In the following image, if we intend to increase the theoretical efficiency of this engine, we can raise TH by AT or by decreasing T by the same AT. Which one of the two scenarios will be more effective?
In the following image, if we intend to increase the theoretical efficiency of this engine, we can raise TH by AT or by decreasing T by the same AT. Which one of the two scenarios will be more effective?
Chapter5: Temperature And Heat
Section: Chapter Questions
Problem 29P: . As a gasoline engine is miming, an amount of gasoline containing 15,000 J of chemical potential...
Related questions
Question
100%
Solved ?
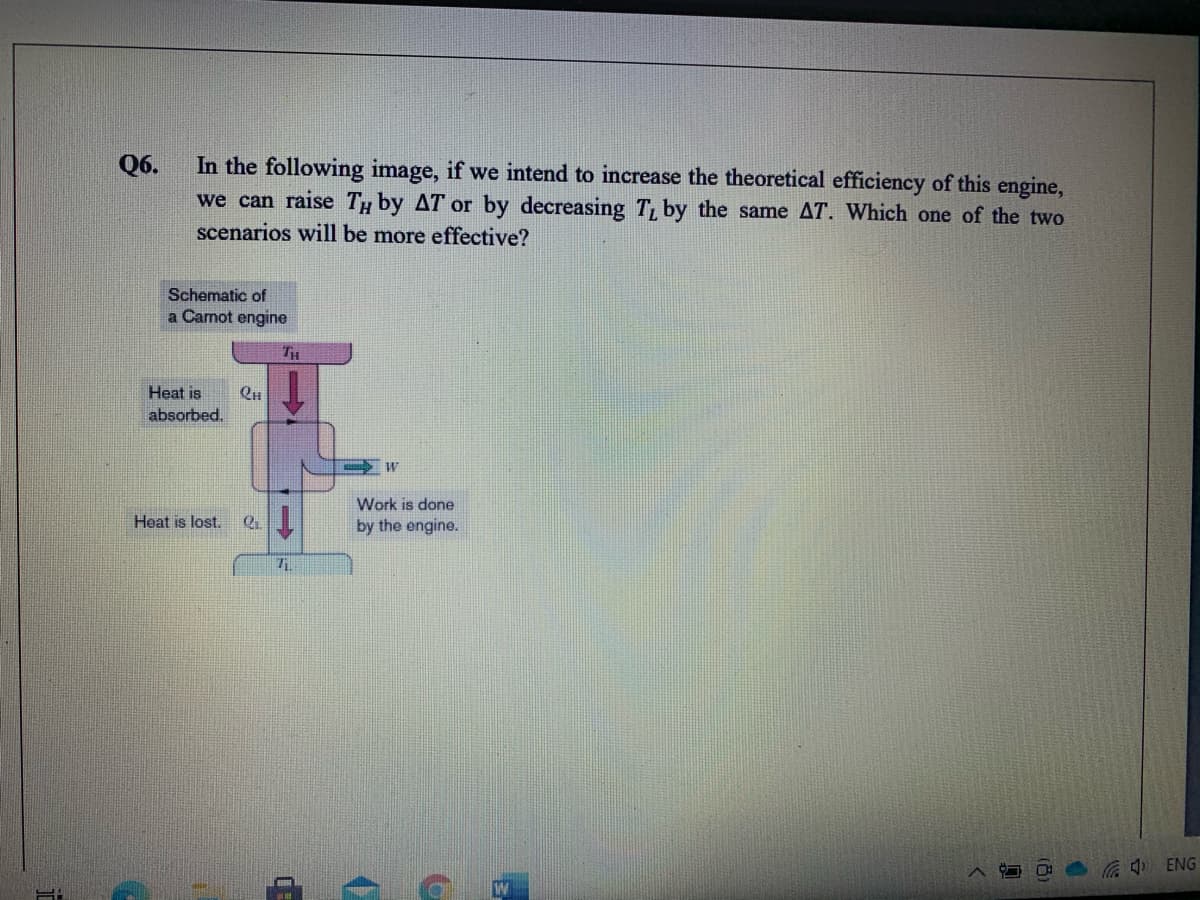
Transcribed Image Text:In the following image, if we intend to increase the theoretical efficiency of this engine,
we can raise Ty by AT or by decreasing T, by the same AT. Which one of the two
scenarios will be more effective?
Q6.
Schematic of
a Carnot engine
TH
Heat is
absorbed.
Work is done
1.
Heat is lost.
by the engine.
A ENG
Expert Solution
Step 1: Carnot heat engine
As we know the efficiency of Carnot heat engine
Efficiency
Now when increase in source then the efficiency becomes
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you



College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College



College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College

College Physics
Physics
ISBN:
9781285737027
Author:
Raymond A. Serway, Chris Vuille
Publisher:
Cengage Learning

Glencoe Physics: Principles and Problems, Student…
Physics
ISBN:
9780078807213
Author:
Paul W. Zitzewitz
Publisher:
Glencoe/McGraw-Hill

Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning