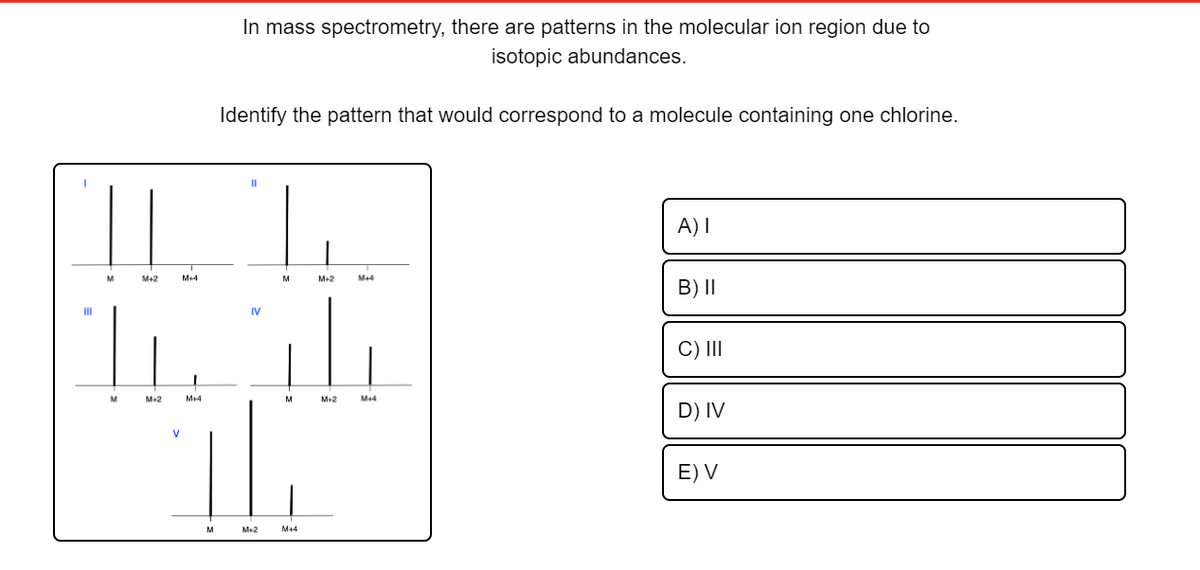
In mass spectrometry, there are patterns in the molecular ion region due to isotopic abundances. Identify the pattern that would correspond to a molecule containing one chlorine.
Q: Explain how a mass spectrometer works. What kind of information can be determined from a mass…
A: Mass spectrum was obtained from mass spectrometer. Mass to charge ratio of positively charged atoms…
Q: After reading the mass spectrometer we have found that the substance under investigation contains…
A: The percentage of the different elements for the given compound is as follows: The percentage of…
Q: Upon burning Mg in air, magnesium oxide and magnesium nitride are formed. The correct statement…
A: In this question, we will see the correct statement for Magnisium nitride. You can see details…
Q: Upon burning Mg in air, magnesium oxide and magnesium nitride are formed. The correct statement…
A: Here, it is given that, upon burning magnesium in the air, magnesium oxide and magnesium nitride are…
Q: What are the labels on the axes of a mass spectrum?
A: Solution: We know a typical mass spectrum is a plot of relative abundance (%) vs m/z.
Q: Calculate the molecular (formula) mass of each compound:(a) dinitrogen pentaoxide; (b) lead(II)…
A: The molecular formula is known as the chemical formula that specifies the total number of atoms…
Q: Calculate the molecular or formula mass of each of the following:(a) P4(b) H2O(c) Ca(NO3)2(d)…
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and…
Q: A compound contained 4.6g Na, 2.8g N, 9.6g O. Find the empirical formula. (RAM data Na = 22.99, N =…
A: In empirical formula problem first we determine mole ratio of given atom
Q: Determine the number of atoms of C in 12.1g of C12H22O11?
A:
Q: The mass spectrum of an organic compound shows the relative abundances of MM to be 48.30%48.30% and…
A: Mass spectrometry is analytical that is used to determine the mass-to-charge ratio. The mass to…
Q: What is the mass of the molecular ion formed from compounds formed from having molecular formula:…
A: A molecular ion (a cation) is formed on removing a single electron from a molecule using a high…
Q: Calculate the empirical formula of each substance from their spectrograms. 2) naproxen ( The drug:…
A: Given : Naproxen molecule which contains only H, C and O. Mass percent of C = 73.03 % Mass percent…
Q: What is the mass percent composition of platinum in the carboplatin, C6H12N2O4Pt? Relative atomic…
A: Introduction : The percent composition gives the mass percentage of each chemical element in the…
Q: Describe the type and nature of the bonding that occurs between reactive metals and nonmetals.
