following the IUPAC naming conventions. ...
A: IUPAC NOMENCLATURE RULES :
Q: Calculate the Equillibrium constant at 25 Degrees Celsius for the following Reaction: From the data ...
A: We have to find equilibrium constant from the given data. Temperature = 25°C or 298 K
Q: Ammonia (NH,) chemically reacts with oxygen gas (o,) to produce nitric oxide (NO) and water (H,0). W...
A: Given data is as follows: The mass of ammonia given = 1.2 g The mass of oxygen gas consumed = ?
Q: Aqueous sulfuric acid (H, SO,) reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium s...
A:
Q: Based on the information from ppt file “add 6M HCl dropwise until the evolution of H2 gas stops and ...
A: According to information given in the question, measurement are taken on the basis of evalation of H...
Q: Calculate the volume in liters of a 3.20 M KCl solution to obtain 0.25 mol of KCl. Express your answ...
A: given, the molarity of the solution = 3.20 M moles of KCl = 0.25 mole
Q: Draw the crystal-field splitting energy (CFSE) of the following complexes: a. [Cr(NH3)6]3* b. [MnF6]...
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for you...
Q: Gaseous ethane (CH,CH,) reacts with gaseous oxygen gas (0,) to produce gaseous carbon dioxide (CO,) ...
A: Given data, mass of C2H6=0.60 g , molar mass=30 g mol-1 mass of oxygen=0.99 g, molar mass= 32 g mol-...
Q: What is the name of the sugar shown here? OH OH O L-xylose O D-xylose O D-erythrose O L-erythrose O ...
A: To identify the name for given Sugar.
Q: SUMMA ASS: Phosphorous trichloride and phosphorous pentachloride equilibrate in the presence of mole...
A: Given , Reaction : PCl3(g) + Cl2(g) → PCl5(g). The equilibrium concentration : PPCl3=0.202 atmPCl2=...
Q: HF is a weak acid and NH3 is a weak base. Which of the following shows the correct order of increasi...
A: The acid-base nature of the salt solution depends on the nature of the acid and base from which the ...
Q: A mixture of gaseous reactants is put into a cylinder, where a chemical reaction turns them into gas...
A:
Q: e the percent yield of se
A:
Q: (ii) Calculate the enthalpy change for the dissolution of (NH4)2SO4(s) using the enthalpy of formati...
A:
Q: NH2 CH3 C-OH NH Br Bromoaniline Acetanilide Benzoic acid Now you've received a mixture contains thre...
A: Practical organic chemistry:
Q: An unknown compound has the following chemical formula: PAS where x stands for a whole number. Measu...
A: Molecular formula: In a molecular formula we describe the number of atoms of all the elements presen...
Q: For the following two compounds, the given names do not correctly follow IUPAC naming conventions. F...
A:
Q: concentration of the H
A:
Q: (CH,(CH.),CH;) CH; reacts with gaseous oxygen gas (0,) to produce gaseous carbon dioxide (Co,) and g...
A: We first need to determine the limiting reagent. After determining the limiting reagent, we can calc...
Q: Find the apparent Km and apparent Vmax for the enzyme reaction containing the inhibitor and identify...
A:
Q: This question is based on the five compounds (A-E) given below. (A) CH;-CH=CH, (B) CH;-CHBr-CH2-CH; ...
A:
Q: Determine the average and standard deviation for the following set of numbers. 45.6g, 52.1g, and...
A: Solution - According to the question - Given number - 45.6g, 52.1g, and 53.8g Average = (45.6 + 52...
Q: Gaseous butane (CH;(CH,),CH,) reacts with gaseous oxygen gas (0,) to produce gaseous carbon dioxide ...
A:
Q: Why do you feel thirsty after playing or working outside? Sometimes, you suffer from cramps or heada...
A:
Q: A virus has a mass of 9.0 x 10 -12 mg and an oil tanker has a mass of 3.0 x 10' kg. Use this informa...
A: We have 1 mole of any substance contain Avogadro number6.022×1023 of atoms. Hence 1 mole of a viruse...
Q: Name types of gels (sieving matrix) that may be used for gel filtration chromatography and discuss t...
A: The types of gels for gel filtration chromatography include, Dextran Polyacrylamide beads/ agarose ...
Q: Draw the structure of the following complexes: a. [Co(en)2(NH3)CI]2+ b. dichlorobis(ethylenediamine)...
A:
Q: What would happen if the solute (salt or sugar) concentration exceeds that of the solvent’s? Would i...
A: When the solute concentration exceeds to that of solvent then the solute particles become more in nu...
Q: Is there a difference when the needle or metal clip is placed on the surface of distilled water and ...
