Chapter5: Protein
Section: Chapter Questions
Problem 1SC
Related questions
Question
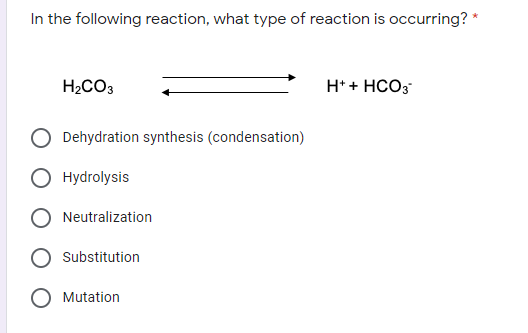
Transcribed Image Text:In the following reaction, what type of reaction is occurring? *
H2CO3
H* + HCO3
Dehydration synthesis (condensation)
Hydrolysis
Neutralization
Substitution
Mutation

Transcribed Image Text:Which of the following molecules are considered to be inorganic?
Carbohydrates and protein
Lipids and minerals
Lipids and proteins
Water and minerals
O None of the above are inorganic
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you


Biology Today and Tomorrow without Physiology (Mi…
Biology
ISBN:
9781305117396
Author:
Cecie Starr, Christine Evers, Lisa Starr
Publisher:
Cengage Learning

Human Biology (MindTap Course List)
Biology
ISBN:
9781305112100
Author:
Cecie Starr, Beverly McMillan
Publisher:
Cengage Learning


Biology Today and Tomorrow without Physiology (Mi…
Biology
ISBN:
9781305117396
Author:
Cecie Starr, Christine Evers, Lisa Starr
Publisher:
Cengage Learning

Human Biology (MindTap Course List)
Biology
ISBN:
9781305112100
Author:
Cecie Starr, Beverly McMillan
Publisher:
Cengage Learning


Concepts of Biology
Biology
ISBN:
9781938168116
Author:
Samantha Fowler, Rebecca Roush, James Wise
Publisher:
OpenStax College

Lifetime Physical Fitness & Wellness
Health & Nutrition
ISBN:
9781337677509
Author:
HOEGER
Publisher:
Cengage