A: The nature of the bond present between two elements depends on the difference in the…
Q: TRUE or FALSE Molecular mass is the sum of the atomic masses in a molecule.
A: Molecular mass gives the mass of the molecule relative to that of 12C atom which has a mass equal to…
Q: The mass spectrum of an organic compound shows the relative abundances of M to be 87.83% and M+ 1 to…
A: Relative abundance/intensity of M+1 peak = 11.40 % Relative abundance/intensity of M peak = 87.83 %…
Q: Calculate the average atomic mass of Amazium, a newly discovered element (Amazium, symbol Az) which…
A: Given :- There are only 2 naturally occurring isotopes of Amazium. Mass of isotope Az-230 = 229.995…
Q: In mass analyzer ions are separated by mass to charge ratio why?
A: In a mass analyzer, ions are deflected from a straight line path and attracted to the negative side…
Q: Mass Spectrum for Tin (Sn) 120 100 100 80 58 60 40 20 0. 115 116 117 118 119 120 121 mass (amu)…
A: Welcome to bartleby ! We have to calculate average weight of Sn
Q: What is the molecular weight of a sample of ferrous sulfide heptahydrate (FeS-7H20)? (Round all…
A: Molecular weight of a compound is the sum of its individual atoms' malar mass.
Q: Sketch a simple schematic diagram of a mass spectrometer. Briefly explain how mass spectrometers…
A:
Q: Calculate (to the nearest 0.1 u) the formula mass of these compounds. (a) carbon monoxide, CO u…
A: Formula mass is the mass which is obtained from the formula of any compound. Recall the atomic…
Q: Calculate the number of hydrogen atoms in 1.11 mol of acetaldehyde, C2H4O.
A: Introduction: As per the international system of unit (SI), mole is the unit of measurement for the…
Q: Determine the formula weights of each of the following compounds: (a) H2S; (b) NiCO3; (c)…
A: For calculating the formula weight, add the molecular mass of all atoms with the no of atoms present…
Q: The following question refers to the mass spectrum of atom Y, shown below. Mass Spectrum of Atom Y…
A: Atoms are everywhere. They are the building blocks of elements. There are three primary atomic…
Q: A chemist records an ESI accurate mass spectrum of an intermediate during a synthesis. The desired…
A: The protonated ion for C29H31N3O8S is C29H31N3O8SH+
Q: Instru Determine the formula mass/ following compound: 1. Ammonia ( NHa ) 2. Glucose (CoH1206) 3.…
A:
Q: Explain the concept of molecular weight ?
A: Molecular weight can be described as a sum of atomic mass of all the atoms present in a molecule.…
Q: If the mass spectrum of a sample has a 7.7% M+1 peak compared to a 100% M+ peak, how many carbon…
A: A question based on mass spectrum, which is to be accomplished.
Q: Enter the precise mass numbers to the nearest 0.0001 mass unit. Compound A: C4H6O2 Compound B: C4H6S…
A:
Q: b. 26. Identify Which of the following formulas are incorrect? Explain your answer. a. Mg, (SO4)3 b.…
A:
Q: molecular weight is the summation of Atomic numbers Atomics weight Number of molecules
A:
Q: Gallium consists of 60. % 69Ga and 40. % 71Ga. Arsenic has only one naturally occurring isotope.…
A: Gallium has two isotopes 69Ga and 71Ga with ratio of abundant 60 % and 40 % . Therefore intensity…
Q: Calculate the formula mass of the molecule from its structure. IN formula mass: amu N N
A: Formula mass of a molecule is defined as the sum of atomic mass of each atom present in the molecule…
Q: How can mass spectrometry be used to determine?