A: Yes, there is a difference when the needle or metal clip is placed on the surface of distilled water...
Q: Calculate ∆H for the reaction 4NH3 (g) + 5O2 (g) → 4NO (g) + 6H2O (g), from the following data. 1. N...
A: 4NH3 (g) + 5O2 (g) → 4NO (g) + 6H2O (g) 1. N2 (g) + O2 (g) → 2NO (g) , ∆H = -180.5 kJ 2. N2 (g) + 3H...
Q: The decomposition of phosphorus pentachloride (PCI5) into phosphorus trichloride (PCI3) and chlorine...
A:
Q: A mixture of nitrogen and argon gas is expanded from a volume of 69.0 L to a volume of 89.0 L, while...
A: Given data, Pressure P = 55.0 atm Initial volume = 69.0 L Final volume = 89.0 L work done = ?
Q: rite the net chemical equation for
A:
Q: If K, for HCN is 6.2 x 10-10, what is K, for CN ? O 6.2 x 104 O 6.2 x 10-10 O none of these O 6.2 x ...
A:
Q: Consider the following balanced chemical equation: H2O2 (aq) + 3 r (aq) + 2 H (aq) (aq) + 2 H,O (I) ...
A:
Q: Consider these three reactions and equilibrium constants. Assume all components are gases. 1. C2 20 ...
A:
Q: Which of the following is the most basic solution (highest [OH ])? O pOH = 1.0 O pH = 12 O [OH¯] = 0...
A:
Q: ÇI CI -CH3 +2Zn 2ZnCl2 + Č CI Cao +NazCO3 + CH;CHCOONA + NaOH CH3 d.e. 2 CH3CHBR + 2Na 2NaBr + CH3
A: Given,
Q: 1. Write the structural formula of the major product(s) formed in each of the following reactions. S...
A:
Q: The decomposition of phosphene (COCI,) gas into carbon monoxide (CO) and chlorine (Cl2) gases is ver...
A: Equilibrium constant ( Kp ) is the ratio of partial pressures of products to partial pressures of re...
Q: ACTIVITY (solution stoichiometry) 1. H2SO̟ reacts with NaOH, producing water and sodium sulfate. Wha...
A: Since you have asked multiple question, we will solve the first question for you.If you want any spe...
Q: Draw the products of each reaction
A:
Q: In an aqueous solution of potassium chloride, the solute is (1) Cl-only (2) KCI (3) K+ only (4) H20
A:
Q: It is emphasized in the procedure that the water bath should be continuously stirred while heating a...
A: Heating or cooling bath allows to set temperatures above and below room temperatures. Temperature-ba...
Q: Illustrate with equation the preparation of alkanes by Wurtz synthesis.
A:
Step by step
Solved in 2 steps with 2 images

- What percent transmittance range and what absorbance range are considered to be the optimum working ranges for spectrochemical measurements?Peaks of scattered intensity are observed when the path difference would becomes equal to __________ λ/2 λ 2λ 3λ/2State the difference between transmittance, absorbance, and molar absorptivity. Which one is proportional to concentration?
- Write a short note about IR spectroscopy instrumentation (source)? Please answer at your own words(to the pount).An absorption measurement with a 1 cm path length yields a reading of 0.004 absorbance units with a noise of 0.001 absorbance units and a mean noise reading of 0 for 10 scans averaged together. Assuming the noise for a single scan does not change if the path length is increased to 6 cm and the number of scans is increase to 100, what is the signal to noise of the new measurement.Explain why the information from an XPS chemical shift must also be contained in an X-ray absorption edge.
- Why do you have to use the same cuvette after doing the baseline correction in Ultraviolet-Visible Spectrophotometry? (Minimum of 8 sentences)A Raman spectrum of undoped GaAs at room temperature is performed with a 514.5 nm laser. The spectrum of scattered light exhibits peaks at wavelengths of 507.1 nm and 591.6 nm due to interaction of the incident light with LO phonons. Calculate the LO phonon frequency and the ratio of the intensity of scattered light at 507.1 nm to that at 591.6 nm.Draw and label a schematic of an ICP-OES spectrophotometer. Briefly (point form is acceptable) explain the purpose or function of each component
- the transmittance concentration pairs could have been used to determine a calibration curve. state at least one important problem associated with such a calibration curveThe following are the reasons why instrumental analysis methods are used, except for: a. Narrow concentration ranges may be detected. b. Extreme low sensitivity, with molecule and noise signal detection. c. Separate equipment or facilities for control, data handling, and optimization procedures. d. All of the aboveCalculate % transmittance if 85% of light was absorbed by an analyte sample in an UV-Vis spetrophotometric experiment