A: The objective of the question is to explain how the mass spectrometry is useful in determination of…
Q: 27. The elemental analysis of a substance shows that it consists of 52.2% C, 34.7% 0, and 13.0% H by…
A: Since you have posted multiple questions, we are entitled to answer the first only. 27) Given:…
Q: What is the mass percent composition of platinum in the carboplatin, C6H12N2O4Pt? Relative atomic…
A:
Q: Carbon has two stable isotopes, 12C, abundance 98.892%, and 13C, abundance 1.108%. In the mass…
A: The molecular weight peak is called the Molecular Ion peak (M). Since this peak is a radical cation,…
Q: The empirical formula c the mass of each eleme the ratio. This will give gives us the ratio of ato…
A: “Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: What does TIC stand for in mass spec? Also explain what each point in the graph represents.
A: TIC means total ion Chromatogram. the entire range of masses found at all points in the analysis.
Q: One form of asbestos called chrysotile is considered to be a human carcinogen. Mass analysis shows…
A: Given: 26.3% Mg, 20.2% Si, 1.45% H Let mass of compound be 100 g. then it contain 26.3 g of Mg,…
Q: Balance the following equation by filling the gaps with appropriate numbers. КОН + H3PO4 ]KSPO4…
A: According to the Law of conservation of mass " all atoms of different elements must be equal on both…
Q: How many molecular ions (compounds with different weights) would you expect to see in a molecule…
A:
Q: Mass spectrometry is more often applied to molecules than to atoms. We will see in Chapter that the…
A: (a)A mass spectrum is defined as the plot between intensity and m/z (mass-to-charge) ratio that…
Q: 1. An unknown compound containing tellurium and bromine is analyzed and it is determined that 28.53%…
A: Moles is the amount of substance present. It is determined usung molar mass of the compound. Moles…
Q: Given the following mass spec data, what is the molecular formula of the compound? (Show pertinent…
A: We have the mass spectrum data and we need to figure out the molecular formula of the compound.…
Step by step
Solved in 2 steps with 1 images

- a) What is a centroid in mass spectrometry? How is it calculated? b) How does a monoisotopic molecular mass differ from an average molecular mass? c) What are the benefits and limitations of each of these?Both UV/Vis and IR spectrometers can be used for quantitative analysis of analytes. True False1 Experimental results describing a protein's amino acid composition are useful for estimating the molecular weight (MW) of the entire protein. A quantitative amino acid analysis reveals that bovine cytochrome c contains 2% cysteine (Mr 121) by weight. Calculate the approximate molecular weight of bovine cytochrome c if there are 2 cysteine residues. approximate bovine cytochrome c MW: ? Da
- Please Explain and show work clearly step by step thank you! Consider a mixture of the two solids BaCl22H20 (FM 244.26) and KCl (FM 74.551). When the mixture is heated to 160 °C for 1 h, the water of crystallization is driven off. A sample originally weighing 1.7839 g weighed 1.5623 g after heating. Calculate the weight percent of Ba, K, and Cl in the original sample1. Mass spectrometry will give information about molecular mass, chemical formula and structure of the molecule. Select one: True False 2. The strongest peak with 100% abundance in the mass spectrum is known as ........................................... a.Prominent ion peak b.Fragment ion peak c.Molecular ion peak d.Base peak1. what resolution is needed to differentiate the molecular ions of a substance a. CO and C2H4 b. C10H21CHO and C12H26
- Calculate CFSE of K2[Fe(NCS)5]INORGANIC Chemistry Starting product was [(Me5dien)CoCl2] and reacted with NaSCN to produce [(Me5dien)Co(SCN)2] Using the graphs attached, the IR data tells you that the binding mode is N coordination. How does the IR data agree (or disagree) with the peak shifts (Uv-Vis spect) IR Product 1, [(Me5dien)CoCl2] 29041.04, 2812.76, 2761.62 cm-1 (carbon bond) 1457.35 cm-1 (CH2) 1263.91 cm-1 (possibly C-N stretch) Product 2, [(Me5dien)Co(SCN)2] 2968.54-2870.67 cm-1 (C-C) 2059.41 cm-1 (N=C=S) 1470.28 cm-1 (CH2) Uv-vis [(Me5dien)CoCl2] Peak A: 531.8 nm, 0.722 Abs Peak B: 609.7 nm, 0.666 Abs [(Me5dien)Co(SCN)2] Peak A: 510.20 nm, 0.357 Abs Peak B: 608.20 nm, 0.401 AbsDetermine structure. Ignore the 77 ppm in the Carbon 13 NMR. Atomic Mass of structure is 87 g

